+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1a49 | ||||||
|---|---|---|---|---|---|---|---|
| Title | BIS MG-ATP-K-OXALATE COMPLEX OF PYRUVATE KINASE | ||||||
 Components Components | PYRUVATE KINASE | ||||||
 Keywords Keywords | TRANSFERASE / PYRUVATE KINASE / RABBIT MUSCLE / DOMAIN MOVEMENT / POTASSIUM BINDING | ||||||
| Function / homology |  Function and homology information Function and homology informationpyruvate kinase / pyruvate kinase activity / positive regulation of cytoplasmic translation / positive regulation of sprouting angiogenesis / potassium ion binding / rough endoplasmic reticulum / non-specific protein-tyrosine kinase / non-membrane spanning protein tyrosine kinase activity / non-specific serine/threonine protein kinase / protein serine/threonine kinase activity ...pyruvate kinase / pyruvate kinase activity / positive regulation of cytoplasmic translation / positive regulation of sprouting angiogenesis / potassium ion binding / rough endoplasmic reticulum / non-specific protein-tyrosine kinase / non-membrane spanning protein tyrosine kinase activity / non-specific serine/threonine protein kinase / protein serine/threonine kinase activity / mRNA binding / magnesium ion binding / ATP binding / nucleus Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.1 Å MOLECULAR REPLACEMENT / Resolution: 2.1 Å | ||||||
 Authors Authors | Larsen, T.M. / Benning, M.M. / Rayment, I. / Reed, G.H. | ||||||
 Citation Citation |  Journal: Biochemistry / Year: 1998 Journal: Biochemistry / Year: 1998Title: Structure of the bis(Mg2+)-ATP-oxalate complex of the rabbit muscle pyruvate kinase at 2.1 A resolution: ATP binding over a barrel. Authors: Larsen, T.M. / Benning, M.M. / Rayment, I. / Reed, G.H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1a49.cif.gz 1a49.cif.gz | 852.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1a49.ent.gz pdb1a49.ent.gz | 684.9 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1a49.json.gz 1a49.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/a4/1a49 https://data.pdbj.org/pub/pdb/validation_reports/a4/1a49 ftp://data.pdbj.org/pub/pdb/validation_reports/a4/1a49 ftp://data.pdbj.org/pub/pdb/validation_reports/a4/1a49 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1a5uC  1aqfS S: Starting model for refinement C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 8 molecules ABCDEFGH
| #1: Protein | Mass: 57998.820 Da / Num. of mol.: 8 / Source method: isolated from a natural source / Source: (natural)  |
|---|
-Non-polymers , 5 types, 1957 molecules 
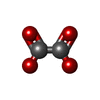







| #2: Chemical | ChemComp-K / #3: Chemical | ChemComp-OXL / #4: Chemical | ChemComp-MG / #5: Chemical | ChemComp-ATP / #6: Water | ChemComp-HOH / | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.74 Å3/Da / Density % sol: 57.2 % | ||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Crystal grow | pH: 6 / Details: pH 6.0 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: batch method | ||||||||||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 113 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SSRL SSRL  / Beamline: BL7-1 / Beamline: BL7-1 |
| Detector | Date: Aug 1, 1996 |
| Radiation | Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Relative weight: 1 |
| Reflection | Resolution: 2.1→30 Å / Num. obs: 267793 / % possible obs: 89 % |
| Reflection shell | Resolution: 2.1→2.18 Å / Rsym value: 0.198 / % possible all: 78 |
| Reflection | *PLUS Num. all: 301829 / Num. measured all: 815369 / Rmerge(I) obs: 0.047 |
| Reflection shell | *PLUS % possible obs: 78 % / Rmerge(I) obs: 0.212 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure:  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: PDB ENTRY 1AQF Resolution: 2.1→30 Å /
| ||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.1→30 Å
| ||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||
| Software | *PLUS Name: TNT / Version: 5D / Classification: refinement | ||||||||||||||||||||||||||||||
| Refinement | *PLUS Rfactor obs: 0.198 | ||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | ||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller













 PDBj
PDBj




