[English] 日本語
 Yorodumi
Yorodumi- EMDB-7435: Cryo-EM structure of mouse TPC1 channel in the PtdIns(3,5)P2-boun... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7435 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of mouse TPC1 channel in the PtdIns(3,5)P2-bound state | ||||||||||||||||||
 Map data Map data | TPC1 channel in the PtdIns(3,5)P2-bound state | ||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||
 Keywords Keywords | Ion channel / MEMBRANE PROTEIN | ||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationvoltage-gated channel activity / endolysosome / intracellularly phosphatidylinositol-3,5-bisphosphate-gated monatomic cation channel activity / NAADP-sensitive calcium-release channel activity / Stimuli-sensing channels / phosphatidylinositol-3,5-bisphosphate binding / syntaxin binding / voltage-gated sodium channel activity / ligand-gated sodium channel activity / monoatomic ion channel complex ...voltage-gated channel activity / endolysosome / intracellularly phosphatidylinositol-3,5-bisphosphate-gated monatomic cation channel activity / NAADP-sensitive calcium-release channel activity / Stimuli-sensing channels / phosphatidylinositol-3,5-bisphosphate binding / syntaxin binding / voltage-gated sodium channel activity / ligand-gated sodium channel activity / monoatomic ion channel complex / positive regulation of autophagy / sodium ion transmembrane transport / recycling endosome membrane / early endosome membrane / endosome membrane / endocytosis involved in viral entry into host cell / lysosomal membrane / protein homodimerization activity / membrane / identical protein binding Similarity search - Function | ||||||||||||||||||
| Biological species |  | ||||||||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.2 Å | ||||||||||||||||||
 Authors Authors | She J / Guo J | ||||||||||||||||||
| Funding support |  United States, 5 items United States, 5 items
| ||||||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Structural insights into the voltage and phospholipid activation of the mammalian TPC1 channel. Authors: Ji She / Jiangtao Guo / Qingfeng Chen / Weizhong Zeng / Youxing Jiang / Xiao-Chen Bai /   Abstract: The organellar two-pore channel (TPC) functions as a homodimer, in which each subunit contains two homologous Shaker-like six-transmembrane (6-TM)-domain repeats. TPCs belong to the voltage-gated ion ...The organellar two-pore channel (TPC) functions as a homodimer, in which each subunit contains two homologous Shaker-like six-transmembrane (6-TM)-domain repeats. TPCs belong to the voltage-gated ion channel superfamily and are ubiquitously expressed in animals and plants. Mammalian TPC1 and TPC2 are localized at the endolysosomal membrane, and have critical roles in regulating the physiological functions of these acidic organelles. Here we present electron cryo-microscopy structures of mouse TPC1 (MmTPC1)-a voltage-dependent, phosphatidylinositol 3,5-bisphosphate (PtdIns(3,5)P)-activated Na-selective channel-in both the apo closed state and ligand-bound open state. Combined with functional analysis, these structures provide comprehensive structural insights into the selectivity and gating mechanisms of mammalian TPC channels. The channel has a coin-slot-shaped ion pathway in the filter that defines the selectivity of mammalian TPCs. Only the voltage-sensing domain from the second 6-TM domain confers voltage dependence on MmTPC1. Endolysosome-specific PtdIns(3,5)P binds to the first 6-TM domain and activates the channel under conditions of depolarizing membrane potential. Structural comparisons between the apo and PtdIns(3,5)P-bound structures show the interplay between voltage and ligand in channel activation. These MmTPC1 structures reveal lipid binding and regulation in a 6-TM voltage-gated channel, which is of interest in light of the emerging recognition of the importance of phosphoinositide regulation of ion channels. | ||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7435.map.gz emd_7435.map.gz | 48.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7435-v30.xml emd-7435-v30.xml emd-7435.xml emd-7435.xml | 19.9 KB 19.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7435.png emd_7435.png | 94.6 KB | ||
| Filedesc metadata |  emd-7435.cif.gz emd-7435.cif.gz | 7 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7435 http://ftp.pdbj.org/pub/emdb/structures/EMD-7435 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7435 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7435 | HTTPS FTP |
-Related structure data
| Related structure data |  6c9aMC  7434C  6c96C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7435.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7435.map.gz / Format: CCP4 / Size: 52.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | TPC1 channel in the PtdIns(3,5)P2-bound state | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
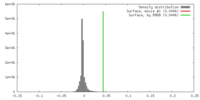
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : PtdIns(3,5)P2-bound mouse TPC1
| Entire | Name: PtdIns(3,5)P2-bound mouse TPC1 |
|---|---|
| Components |
|
-Supramolecule #1: PtdIns(3,5)P2-bound mouse TPC1
| Supramolecule | Name: PtdIns(3,5)P2-bound mouse TPC1 / type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Two pore calcium channel protein 1
| Macromolecule | Name: Two pore calcium channel protein 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 95.414758 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAVSLDDDVP LILTLDEAES APLPPSNSLG QEQLPSKNGG SHSIHNSQVP SLVSGADSPP SSPTGHNWEM NYQEAAIYLQ EGQNNDKFF THPKDARALA AYLFVHNHFF YMMELLTALL LLLLSLCESP AVPVLKLHTY VHATLELFAL MVVVFELCMK L RWLGFHTF ...String: MAVSLDDDVP LILTLDEAES APLPPSNSLG QEQLPSKNGG SHSIHNSQVP SLVSGADSPP SSPTGHNWEM NYQEAAIYLQ EGQNNDKFF THPKDARALA AYLFVHNHFF YMMELLTALL LLLLSLCESP AVPVLKLHTY VHATLELFAL MVVVFELCMK L RWLGFHTF VRHKRTMVKT SVLVVQFIEA IVVLVRQTSH VRVTRALRCI FLVDCRYCGG VRRNLRQIFQ SLPPFMDILL LL LFFMIIF AILGFYLFST NPSDPYFSTL ENSIVNLFVL LTTANFPDVM MPSYSRNPWS CVFFIVYLSI ELYFIMNLLL AVV FDTFND IEKHKFKSLL LHKRTAIQHA YGLLASQRRP AGISYRQFEG LMRFYKPRMS ARERFLTFKA LNQSNTPLLS LKDF YDIYE VAALQWKAKR NRQHWFDELP RTAFLIFKGI NILVNSKAFQ YFMYLVVAVN GVWILVETFM LKGGNFTSKH VPWSY LVFL TIYGVELFMK VAGLGPVEYL SSGWNLFDFS VTAFAFLGLL ALTLNMEPFY FIVVLRPLQL LRLFKLKKRY RNVLDT MFE LLPRMASLGL TLLTFYYSFA IVGMEFFNGR LTPNCCNTST VADAYRFINH TVGNKTKVEE GYYYLNNFDN ILNSFVT LF ELTVVNNWYI IMEGVTSQTS HWSRLYFMTF YIVTMVVMTI IVAFILEAFV FRMNYSRKSQ DSEVDSGIVI EKEMSKEE L MAVLELYREE RGTSSDVTRL LDTLSQMEKY QQNSMVFLGR RSRTKSDLSL KMYQEEIQEW YEEHAREQEQ QKLRGSVPG PAAQQPPGSR QRSQTVTVEA GLVPR UniProtKB: Two pore calcium channel protein 1 |
-Macromolecule #3: (2R)-3-{[(S)-hydroxy{[(1S,2R,3R,4S,5S,6R)-2,4,6-trihydroxy-3,5-bi...
| Macromolecule | Name: (2R)-3-{[(S)-hydroxy{[(1S,2R,3R,4S,5S,6R)-2,4,6-trihydroxy-3,5-bis(phosphonooxy)cyclohexyl]oxy}phosphoryl]oxy}propane-1,2-diyl dioctanoate type: ligand / ID: 3 / Number of copies: 2 / Formula: EUJ |
|---|---|
| Molecular weight | Theoretical: 746.566 Da |
| Chemical component information | 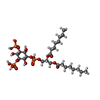 ChemComp-EUJ: |
-Macromolecule #4: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 4 / Number of copies: 2 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)