+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Rabbit RyR1 disease mutant Y523S in complex with FKBP12.6 embedded in lipidic nanodisc in the open state | |||||||||||||||
 マップデータ マップデータ | primary map | |||||||||||||||
 試料 試料 |
| |||||||||||||||
 キーワード キーワード | Ryanodine receptor / RyR / RyR1 / calcium channel / mutation / NTDC mutation / malignant hyperthermia / central core disease / membrane protein / excitation-contraction coupling | |||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報ATP-gated ion channel activity / : / negative regulation of calcium-mediated signaling / negative regulation of insulin secretion involved in cellular response to glucose stimulus / neuronal action potential propagation / insulin secretion involved in cellular response to glucose stimulus / negative regulation of release of sequestered calcium ion into cytosol / terminal cisterna / ryanodine-sensitive calcium-release channel activity / ryanodine receptor complex ...ATP-gated ion channel activity / : / negative regulation of calcium-mediated signaling / negative regulation of insulin secretion involved in cellular response to glucose stimulus / neuronal action potential propagation / insulin secretion involved in cellular response to glucose stimulus / negative regulation of release of sequestered calcium ion into cytosol / terminal cisterna / ryanodine-sensitive calcium-release channel activity / ryanodine receptor complex / release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / response to redox state / ossification involved in bone maturation / negative regulation of heart rate / cellular response to caffeine / 'de novo' protein folding / skin development / FK506 binding / organelle membrane / intracellularly gated calcium channel activity / smooth endoplasmic reticulum / outflow tract morphogenesis / smooth muscle contraction / T cell proliferation / toxic substance binding / voltage-gated calcium channel activity / calcium channel inhibitor activity / striated muscle contraction / skeletal muscle fiber development / Ion homeostasis / regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / release of sequestered calcium ion into cytosol / regulation of cardiac muscle contraction by regulation of the release of sequestered calcium ion / sarcoplasmic reticulum membrane / calcium channel complex / cellular response to calcium ion / muscle contraction / sarcoplasmic reticulum / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / calcium channel regulator activity / calcium-mediated signaling / protein maturation / sarcolemma / Stimuli-sensing channels / calcium ion transmembrane transport / calcium channel activity / Z disc / intracellular calcium ion homeostasis / disordered domain specific binding / protein refolding / positive regulation of cytosolic calcium ion concentration / protein homotetramerization / transmembrane transporter binding / calmodulin binding / signaling receptor binding / calcium ion binding / ATP binding / membrane / identical protein binding / cytoplasm 類似検索 - 分子機能 | |||||||||||||||
| 生物種 |   Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||||||||
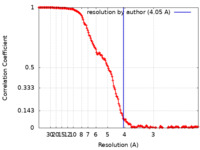
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 4.05 Å | |||||||||||||||
 データ登録者 データ登録者 | Iyer KA / Hu Y / Murayama T / Samso M | |||||||||||||||
| 資金援助 |  米国, 4件 米国, 4件
| |||||||||||||||
 引用 引用 |  ジャーナル: Proc Natl Acad Sci U S A / 年: 2022 ジャーナル: Proc Natl Acad Sci U S A / 年: 2022タイトル: Molecular mechanism of the severe MH/CCD mutation Y522S in skeletal ryanodine receptor (RyR1) by cryo-EM. 著者: Kavita A Iyer / Yifan Hu / Thomas Klose / Takashi Murayama / Montserrat Samsó /   要旨: Ryanodine receptors (RyRs) are main regulators of intracellular Ca release and muscle contraction. The Y522S mutation of RyR1 causes central core disease, a weakening myopathy, and malignant ...Ryanodine receptors (RyRs) are main regulators of intracellular Ca release and muscle contraction. The Y522S mutation of RyR1 causes central core disease, a weakening myopathy, and malignant hyperthermia, a sudden and potentially fatal response to anesthetics or heat. Y522 is in the core of the N-terminal subdomain C of RyR1 and the mechanism of how this mutation orchestrates malfunction is unpredictable for this 2-MDa ion channel, which has four identical subunits composed of 15 distinct cytoplasmic domains each. We expressed and purified the RyR1 rabbit homolog, Y523S, from HEK293 cells and reconstituted it in nanodiscs under closed and open states. The high-resolution cryogenic electron microscopic (cryo-EM) three-dimensional (3D) structures show that the phenyl ring of Tyr functions in a manner analogous to a "spacer" within an α-helical bundle. Mutation to the much smaller Ser alters the hydrophobic network within the bundle, triggering rearrangement of its α-helices with repercussions in the orientation of most cytoplasmic domains. Examining the mutation-induced readjustments exposed a series of connected α-helices acting as an ∼100 Å-long lever: One end protrudes toward the dihydropyridine receptor, its molecular activator (akin to an antenna), while the other end reaches the Ca activation site. The Y523S mutation elicits channel preactivation in the absence of any activator and full opening at 1.5 µM free Ca, increasing by ∼20-fold the potency of Ca to activate the channel compared with RyR1 wild type (WT). This study identified a preactivated pathological state of RyR1 and a long-range lever that may work as a molecular switch to open the channel. | |||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_25710.map.gz emd_25710.map.gz | 286.3 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-25710-v30.xml emd-25710-v30.xml emd-25710.xml emd-25710.xml | 26.1 KB 26.1 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_25710_fsc.xml emd_25710_fsc.xml | 19.7 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_25710.png emd_25710.png | 126 KB | ||
| Filedesc metadata |  emd-25710.cif.gz emd-25710.cif.gz | 9.8 KB | ||
| その他 |  emd_25710_half_map_1.map.gz emd_25710_half_map_1.map.gz emd_25710_half_map_2.map.gz emd_25710_half_map_2.map.gz | 281.2 MB 281.2 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25710 http://ftp.pdbj.org/pub/emdb/structures/EMD-25710 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25710 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25710 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7t65MC  7t64C M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_25710.map.gz / 形式: CCP4 / 大きさ: 303.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_25710.map.gz / 形式: CCP4 / 大きさ: 303.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | primary map | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.078 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: half map 1
| ファイル | emd_25710_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half map 1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: half map 2
| ファイル | emd_25710_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | half map 2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Rabbit RyR1 disease mutant Y523S in complex with FKBP12.6 embedde...
| 全体 | 名称: Rabbit RyR1 disease mutant Y523S in complex with FKBP12.6 embedded in lipidic nanodisc in the open state |
|---|---|
| 要素 |
|
-超分子 #1: Rabbit RyR1 disease mutant Y523S in complex with FKBP12.6 embedde...
| 超分子 | 名称: Rabbit RyR1 disease mutant Y523S in complex with FKBP12.6 embedded in lipidic nanodisc in the open state タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#2 |
|---|---|
| 分子量 | 理論値: 2.26 MDa |
-分子 #1: Ryanodine receptor 1
| 分子 | 名称: Ryanodine receptor 1 / タイプ: protein_or_peptide / ID: 1 / コピー数: 4 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 565.832562 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: MGDGGEGEDE VQFLRTDDEV VLQCSATVLK EQLKLCLAAE GFGNRLCFLE PTSNAQNVPP DLAICCFTLE QSLSVRALQE MLANTVEAG VESSQGGGHR TLLYGHAILL RHAHSRMYLS CLTTSRSMTD KLAFDVGLQE DATGEACWWT MHPASKQRSE G EKVRVGDD ...文字列: MGDGGEGEDE VQFLRTDDEV VLQCSATVLK EQLKLCLAAE GFGNRLCFLE PTSNAQNVPP DLAICCFTLE QSLSVRALQE MLANTVEAG VESSQGGGHR TLLYGHAILL RHAHSRMYLS CLTTSRSMTD KLAFDVGLQE DATGEACWWT MHPASKQRSE G EKVRVGDD LILVSVSSER YLHLSTASGE LQVDASFMQT LWNMNPICSC CEEGYVTGGH VLRLFHGHMD ECLTISAADS DD QRRLVYY EGGAVCTHAR SLWRLEPLRI SWSGSHLRWG QPLRIRHVTT GRYLALTEDQ GLVVVDACKA HTKATSFCFR VSK EKLDTA PKRDVEGMGP PEIKYGESLC FVQHVASGLW LTYAAPDPKA LRLGVLKKKA ILHQEGHMDD ALFLTRCQQE ESQA ARMIH STAGLYNQFI KGLDSFSGKP RGSGPPAGPA LPIEAVILSL QDLIGYFEPP SEELQHEEKQ SKLRSLRNRQ SLFQE EGML SLVLNCIDRL NVYTTAAHFA EYAGEEAAES WKEIVNLLSE LLASLIRGNR ANCALFSTNL DWVVSKLDRL EASSGI LEV LYCVLIESPE VLNIIQENHI KSIISLLDKH GRNHKVLDVL CSLCVCNGVA VRSNQDLITE NLLPGRELLL QTNLINY VT SIRPNIFVGR AEGSTQYGKW YFEVMVDEVV PFLTAQATHL RVGWALTEGY SPYPGGGEGW GGNGVGDDLY SYGFDGLH L WTGHVARPVT SPGQHLLAPE DVVSCCLDLS VPSISFRING CPVQGVFEAF NLDGLFFPVV SFSAGVKVRF LLGGRHGEF KFLPPPGYAP CHEAVLPRER LRLEPIKEYR REGPRGPHLV GPSRCLSHTD FVPCPVDTVQ IVLPPHLERI REKLAENIHE LWALTRIEQ GWTYGPVRDD NKRLHPCLVN FHSLPEPERN YNLQMSGETL KTLLALGCHV GMADEKAEDN LKKTKLPKTY M MSNGYKPA PLDLSHVRLT PAQTTLVDRL AENGHNVWAR DRVAQGWSYS AVQDIPARRN PRLVPYRLLD EATKRSNRDS LC QAVRTLL GYGYNIEPPD QEPSQVENQS RWDRVRIFRA EKSYTVQSGR WYFEFEAVTT GEMRVGWARP ELRPDVELGA DEL AYVFNG HRGQRWHLGS EPFGRPWQSG DVVGCMIDLT ENTIIFTLNG EVLMSDSGSE TAFREIEIGD GFLPVCSLGP GQVG HLNLG QDVSSLRFFA ICGLQEGFEP FAINMQRPVT TWFSKSLPQF EPVPPEHPHY EVARMDGTVD TPPCLRLAHR TWGSQ NSLV EMLFLRLSLP VQFHQHFRCT AGATPLAPPG LQPPAEDEAR AAEPDPDYEN LRRSAGGWGE AEGGKEGTAK EGTPGG TPQ PGVEAQPVRA ENEKDATTEK NKKRGFLFKA KKAAMMTQPP ATPALPRLPH DVVPADNRDD PEIILNTTTY YYSVRVF AG QEPSCVWVGW VTPDYHQHDM NFDLSKVRAV TVTMGDEQGN VHSSLKCSNC YMVWGGDFVS PGQQGRISHT DLVIGCLV D LATGLMTFTA NGKESNTFFQ VEPNTKLFPA VFVLPTHQNV IQFELGKQKN IMPLSAAMFL SERKNPAPQC PPRLEVQML MPVSWSRMPN HFLQVETRRA GERLGWAVQC QDPLTMMALH IPEENRCMDI LELSERLDLQ RFHSHTLRLY RAVCALGNNR VAHALCSHV DQAQLLHALE DAHLPGPLRA GYYDLLISIH LESACRSRRS MLSEYIVPLT PETRAITLFP PGRKGGNARR H GLPGVGVT TSLRPPHHFS PPCFVAALPA AGVAEAPARL SPAIPLEALR DKALRMLGEA VRDGGQHARD PVGGSVEFQF VP VLKLVST LLVMGIFGDE DVKQILKMIE PEVFTEEEEE EEEEEEEEEE EEEDEEEKEE DEEEEEKEDA EKEEEEAPEG EKE DLEEGL LQMKLPESVK LQMCNLLEYF CDQELQHRVE SLAAFAERYV DKLQANQRSR YALLMRAFTM SAAETARRTR EFRS PPQEQ INMLLHFKDE ADEEDCPLPE DIRQDLQDFH QDLLAHCGIQ LEGEEEEPEE ETSLSSRLRS LLETVRLVKK KEEKP EEEL PAEEKKPQSL QELVSHMVVR WAQEDYVQSP ELVRAMFSLL HRQYDGLGEL LRALPRAYTI SPSSVEDTMS LLECLG QIR SLLIVQMGPQ EENLMIQSIG NIMNNKVFYQ HPNLMRALGM HETVMEVMVN VLGGGETKEI RFPKMVTSCC RFLCYFC RI SRQNQRSMFD HLSYLLENSG IGLGMQGSTP LDVAAASVID NNELALALQE QDLEKVVSYL AGCGLQSCPM LLAKGYPD I GWNPCGGERY LDFLRFAVFV NGESVEENAN VVVRLLIRKP ECFGPALRGE GGSGLLAAIE EAIRISEDPA RDGPGVRRD RRREHFGEEP PEENRVHLGH AIMSFYAALI DLLGRCAPEM HLIQAGKGEA LRIRAILRSL VPLDDLVGII SLPLQIPTLG KDGALVQPK MSASFVPDHK ASMVLFLDRV YGIENQDFLL HVLDVGFLPD MRAAASLDTA TFSTTEMALA LNRYLCLAVL P LITKCAPL FAGTEHRAIM VDSMLHTVYR LSRGRSLTKA QRDVIEDCLM ALCRYIRPSM LQHLLRRLVF DVPILNEFAK MP LKLLTNH YERCWKYYCL PTGWANFGVT SEEELHLTRK LFWGIFDSLA HKKYDQELYR MAMPCLCAIA GALPPDYVDA SYS SKAEKK ATVDAEGNFD PRPVETLNVI IPEKLDSFIN KFAEYTHEKW AFDKIQNNWS YGENVDEELK THPMLRPYKT FSEK DKEIY RWPIKESLKA MIAWEWTIEK AREGEEERTE KKKTRKISQT AQTYDPREGY NPQPPDLSGV TLSRELQAMA EQLAE NYHN TWGRKKKQEL EAKGGGTHPL LVPYDTLTAK EKARDREKAQ ELLKFLQMNG YAVTRGLKDM ELDTSSIEKR FAFGFL QQL LRWMDISQEF IAHLEAVVSS GRVEKSPHEQ EIKFFAKILL PLINQYFTNH CLYFLSTPAK VLGSGGHASN KEKEMIT SL FCKLAALVRH RVSLFGTDAP AVVNCLHILA RSLDARTVMK SGPEIVKAGL RSFFESASED IEKMVENLRL GKVSQART Q VKGVGQNLTY TTVALLPVLT TLFQHIAQHQ FGDDVILDDV QVSCYRTLCS IYSLGTTKNT YVEKLRPALG ECLARLAAA MPVAFLEPQL NEYNACSVYT TKSPRERAIL GLPNSVEEMC PDIPVLDRLM ADIGGLAESG ARYTEMPHVI EITLPMLCSY LPRWWERGP EAPPPALPAG APPPCTAVTS DHLNSLLGNI LRIIVNNLGI DEATWMKRLA VFAQPIVSRA RPELLHSHFI P TIGRLRKR AGKVVAEEEQ LRLEAKAEAE EGELLVRDEF SVLCRDLYAL YPLLIRYVDN NRAHWLTEPN ANAEELFRMV GE IFIYWSK SHNFKREEQN FVVQNEINNM SFLTADSKSK MAKAGDAQSG GSDQERTKKK RRGDRYSVQT SLIVATLKKM LPI GLNMCA PTDQDLIMLA KTRYALKDTD EEVREFLQNN LHLQGKVEGS PSLRWQMALY RGLPGREEDA DDPEKIVRRV QEVS AVLYH LEQTEHPYKS KKAVWHKLLS KQRRRAVVAC FRMTPLYNLP THRACNMFLE SYKAAWILTE DHSFEDRMID DLSKA GEQE EEEEEVEEKK PDPLHQLVLH FSRTALTEKS KLDEDYLYMA YADIMAKSCH LEEGGENGEA EEEEVEVSFE EKEMEK QRL LYQQSRLHTR GAAEMVLQMI SACKGETGAM VSSTLKLGIS ILNGGNAEVQ QKMLDYLKDK KEVGFFQSIQ ALMQTCS VL DLNAFERQNK AEGLGMVNED GTVINRQNGE KVMADDEFTQ DLFRFLQLLC EGHNNDFQNY LRTQTGNTTT INIIICTV D YLLRLQESIS DFYWYYSGKD VIEEQGKRNF SKAMSVAKQV FNSLTEYIQG PCTGNQQSLA HSRLWDAVVG FLHVFAHMM MKLAQDSSQI ELLKELLDLQ KDMVVMLLSL LEGNVVNGMI ARQMVDMLVE SSSNVEMILK FFDMFLKLKD IVGSEAFQDY VTDPRGLIS KKDFQKAMDS QKQFTGPEIQ FLLSCSEADE NEMINFEEFA NRFQEPARDI GFNVAVLLTN LSEHVPHDPR L RNFLELAE SILEYFRPYL GRIEIMGASR RIERIYFEIS ETNRAQWEMP QVKESKRQFI FDVVNEGGEA EKMELFVSFC ED TIFEMQI AAQISEPEGE PEADEDEGMG EAAAEGAEEG AAGAEGAAGT VAAGATARLA AAAARALRGL SYRSLRRRVR RLR RLTARE AATALAALLW AVVARAGAAG AGAAAGALRL LWGSLFGGGL VEGAKKVTVT ELLAGMPDPT SDEVHGEQPA GPGG DADGA GEGEGEGDAA EGDGDEEVAG HEAGPGGAEG VVAVADGGPF RPEGAGGLGD MGDTTPAEPP TPEGSPILKR KLGVD GEEE ELVPEPEPEP EPEPEKADEE NGEKEEVPEA PPEPPKKAPP SPPAKKEEAG GAGMEFWGEL EVQRVKFLNY LSRNFY TLR FLALFLAFAI NFILLFYKVS DSPPGEDDME GSAAGDLAGA GSGGGSGWGS GAGEEAEGDE DENMVYYFLE ESTGYME PA LWCLSLLHTL VAFLCIIGYN CLKVPLVIFK REKELARKLE FDGLYITEQP GDDDVKGQWD RLVLNTPSFP SNYWDKFV K RKVLDKHGDI FGRERIAELL GMDLASLEIT AHNERKPDPP PGLLTWLMSI DVKYQIWKFG VIFTDNSFLY LGWYMVMSL LGHYNNFFFA AHLLDIAMGV KTLRTILSSV THNGKQLVMT VGLLAVVVYL YTVVAFNFFR KFYNKSEDED EPDMKCDDMM TCYLFHMYV GVRAGGGIGD EIEDPAGDEY ELYRVVFDIT FFFFVIVILL AIIQGLIIDA FGELRDQQEQ VKEDMETKCF I CGIGSDYF DTTPHGFETH TLEEHNLANY MFFLMYLINK DETEHTGQES YVWKMYQERC WDFFPAGDCF RKQYEDQLS UniProtKB: Ryanodine receptor 1 |
-分子 #2: Peptidyl-prolyl cis-trans isomerase FKBP1B
| 分子 | 名称: Peptidyl-prolyl cis-trans isomerase FKBP1B / タイプ: protein_or_peptide / ID: 2 / コピー数: 4 / 光学異性体: LEVO / EC番号: peptidylprolyl isomerase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 11.667305 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: GVEIETISPG DGRTFPKKGQ TCVVHYTGML QNGKKFDSSR DRNKPFKFRI GKQEVIKGFE EGAAQMSLGQ RAKLTCTPDV AYGATGHPG VIPPNATLIF DVELLNLE UniProtKB: Peptidyl-prolyl cis-trans isomerase FKBP1B |
-分子 #3: ADENOSINE-5'-TRIPHOSPHATE
| 分子 | 名称: ADENOSINE-5'-TRIPHOSPHATE / タイプ: ligand / ID: 3 / コピー数: 4 / 式: ATP |
|---|---|
| 分子量 | 理論値: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-分子 #4: CALCIUM ION
| 分子 | 名称: CALCIUM ION / タイプ: ligand / ID: 4 / コピー数: 4 / 式: CA |
|---|---|
| 分子量 | 理論値: 40.078 Da |
-分子 #5: ZINC ION
| 分子 | 名称: ZINC ION / タイプ: ligand / ID: 5 / コピー数: 4 / 式: ZN |
|---|---|
| 分子量 | 理論値: 65.409 Da |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 4.5 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.4 構成要素:
| ||||||||||||||||||
| グリッド | モデル: Quantifoil R1.2/1.3 / 材質: GOLD / メッシュ: 300 | ||||||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 95 % / チャンバー内温度: 277.15 K / 装置: FEI VITROBOT MARK IV / 詳細: Blot force 2; blot for 1.0s before plunging. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 特殊光学系 | エネルギーフィルター - スリット幅: 20 eV |
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 撮影したグリッド数: 1 / 実像数: 11018 / 平均露光時間: 3.12 sec. / 平均電子線量: 53.19 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.5 µm 最小 デフォーカス(公称値): 0.7000000000000001 µm 倍率(公称値): 81000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)