+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9757 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | pCBH ParM filament | ||||||||||||
 Map data Map data | Whole map of a ParM filament from pCBH of Clostridium botulinum | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | ParM / filaments / cytoskeleton / PROTEIN FIBRIL | ||||||||||||
| Function / homology | ParM-like / Actin-like protein, N-terminal / Actin like proteins N terminal domain / ATPase, nucleotide binding domain / Actin-like protein N-terminal domain-containing protein Function and homology information Function and homology information | ||||||||||||
| Biological species |  Clostridium botulinum F str. 230613 (bacteria) / Clostridium botulinum F str. 230613 (bacteria) /  Clostridium botulinum Prevot_594 (bacteria) Clostridium botulinum Prevot_594 (bacteria) | ||||||||||||
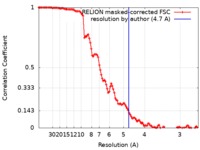
| Method | helical reconstruction / cryo EM / Resolution: 4.7 Å | ||||||||||||
 Authors Authors | Koh F / Narita A | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2019 Journal: Nat Commun / Year: 2019Title: The structure of a 15-stranded actin-like filament from Clostridium botulinum. Authors: Fujiet Koh / Akihiro Narita / Lin Jie Lee / Kotaro Tanaka / Yong Zi Tan / Venkata P Dandey / David Popp / Robert C Robinson /     Abstract: Microfilaments (actin) and microtubules represent the extremes in eukaryotic cytoskeleton cross-sectional dimensions, raising the question of whether filament architectures are limited by protein ...Microfilaments (actin) and microtubules represent the extremes in eukaryotic cytoskeleton cross-sectional dimensions, raising the question of whether filament architectures are limited by protein fold. Here, we report the cryoelectron microscopy structure of a complex filament formed from 15 protofilaments of an actin-like protein. This actin-like ParM is encoded on the large pCBH Clostridium botulinum plasmid. In cross-section, the ~26 nm diameter filament comprises a central helical protofilament surrounded by intermediate and outer layers of six and eight twisted protofilaments, respectively. Alternating polarity of the layers allows for similar lateral contacts between each layer. This filament design is stiffer than the actin filament, and has likely been selected for during evolution to move large cargos. The comparable sizes of microtubule and pCBH ParM filaments indicate that larger filament architectures are not limited by the protomer fold. Instead, function appears to have been the evolutionary driving force to produce broad, complex filaments. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9757.map.gz emd_9757.map.gz | 15 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9757-v30.xml emd-9757-v30.xml emd-9757.xml emd-9757.xml | 11.3 KB 11.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_9757_fsc.xml emd_9757_fsc.xml | 11.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_9757.png emd_9757.png | 325.6 KB | ||
| Filedesc metadata |  emd-9757.cif.gz emd-9757.cif.gz | 5.7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9757 http://ftp.pdbj.org/pub/emdb/structures/EMD-9757 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9757 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9757 | HTTPS FTP |
-Related structure data
| Related structure data |  6izrMC  9758C  6ixwC  6izvC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9757.map.gz / Format: CCP4 / Size: 35.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9757.map.gz / Format: CCP4 / Size: 35.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Whole map of a ParM filament from pCBH of Clostridium botulinum | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.331 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : ParM filament coded on pCBH plasmid from Clostridium botulinum
| Entire | Name: ParM filament coded on pCBH plasmid from Clostridium botulinum |
|---|---|
| Components |
|
-Supramolecule #1: ParM filament coded on pCBH plasmid from Clostridium botulinum
| Supramolecule | Name: ParM filament coded on pCBH plasmid from Clostridium botulinum type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Clostridium botulinum F str. 230613 (bacteria) / Organelle: pCBH plasmid Clostridium botulinum F str. 230613 (bacteria) / Organelle: pCBH plasmid |
-Macromolecule #1: Putative plasmid segregation protein ParM
| Macromolecule | Name: Putative plasmid segregation protein ParM / type: protein_or_peptide / ID: 1 / Number of copies: 30 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Clostridium botulinum Prevot_594 (bacteria) Clostridium botulinum Prevot_594 (bacteria) |
| Molecular weight | Theoretical: 39.560168 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MNKYTIAIDL GYGQIKGINQ DNKRVIFPSI ISSGKDRSLD TFFNSIDNIV DNIHVKILDE YFNEKEYFVG ELAKRQPSNS SFINRDNKI NSEENKVLLA TALGLLIPND LPNDTKIHIV TGLPLEHFIK QKQALNDMLK DFEHTIKFVD HNFSRNIKFE E SNITLFPQ ...String: MNKYTIAIDL GYGQIKGINQ DNKRVIFPSI ISSGKDRSLD TFFNSIDNIV DNIHVKILDE YFNEKEYFVG ELAKRQPSNS SFINRDNKI NSEENKVLLA TALGLLIPND LPNDTKIHIV TGLPLEHFIK QKQALNDMLK DFEHTIKFVD HNFSRNIKFE E SNITLFPQ GAGAIFSKIN NDISSLLIKE TFIGLIDVGF KTTDIVVFRI NKDKEPVFEQ EMSATLDGLG MINIYNTMDK AF TDNSRDG SKLNTEQLML LCEEGKIFFK GDYIDLKKDL IKARKTLSTN IINKADGLWG SRKNSFNSIM IAGGGGKVLY NHL KLIEPN MCQLIDNPEF ANAIGYLEFG KQF UniProtKB: Actin-like protein N-terminal domain-containing protein |
-Macromolecule #2: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 30 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 30 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 0.8 mg/mL |
|---|---|
| Buffer | pH: 7.4 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: MOLYBDENUM / Mesh: 400 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 30.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller









 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)