[English] 日本語
 Yorodumi
Yorodumi- EMDB-5797: Structure of the Ribosome with Elongation Factor G Trapped in the... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5797 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the Ribosome with Elongation Factor G Trapped in the Pre-Translocation State | |||||||||
 Map data Map data | Ribosome reconstruction | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | protein structure / translation / EF-G / electron cryo-microscopy / single particle analysis | |||||||||
| Function / homology |  Function and homology information Function and homology informationintracellular anatomical structure / ribosome disassembly / guanosine tetraphosphate binding / translational elongation / translation elongation factor activity / Hydrolases; Acting on acid anhydrides; Acting on GTP to facilitate cellular and subcellular movement / GTPase activity / GTP binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 6.2 Å | |||||||||
 Authors Authors | Brilot AF / Korostelev AA / Ermolenko DN / Grigorieff N | |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2013 Journal: Proc Natl Acad Sci U S A / Year: 2013Title: Structure of the ribosome with elongation factor G trapped in the pretranslocation state. Authors: Axel F Brilot / Andrei A Korostelev / Dmitri N Ermolenko / Nikolaus Grigorieff /  Abstract: During protein synthesis, tRNAs and their associated mRNA codons move sequentially on the ribosome from the A (aminoacyl) site to the P (peptidyl) site to the E (exit) site in a process catalyzed by ...During protein synthesis, tRNAs and their associated mRNA codons move sequentially on the ribosome from the A (aminoacyl) site to the P (peptidyl) site to the E (exit) site in a process catalyzed by a universally conserved ribosome-dependent GTPase [elongation factor G (EF-G) in prokaryotes and elongation factor 2 (EF-2) in eukaryotes]. Although the high-resolution structure of EF-G bound to the posttranslocation ribosome has been determined, the pretranslocation conformation of the ribosome bound with EF-G and A-site tRNA has evaded visualization owing to the transient nature of this state. Here we use electron cryomicroscopy to determine the structure of the 70S ribosome with EF-G, which is trapped in the pretranslocation state using antibiotic viomycin. Comparison with the posttranslocation ribosome shows that the small subunit of the pretranslocation ribosome is rotated by ∼12° relative to the large subunit. Domain IV of EF-G is positioned in the cleft between the body and head of the small subunit outwardly of the A site and contacts the A-site tRNA. Our findings suggest a model in which domain IV of EF-G promotes the translocation of tRNA from the A to the P site as the small ribosome subunit spontaneously rotates back from the hybrid, rotated state into the nonrotated posttranslocation state. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5797.map.gz emd_5797.map.gz | 105.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5797-v30.xml emd-5797-v30.xml emd-5797.xml emd-5797.xml | 14.7 KB 14.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_5797_1.jpg emd_5797_1.jpg emd_5797_2.png emd_5797_2.png | 224.6 KB 236.6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5797 http://ftp.pdbj.org/pub/emdb/structures/EMD-5797 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5797 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5797 | HTTPS FTP |
-Related structure data
| Related structure data |  5796C  5798C  5799C  5800C  4v7cC  4v7dC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_5797.map.gz / Format: CCP4 / Size: 122.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5797.map.gz / Format: CCP4 / Size: 122.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Ribosome reconstruction | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.04 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
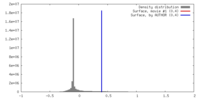
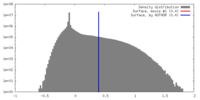
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Post-translocation ribosome bound to EF-G with P, E sites occupie...
| Entire | Name: Post-translocation ribosome bound to EF-G with P, E sites occupied by tRNA |
|---|---|
| Components |
|
-Supramolecule #1000: Post-translocation ribosome bound to EF-G with P, E sites occupie...
| Supramolecule | Name: Post-translocation ribosome bound to EF-G with P, E sites occupied by tRNA type: sample / ID: 1000 / Details: Sample was monodisperse / Number unique components: 6 |
|---|---|
| Molecular weight | Theoretical: 3 MDa |
-Supramolecule #1: 70S ribosome
| Supramolecule | Name: 70S ribosome / type: complex / ID: 1 / Recombinant expression: No / Database: NCBI / Ribosome-details: ribosome-prokaryote: ALL |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 3 MDa |
-Macromolecule #1: Elongation Factor G
| Macromolecule | Name: Elongation Factor G / type: protein_or_peptide / ID: 1 / Name.synonym: EF-G / Number of copies: 1 / Oligomeric state: monomer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 78 KDa |
| Recombinant expression | Organism:  |
| Sequence | UniProtKB: Elongation factor G GO: translational elongation, GTP binding, translation elongation factor activity, intracellular anatomical structure InterPro: Translation elongation factor EFG/EF2 |
-Macromolecule #2: Transfer RNA
| Macromolecule | Name: Transfer RNA / type: rna / ID: 2 / Name.synonym: tRNA / Details: Two tRNA present (P and E sites). / Classification: TRANSFER / Structure: DOUBLE HELIX / Synthetic?: No |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Experimental: 25 KDa |
-Macromolecule #3: Messenger RNA
| Macromolecule | Name: Messenger RNA / type: rna / ID: 3 / Name.synonym: mRNA / Classification: OTHER / Structure: SINGLE STRANDED / Synthetic?: Yes |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 12 KDa |
| Sequence | String: GGCAAGGAGG UAAAAAUGUU UAAACGUAAA UCUACU |
-Macromolecule #4: Fusidic Acid
| Macromolecule | Name: Fusidic Acid / type: ligand / ID: 4 / Name.synonym: Fus / Number of copies: 1 / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Theoretical: 1 KDa |
| Chemical component information | 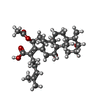 ChemComp-FUA: |
-Macromolecule #5: Viomycin
| Macromolecule | Name: Viomycin / type: ligand / ID: 5 / Name.synonym: Vio / Number of copies: 1 / Recombinant expression: No / Database: NCBI |
|---|---|
| Source (natural) | Organism: unidentified (others) |
| Molecular weight | Theoretical: 1 KDa |
| Chemical component information | 
ChemComp-PRD_000226: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL |
|---|---|
| Buffer | pH: 7.6 Details: 10 mM HEPES-KOH, 5 mM MgCl2, 90 mM NH4Cl, 2 mM spermidine, 0.1 mM spermine, 6 mM BME, 0.5 mM viomycin, 0.5 mM GTP, 0.5 mM fusidic acid |
| Grid | Details: C-flat 1.2/1.3 holey carbon 400 mesh copper grid, glow discharged with a current of -20 mA for 45 seconds in an EMITECH K100X glow discharge unit |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 95 % / Instrument: FEI VITROBOT MARK II Method: Freshly glow-disharged grids were loaded into an FEI Mark II Vitrobot and equilibrated to 95% relative humidity at 22 degrees Celsius. 2 microliters of sample was applied through the side ...Method: Freshly glow-disharged grids were loaded into an FEI Mark II Vitrobot and equilibrated to 95% relative humidity at 22 degrees Celsius. 2 microliters of sample was applied through the side port, blotted for 7 seconds with a positional offset of 2, and plunged into liquid ethane. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Alignment procedure | Legacy - Astigmatism: Automatically corrected using FEI software |
| Date | Nov 2, 2012 |
| Image recording | Category: CCD / Film or detector model: FEI FALCON I (4k x 4k) / Digitization - Sampling interval: 14.0 µm / Number real images: 13341 / Average electron dose: 30 e/Å2 / Bits/pixel: 16 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 134615 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 0.01 mm / Nominal defocus max: 6.95 µm / Nominal defocus min: 1.15 µm / Nominal magnification: 133333 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Details | Refinement and 3D classification performed by Frealign. See primary citation Supplementary Information for details. |
|---|---|
| CTF correction | Details: CTFFIND3, FREALIGN per micrograph |
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 6.2 Å / Resolution method: OTHER / Software - Name: EMAN2, IMAGIC, FREALIGN, RSAMPLE, CTFFIND3 Details: Refinement included data to 12 Angstrom resolution to limit FSC bias. See primary citation Supplementary Information for details. Number images used: 1341961 |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)