+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-7301 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Atomic resolution structure of human bufavirus 2 | |||||||||
 Map data Map data | Atomic resolution structure of human bufavirus 2 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | parvovirus / protoparvovirus / Bufavirus / VIRUS LIKE PARTICLE | |||||||||
| Function / homology | Parvovirus coat protein VP2 / Parvovirus coat protein VP1/VP2 / Parvovirus coat protein VP1/VP2 / Capsid/spike protein, ssDNA virus / T=1 icosahedral viral capsid / structural molecule activity / VP2 Function and homology information Function and homology information | |||||||||
| Biological species |  Bufavirus-2 Bufavirus-2 | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.79 Å | |||||||||
 Authors Authors | Mietzsch M / Agbandje-McKenna M | |||||||||
 Citation Citation |  Journal: Viruses / Year: 2018 Journal: Viruses / Year: 2018Title: Atomic Resolution Structures of Human Bufaviruses Determined by Cryo-Electron Microscopy. Authors: Maria Ilyas / Mario Mietzsch / Shweta Kailasan / Elina Väisänen / Mengxiao Luo / Paul Chipman / J Kennon Smith / Justin Kurian / Duncan Sousa / Robert McKenna / Maria Söderlund-Venermo / ...Authors: Maria Ilyas / Mario Mietzsch / Shweta Kailasan / Elina Väisänen / Mengxiao Luo / Paul Chipman / J Kennon Smith / Justin Kurian / Duncan Sousa / Robert McKenna / Maria Söderlund-Venermo / Mavis Agbandje-McKenna /   Abstract: Bufavirus strain 1 (BuV1), a member of the genus of the , was first isolated from fecal samples of children with acute diarrhea in Burkina Faso. Since this initial discovery, BuVs have been isolated ...Bufavirus strain 1 (BuV1), a member of the genus of the , was first isolated from fecal samples of children with acute diarrhea in Burkina Faso. Since this initial discovery, BuVs have been isolated in several countries, including Finland, the Netherlands, and Bhutan, in pediatric patients exhibiting similar symptoms. Towards their characterization, the structures of virus-like particles of BuV1, BuV2, and BuV3, the current known genotypes, have been determined by cryo-electron microscopy and image reconstruction to 2.84, 3.79, and 3.25 Å, respectively. The BuVs, 65-73% identical in amino acid sequence, conserve the major viral protein, VP2, structure and general capsid surface features of parvoviruses. These include a core β-barrel (βB-βI), α-helix A, and large surface loops inserted between these elements in VP2. The capsid contains depressions at the icosahedral 2-fold and around the 5-fold axes, and has three separated protrusions surrounding the 3-fold axes. Structure comparison among the BuVs and to available parvovirus structures revealed capsid surface variations and capsid 3-fold protrusions that depart from the single pinwheel arrangement of the animal protoparvoviruses. These structures provide a platform to begin the molecular characterization of these potentially pathogenic viruses. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_7301.map.gz emd_7301.map.gz | 60.9 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-7301-v30.xml emd-7301-v30.xml emd-7301.xml emd-7301.xml | 13.4 KB 13.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_7301.png emd_7301.png | 308 KB | ||
| Filedesc metadata |  emd-7301.cif.gz emd-7301.cif.gz | 5.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-7301 http://ftp.pdbj.org/pub/emdb/structures/EMD-7301 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7301 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-7301 | HTTPS FTP |
-Validation report
| Summary document |  emd_7301_validation.pdf.gz emd_7301_validation.pdf.gz | 506 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_7301_full_validation.pdf.gz emd_7301_full_validation.pdf.gz | 505.6 KB | Display | |
| Data in XML |  emd_7301_validation.xml.gz emd_7301_validation.xml.gz | 7.2 KB | Display | |
| Data in CIF |  emd_7301_validation.cif.gz emd_7301_validation.cif.gz | 8.3 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7301 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7301 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7301 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-7301 | HTTPS FTP |
-Related structure data
| Related structure data |  6bx0MC  7300C  7302C  6bwxC  6bx1C C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_7301.map.gz / Format: CCP4 / Size: 176.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_7301.map.gz / Format: CCP4 / Size: 176.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Atomic resolution structure of human bufavirus 2 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.22 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
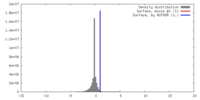
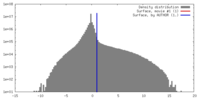
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Bufavirus-2
| Entire | Name:  Bufavirus-2 Bufavirus-2 |
|---|---|
| Components |
|
-Supramolecule #1: Bufavirus-2
| Supramolecule | Name: Bufavirus-2 / type: virus / ID: 1 / Parent: 0 / Macromolecule list: all / NCBI-ID: 1209383 / Sci species name: Bufavirus-2 / Virus type: VIRUS-LIKE PARTICLE / Virus isolate: SEROTYPE / Virus enveloped: No / Virus empty: Yes |
|---|---|
| Host (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: VP2
| Macromolecule | Name: VP2 / type: protein_or_peptide / ID: 1 / Number of copies: 60 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Bufavirus-2 Bufavirus-2 |
| Molecular weight | Theoretical: 61.484789 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGVGHSTGDY NNRTEFIYHG EKVTIICHST RLIHINMSDM EEYRIYDTDR GPLFPTTQDL QGRDTINDSY HAKVETPWKL LHANCWGVW FSPADFQQLI TTCREIKPSE FEQKIENIVI KTVSKTGAGE QETTQYNNDL TALLQVAQDN SNLMPWAADN F YIDSVGYV ...String: SGVGHSTGDY NNRTEFIYHG EKVTIICHST RLIHINMSDM EEYRIYDTDR GPLFPTTQDL QGRDTINDSY HAKVETPWKL LHANCWGVW FSPADFQQLI TTCREIKPSE FEQKIENIVI KTVSKTGAGE QETTQYNNDL TALLQVAQDN SNLMPWAADN F YIDSVGYV PWRASKLPTY CYHVDTWNTI DINQADRPNQ WREIRKGIQW DNIQFTPLET MINIDLLRTG DAWESGKYIF NA KPASLAY HWQSQRHIGS CHPSTAPNNE GGQGTNIANI NCWQWGDRSN PSSASTRVSN EHIGYSWPEW QIHYSTGGPV INP GQPFSQ APWGSTVAGT RLTQGATEKA IYDWNHGDDQ QGMRETWWQN NEHMTGQTDW GPKNAHQSEL NDHIQSASHF WRNS YHNTF GPYTACDDHG PQYPWGAIWG KMPDTTHKPM MSAHAPFLLN GPPGQLFVKL APNYTDVLDN SGTHTSRIVT YGTFW WSGK MIFEAKLRTP RQWNTYNLPT LNEREHMQNT VPNAVGKFEL PYLPGRAMPN YTM UniProtKB: VP2 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: DIRECT ELECTRON DE-20 (5k x 3k) / Digitization - Frames/image: 2-20 / Number real images: 429 / Average electron dose: 57.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Refinement | Protocol: RIGID BODY FIT / Target criteria: Correlation coefficient |
|---|---|
| Output model |  PDB-6bx0: |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) X (Row.)
X (Row.) Y (Col.)
Y (Col.)