[English] 日本語
 Yorodumi
Yorodumi- EMDB-6933: Cryo-EM reconstruction of flexible nanotubes self-assembled from ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-6933 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM reconstruction of flexible nanotubes self-assembled from baculovirus major capsid protein | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Helicoverpa armigera nucleopolyhedrovirus Helicoverpa armigera nucleopolyhedrovirus | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 14.0 Å | |||||||||
 Authors Authors | Rao G / Fu Y / Li N / Yin J / Zhang J / Wang M / Hu Z / Cao S | |||||||||
 Citation Citation |  Journal: ACS Appl Mater Interfaces / Year: 2018 Journal: ACS Appl Mater Interfaces / Year: 2018Title: Controllable Assembly of Flexible Protein Nanotubes for Loading Multifunctional Modules. Authors: Guibo Rao / Yan Fu / Na Li / Jiayi Yin / Jie Zhang / Manli Wang / Zhihong Hu / Sheng Cao /  Abstract: Viruses with filamentous morphologies, such as tobacco mosaic virus (TMV) and M13 bacteriophage, have long been studied as multivalent nanoscaffolds for loading functional motifs. Structural assembly ...Viruses with filamentous morphologies, such as tobacco mosaic virus (TMV) and M13 bacteriophage, have long been studied as multivalent nanoscaffolds for loading functional motifs. Structural assembly of the capsid proteins (CPs) of filamentous viruses often requires the presence of DNA or RNA molecules, which has limited their applications. Here, we describe a strategy for controllable assembly of flexible bio-nanotubes consisting of Escherichia coli expressed CP of baculovirus Helicoverpa armigera nucleopolyhedrovirus (HearNPV) in vitro. These protein-only nanotubes were studied as a new structural platform for high-density presentation of multiple active molecules on the exterior surface by direct fusion of the protein of interest to the N-terminus of HearNPV CP (HaCP). Structural characterization using cryoelectron microscopy demonstrated that the HaCP could assemble into two closely related but structurally distinct tube types, suggesting the tunable HaCP interaction network is the major contributor to the flexibility of HaCP nanotubes. Our flexible nanotubes could tolerate larger molecular modifications compared with TMV-based templates and could be used as promising candidates for versatile molecular loading applications. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_6933.map.gz emd_6933.map.gz | 218.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-6933-v30.xml emd-6933-v30.xml emd-6933.xml emd-6933.xml | 8.2 KB 8.2 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_6933.png emd_6933.png | 98.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-6933 http://ftp.pdbj.org/pub/emdb/structures/EMD-6933 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6933 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-6933 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_6933.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_6933.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.014 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
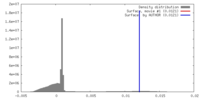
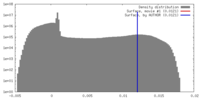
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Helicoverpa armigera nucleopolyhedrovirus
| Entire | Name:  Helicoverpa armigera nucleopolyhedrovirus Helicoverpa armigera nucleopolyhedrovirus |
|---|---|
| Components |
|
-Supramolecule #1: Helicoverpa armigera nucleopolyhedrovirus
| Supramolecule | Name: Helicoverpa armigera nucleopolyhedrovirus / type: virus / ID: 1 / Parent: 0 / Macromolecule list: #1 / NCBI-ID: 51313 / Sci species name: Helicoverpa armigera nucleopolyhedrovirus / Virus type: VIRUS-LIKE PARTICLE / Virus isolate: OTHER / Virus enveloped: No / Virus empty: Yes |
|---|---|
| Host system | Organism:  |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 3.06 Å Applied symmetry - Helical parameters - Δ&Phi: -31.74 ° Applied symmetry - Helical parameters - Axial symmetry: C1 (asymmetric) Resolution.type: BY AUTHOR / Resolution: 14.0 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION / Number images used: 17923 |
|---|---|
| Final angle assignment | Type: NOT APPLICABLE |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)