[English] 日本語
 Yorodumi
Yorodumi- EMDB-4533: Human post-catalytic spliceosome (P complex), focused refinement ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4533 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human post-catalytic spliceosome (P complex), focused refinement on Prp19 | |||||||||
 Map data Map data | Human post-catalytic spliceosome (P complex), focused refinement on Prp19 | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
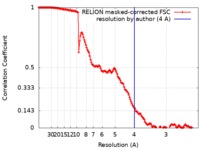
| Method | single particle reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Fica SM / Oubridge C / Wilkinson ME / Newman AJ / Nagai KN | |||||||||
 Citation Citation |  Journal: Science / Year: 2019 Journal: Science / Year: 2019Title: A human postcatalytic spliceosome structure reveals essential roles of metazoan factors for exon ligation. Authors: Sebastian M Fica / Chris Oubridge / Max E Wilkinson / Andrew J Newman / Kiyoshi Nagai /  Abstract: During exon ligation, the spliceosome recognizes the 3'-splice site (3'SS) of precursor messenger RNA (pre-mRNA) through non-Watson-Crick pairing with the 5'SS and the branch adenosine, in a ...During exon ligation, the spliceosome recognizes the 3'-splice site (3'SS) of precursor messenger RNA (pre-mRNA) through non-Watson-Crick pairing with the 5'SS and the branch adenosine, in a conformation stabilized by Prp18 and Prp8. Here we present the 3.3-angstrom cryo-electron microscopy structure of a human postcatalytic spliceosome just after exon ligation. The 3'SS docks at the active site through conserved RNA interactions in the absence of Prp18. Unexpectedly, the metazoan-specific FAM32A directly bridges the 5'-exon and intron 3'SS of pre-mRNA and promotes exon ligation, as shown by functional assays. CACTIN, SDE2, and NKAP-factors implicated in alternative splicing-further stabilize the catalytic conformation of the spliceosome during exon ligation. Together these four proteins act as exon ligation factors. Our study reveals how the human spliceosome has co-opted additional proteins to modulate a conserved RNA-based mechanism for 3'SS selection and to potentially fine-tune alternative splicing at the exon ligation stage. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4533.map.gz emd_4533.map.gz | 238.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4533-v30.xml emd-4533-v30.xml emd-4533.xml emd-4533.xml | 15.7 KB 15.7 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4533_fsc.xml emd_4533_fsc.xml | 14.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_4533.png emd_4533.png | 120.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4533 http://ftp.pdbj.org/pub/emdb/structures/EMD-4533 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4533 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4533 | HTTPS FTP |
-Validation report
| Summary document |  emd_4533_validation.pdf.gz emd_4533_validation.pdf.gz | 280.8 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_4533_full_validation.pdf.gz emd_4533_full_validation.pdf.gz | 279.9 KB | Display | |
| Data in XML |  emd_4533_validation.xml.gz emd_4533_validation.xml.gz | 13.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4533 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4533 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4533 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-4533 | HTTPS FTP |
-Related structure data
| Related structure data |  4525C  4526C  4527C  4528C  4529C  4530C  4532C  4534C  4535C  6qdvC C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_4533.map.gz / Format: CCP4 / Size: 262.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4533.map.gz / Format: CCP4 / Size: 262.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Human post-catalytic spliceosome (P complex), focused refinement on Prp19 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.2 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Human post-catalytic P complex spliceosome
| Entire | Name: Human post-catalytic P complex spliceosome |
|---|---|
| Components |
|
-Supramolecule #1: Human post-catalytic P complex spliceosome
| Supramolecule | Name: Human post-catalytic P complex spliceosome / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#44 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 3.0 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 1 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
| ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK III Details: 3 uL sample was applied to the grid, left for 25s, then blotted for 2.5-3.5s and immediately plunged into liquid ethane.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 60.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.5 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 130000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller





 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)