[English] 日本語
 Yorodumi
Yorodumi- EMDB-9712: Negative stain electron microscopy of the actin/Arp-Nucleosome as... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9712 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Negative stain electron microscopy of the actin/Arp-Nucleosome assembly state II. | |||||||||
 Map data Map data | 3D reconstruction of the actin/Arp-Nucleosome assembly state II. | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 27.493 Å | |||||||||
 Authors Authors | Zhang X / Cai G | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: J Mol Cell Biol / Year: 2019 Journal: J Mol Cell Biol / Year: 2019Title: Structure and functional interactions of INO80 actin/Arp module. Authors: Xuan Zhang / Xuejuan Wang / Zhihui Zhang / Gang Cai /  Abstract: The presence and functions of nuclear actin have been controversial due to the lack of molecular mechanisms. Nuclear actin and actin-related proteins (Arps) are subunits of several chromatin ...The presence and functions of nuclear actin have been controversial due to the lack of molecular mechanisms. Nuclear actin and actin-related proteins (Arps) are subunits of several chromatin remodelers, including the evolutionarily conserved INO80 chromatin-remodeling complex. Here, we present an improved cryo-EM structure of the yeast INO80 complex and the first 3D reconstruction of the INO80 actin/Arp module. The modular and subunit architecture is defined using a combination of subunit deletion analysis and published crosslinking-mass spectrometry. The functional interactions of the INO80 actin/Arp module with a nucleosome is 3D EM reconstructed in two different binding states. Nucleosomes initially bind to the Arp8 subunit and the substantial conformational changes maximize nucleosome contacts of the actin/Arp module, which could promote the bound nucleosome to be engaged onto the INO80 ATPase domain. Our findings suggest that the conserved nuclear actin/Arp module acts a conformational switch of the INO80 for nucleosome binding. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9712.map.gz emd_9712.map.gz | 6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9712-v30.xml emd-9712-v30.xml emd-9712.xml emd-9712.xml | 8.4 KB 8.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9712.png emd_9712.png | 31.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9712 http://ftp.pdbj.org/pub/emdb/structures/EMD-9712 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9712 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9712 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9712.map.gz / Format: CCP4 / Size: 6.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9712.map.gz / Format: CCP4 / Size: 6.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 3D reconstruction of the actin/Arp-Nucleosome assembly state II. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 3.54 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
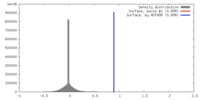
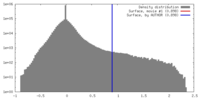
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : actin/Arp-Nucleosome assembly
| Entire | Name: actin/Arp-Nucleosome assembly |
|---|---|
| Components |
|
-Supramolecule #1: actin/Arp-Nucleosome assembly
| Supramolecule | Name: actin/Arp-Nucleosome assembly / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 Details: Negative stain electron microscopy of the actin/Arp-Nucleosome assembly state II. |
|---|---|
| Source (natural) | Organism:  |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | cell |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Staining | Type: NEGATIVE / Material: Uranyl Formate |
| Vitrification | Cryogen name: OTHER |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F20 |
|---|---|
| Image recording | Film or detector model: FEI EAGLE (2k x 2k) / Average electron dose: 36.0 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Tecnai F20 / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 27.493 Å / Resolution method: FSC 0.5 CUT-OFF / Number images used: 1157 |
|---|---|
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)