[English] 日本語
 Yorodumi
Yorodumi- EMDB-7971: cryo-EM structure of the mitochondrial calcium uniporter MCU from... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7971 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | cryo-EM structure of the mitochondrial calcium uniporter MCU from the fungus Cyphellophora europaea | |||||||||
 Map data Map data | Cryo-EM structure of the mitochondrial calcium uniporter | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | mitochondria / calcium / ion channel / eukaryotic / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationuniporter activity / uniplex complex / mitochondrial calcium ion homeostasis / calcium import into the mitochondrion / calcium channel activity / protein homotetramerization / mitochondrial inner membrane / metal ion binding / identical protein binding Similarity search - Function | |||||||||
| Biological species |  Cyphellophora europaea (fungus) / Cyphellophora europaea (fungus) /  Cyphellophora europaea CBS 101466 (fungus) Cyphellophora europaea CBS 101466 (fungus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | |||||||||
 Authors Authors | Long SB / Baradaran R | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Cryo-EM structures of fungal and metazoan mitochondrial calcium uniporters. Authors: Rozbeh Baradaran / Chongyuan Wang / Andrew Francis Siliciano / Stephen Barstow Long /  Abstract: The mitochondrial calcium uniporter (MCU) is a highly selective calcium channel and a major route of calcium entry into mitochondria. How the channel catalyses ion permeation and achieves ...The mitochondrial calcium uniporter (MCU) is a highly selective calcium channel and a major route of calcium entry into mitochondria. How the channel catalyses ion permeation and achieves ion selectivity are not well understood, partly because MCU is thought to have a distinct architecture in comparison to other cellular channels. Here we report cryo-electron microscopy reconstructions of MCU channels from zebrafish and Cyphellophora europaea at 8.5 Å and 3.2 Å resolutions, respectively. In contrast to a previous report of pentameric stoichiometry for MCU, both channels are tetramers. The atomic model of C. europaea MCU shows that a conserved WDXXEP signature sequence forms the selectivity filter, in which calcium ions are arranged in single file. Coiled-coil legs connect the pore to N-terminal domains in the mitochondrial matrix. In C. europaea MCU, the N-terminal domains assemble as a dimer of dimers; in zebrafish MCU, they form an asymmetric crescent. The structures define principles that underlie ion permeation and calcium selectivity in this unusual channel. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7971.map.gz emd_7971.map.gz | 200.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7971-v30.xml emd-7971-v30.xml emd-7971.xml emd-7971.xml | 16 KB 16 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7971.png emd_7971.png | 115.1 KB | ||
| Filedesc metadata |  emd-7971.cif.gz emd-7971.cif.gz | 6.3 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7971 http://ftp.pdbj.org/pub/emdb/structures/EMD-7971 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7971 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7971 | HTTPS FTP |
-Related structure data
| Related structure data |  6dnfMC  7972C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_7971.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7971.map.gz / Format: CCP4 / Size: 216 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Cryo-EM structure of the mitochondrial calcium uniporter | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.861 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
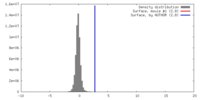
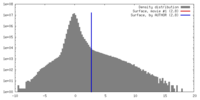
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Mitochondrial Calcium Uniporter (MCU) from the fungus Cyphellopho...
| Entire | Name: Mitochondrial Calcium Uniporter (MCU) from the fungus Cyphellophora europaea. |
|---|---|
| Components |
|
-Supramolecule #1: Mitochondrial Calcium Uniporter (MCU) from the fungus Cyphellopho...
| Supramolecule | Name: Mitochondrial Calcium Uniporter (MCU) from the fungus Cyphellophora europaea. type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Cyphellophora europaea (fungus) Cyphellophora europaea (fungus) |
| Molecular weight | Theoretical: 158.328 KDa |
-Macromolecule #1: Mitochondrial calcium uniporter MCU
| Macromolecule | Name: Mitochondrial calcium uniporter MCU / type: protein_or_peptide / ID: 1 / Number of copies: 4 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Cyphellophora europaea CBS 101466 (fungus) Cyphellophora europaea CBS 101466 (fungus) |
| Molecular weight | Theoretical: 39.637176 KDa |
| Recombinant expression | Organism:  Komagataella pastoris (fungus) Komagataella pastoris (fungus) |
| Sequence | String: MTKGKLLTTP SRLLKLVLPL STVDHNTDRK DVAPLALLVH PQQPLSYLER LIQAELPPPD PQDSKSTTRS VTFRAMEAKD DEIKPRKKA DTEGGGGSDG SVQSYSGAGR EGEGKDEGEF VRWSPSTEIG DFIRDAARAK EFEVEIEGSP GVIKVAVPSF N DRTYYLRQ ...String: MTKGKLLTTP SRLLKLVLPL STVDHNTDRK DVAPLALLVH PQQPLSYLER LIQAELPPPD PQDSKSTTRS VTFRAMEAKD DEIKPRKKA DTEGGGGSDG SVQSYSGAGR EGEGKDEGEF VRWSPSTEIG DFIRDAARAK EFEVEIEGSP GVIKVAVPSF N DRTYYLRQ RLRRTSRKIS KLAAIKEECD KAAHRGAQRI ALAGCGGLIG YWYIVYRLTF ETDLGWDVME PVTYLVGLST LI GGYMWFL WHNREVSYRS ALNITVSARQ NKLYQAKGFS LQDWEGYLEE ANAMRREIKA VASEYDVDWN ETQDEGGDEK VTK ALRDER KNNNGTKNKS KEGEEDDEDD GEGEEF UniProtKB: Calcium uniporter protein, mitochondrial |
-Macromolecule #2: 1-[GLYCEROLYLPHOSPHONYL]-2-[8-(2-HEXYL-CYCLOPROPYL)-OCTANAL-1-YL]...
| Macromolecule | Name: 1-[GLYCEROLYLPHOSPHONYL]-2-[8-(2-HEXYL-CYCLOPROPYL)-OCTANAL-1-YL]-3-[HEXADECANAL-1-YL]-GLYCEROL type: ligand / ID: 2 / Number of copies: 8 / Formula: DGG |
|---|---|
| Molecular weight | Theoretical: 734.981 Da |
| Chemical component information | 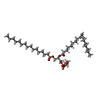 ChemComp-DGG: |
-Macromolecule #3: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 3 / Number of copies: 3 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 5 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.5 Component:
| ||||||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY ARRAY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 10 sec. / Pretreatment - Atmosphere: AIR | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 298 K / Instrument: FEI VITROBOT MARK IV / Details: 2 second blot, blot force of 0. | ||||||||||||||||||
| Details | Monodisperse sample |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-40 / Number grids imaged: 3 / Number real images: 6040 / Average exposure time: 0.2 sec. / Average electron dose: 2.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 29000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL / Overall B value: 136.58 / Target criteria: Correlation coefficient |
|---|---|
| Output model |  PDB-6dnf: |
 Movie
Movie Controller
Controller













 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)