[English] 日本語
 Yorodumi
Yorodumi- EMDB-9341: Structure of the type VI secretion system TssK-TssF-TssG baseplat... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-9341 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the type VI secretion system TssK-TssF-TssG baseplate subcomplex revealed by cryo-electron microscopy - full map sharpened | |||||||||
 Map data Map data | Type VI secretion system TssK-TssF-TssG baseplate subcomplex, sharpened map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Type VI secretion system / enteroaggregative E. coli / protein secretion / baseplate / Structural Genomics / Seattle Structural Genomics Center for Infectious Disease / SSGCID / PROTEIN TRANSPORT | |||||||||
| Function / homology | Type VI secretion system TssF / Type VI secretion system, TssF / Type VI secretion, TssG-like / Type VI secretion, TssG / Bacterial Type VI secretion, VC_A0110, EvfL, ImpJ, VasE / Type VI secretion system TssK / Type VI secretion protein / Type VI secretion protein / Type VI secretion protein Function and homology information Function and homology information | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.7 Å | |||||||||
 Authors Authors | Park YJ / Lacourse KD / Cambillau C / Seattle Structural Genomics Center for Infectious Disease (SSGCID) / DiMaio F / Mougous JD / Veesler D | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2018 Journal: Nat Commun / Year: 2018Title: Structure of the type VI secretion system TssK-TssF-TssG baseplate subcomplex revealed by cryo-electron microscopy. Authors: Young-Jun Park / Kaitlyn D Lacourse / Christian Cambillau / Frank DiMaio / Joseph D Mougous / David Veesler /   Abstract: Type VI secretion systems (T6SSs) translocate effectors into target cells and are made of a contractile sheath and a tube docked onto a multi-protein transmembrane complex via a baseplate. Although ...Type VI secretion systems (T6SSs) translocate effectors into target cells and are made of a contractile sheath and a tube docked onto a multi-protein transmembrane complex via a baseplate. Although some information is available about the mechanisms of tail contraction leading to effector delivery, the detailed architecture and function of the baseplate remain unknown. Here, we report the 3.7 Å resolution cryo-electron microscopy reconstruction of an enteroaggregative Escherichia coli baseplate subcomplex assembled from TssK, TssF and TssG. The structure reveals two TssK trimers interact with a locally pseudo-3-fold symmetrical complex comprising two copies of TssF and one copy of TssG. TssF and TssG are structurally related to each other and to components of the phage T4 baseplate and of the type IV secretion system, strengthening the evolutionary relationships among these macromolecular machines. These results, together with bacterial two-hybrid assays, provide a structural framework to understand the T6SS baseplate architecture. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_9341.map.gz emd_9341.map.gz | 6.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-9341-v30.xml emd-9341-v30.xml emd-9341.xml emd-9341.xml | 17.6 KB 17.6 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_9341.png emd_9341.png | 142.9 KB | ||
| Filedesc metadata |  emd-9341.cif.gz emd-9341.cif.gz | 6.5 KB | ||
| Others |  emd_9341_additional.map.gz emd_9341_additional.map.gz | 71.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-9341 http://ftp.pdbj.org/pub/emdb/structures/EMD-9341 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9341 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-9341 | HTTPS FTP |
-Related structure data
| Related structure data |  6n38MC  9342C  9343C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_9341.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_9341.map.gz / Format: CCP4 / Size: 91.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Type VI secretion system TssK-TssF-TssG baseplate subcomplex, sharpened map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.37 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Type VI secretion system TssK-TssF-TssG baseplate subcomplex, unsharpened...
| File | emd_9341_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Type VI secretion system TssK-TssF-TssG baseplate subcomplex, unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
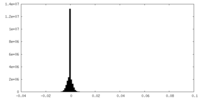
| Density Histograms |
- Sample components
Sample components
-Entire : TssK-TssF-TssG T6SS baseplate subcomplex
| Entire | Name: TssK-TssF-TssG T6SS baseplate subcomplex |
|---|---|
| Components |
|
-Supramolecule #1: TssK-TssF-TssG T6SS baseplate subcomplex
| Supramolecule | Name: TssK-TssF-TssG T6SS baseplate subcomplex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Putative type VI secretion protein
| Macromolecule | Name: Putative type VI secretion protein / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Strain: 042 / EAEC |
| Molecular weight | Theoretical: 66.557125 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MDDLTLRYFD AEMRYLREAG KAFAQAHPDR AAMLDLDKAG TPDPCVERLF EGFAFSMGRL RQKIDDDLPE LTESLVSMLW PHYLRTIPS LSVVALTPRL SVMKMAETVP AGLEVTSRPV GPGNTVCRYR TTRAIPLNPL AVEKVVMTTE PDGRSVLKIG F ACSELADW ...String: MDDLTLRYFD AEMRYLREAG KAFAQAHPDR AAMLDLDKAG TPDPCVERLF EGFAFSMGRL RQKIDDDLPE LTESLVSMLW PHYLRTIPS LSVVALTPRL SVMKMAETVP AGLEVTSRPV GPGNTVCRYR TTRAIPLNPL AVEKVVMTTE PDGRSVLKIG F ACSELADW SQVDLHRLSL YLAAEAPVSS TLHLMMTKRL AALYLRLPGN DERIRIDGWF SPGGFAEEDR LWPKGDSAFS GY QLLLEYF TFREKFMFVH LNGLENVSLP AGISGFDLEV VLSQPWPADL PVTDDALCLH CVPVINLFTL EADPLIINGL ESE YLLRPK RLQDGYTEIY SVDAVTGSGR TGSAEYVPFT SFRHRGGMLR HDAPERYYHT RVKRGVTGMY DTWLILGGQR WEAD RMPER ETLSLRITGT NGQLPRRALQ STLLDRCEQV LQAPVSVRNL CKPTLPVYPP TEDRFHWRVM SHLGTGFLNM LSSAE VLRG TLALYNWRDD ELNHRRLDAI LAVQHHRIQR FEKGFLLRGL DVEVTLDGNG FAGEGDIHLF GEMLNRFLAL YADMNQ FNQ LTLIVQPEGK CIRWKENHNP RLPG UniProtKB: Type VI secretion protein |
-Macromolecule #2: Putative type VI secretion protein
| Macromolecule | Name: Putative type VI secretion protein / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Strain: 042 / EAEC |
| Molecular weight | Theoretical: 33.787875 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGFPASEIKD AVIPEESHLP PIVHVTFMGL YGVTSPLPAH YISDIAQQRE GHEAAADFLD IFSHRLITQY YRIWRKYSYP ATFEAGGQD KTSQYLLGLA RLGIPGCAQN IATPVSRFLA LLPLMLLPGR TAEGLTSLVT LLAPGTQARV WHHDRRRIPL K TPLTMRVH ...String: MGFPASEIKD AVIPEESHLP PIVHVTFMGL YGVTSPLPAH YISDIAQQRE GHEAAADFLD IFSHRLITQY YRIWRKYSYP ATFEAGGQD KTSQYLLGLA RLGIPGCAQN IATPVSRFLA LLPLMLLPGR TAEGLTSLVT LLAPGTQARV WHHDRRRIPL K TPLTMRVH HPVSLKSRPV MGDHATDVNG QVLLQLSTQT GSEVQGWLPG GHLYSDLLAL LHVYLGSRLD VRLQLCVERS LL PDARLSC RPAAGSPQLG RTAVMRTQAK IATSAARVMT ISLGRYQRVQ EHYQRKETQE NGDYRW UniProtKB: Type VI secretion protein |
-Macromolecule #3: Unassigned protein
| Macromolecule | Name: Unassigned protein / type: protein_or_peptide / ID: 3 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Strain: strain 042 / EAEC |
| Molecular weight | Theoretical: 1.805216 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: (UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK)(UNK)(UNK)(UNK) (UNK)(UNK)(UNK) (UNK)(UNK) |
-Macromolecule #4: Putative type VI secretion protein
| Macromolecule | Name: Putative type VI secretion protein / type: protein_or_peptide / ID: 4 / Number of copies: 6 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Strain: 042 / EAEC |
| Molecular weight | Theoretical: 36.57768 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKIYRPLWED GAFLMPQQFQ QQAAWDVHLA DSVARMGLAH PWGVVAAEFD DSLLPLSRLN ATRLIVRFPD GTLIDTERAD NLPPVCDLS TVSDRSLVDI VLALPLLNAN GGNLDNGSES ERPRRWKSER VNVQELAGHE QSEVAVLRHN LTLRMAHQEN A AWLTCPVT ...String: MKIYRPLWED GAFLMPQQFQ QQAAWDVHLA DSVARMGLAH PWGVVAAEFD DSLLPLSRLN ATRLIVRFPD GTLIDTERAD NLPPVCDLS TVSDRSLVDI VLALPLLNAN GGNLDNGSES ERPRRWKSER VNVQELAGHE QSEVAVLRHN LTLRMAHQEN A AWLTCPVT RLVRDAQGQW CRDPRFIPPL LTLSASPSLM TELAELLHHL QARRQRLMSM RRENNARLAD FAVADVSLFW LL NALNSAE PVLKELLDMP YRHPELLYRE LARLAGSLLT FSLEHNVDAV PAYHHETPEN VFPPLLSLLN RLLEASLPSH HHH HH UniProtKB: Type VI secretion protein |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.12 mg/mL |
|---|---|
| Buffer | pH: 7.8 |
| Grid | Model: C-flat-1.2/1.3 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: CONTINUOUS / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER Details: An initial model was obtained using cryosparc abinitio. |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.7 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 36496 |
| Initial angle assignment | Type: PROJECTION MATCHING |
| Final angle assignment | Type: PROJECTION MATCHING / Software - Name: RELION (ver. 3.0) |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)