[English] 日本語
 Yorodumi
Yorodumi- EMDB-20554: Cryo-EM structure of DDB1-DCAF15 bound to E7820 and the second RR... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-20554 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of DDB1-DCAF15 bound to E7820 and the second RRM of RBM39 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 10.0 Å | |||||||||
 Authors Authors | Faust T / Yoon H / Nowak RP / Donovan KA / Li Z / Cai Q / Eleuteri NA / Zhang T / Gray NS / Fischer ES | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Chem Biol / Year: 2020 Journal: Nat Chem Biol / Year: 2020Title: Structural complementarity facilitates E7820-mediated degradation of RBM39 by DCAF15. Authors: Tyler B Faust / Hojong Yoon / Radosław P Nowak / Katherine A Donovan / Zhengnian Li / Quan Cai / Nicholas A Eleuteri / Tinghu Zhang / Nathanael S Gray / Eric S Fischer /  Abstract: The investigational drugs E7820, indisulam and tasisulam (aryl-sulfonamides) promote the degradation of the splicing factor RBM39 in a proteasome-dependent mechanism. While the activity critically ...The investigational drugs E7820, indisulam and tasisulam (aryl-sulfonamides) promote the degradation of the splicing factor RBM39 in a proteasome-dependent mechanism. While the activity critically depends on the cullin RING ligase substrate receptor DCAF15, the molecular details remain elusive. Here we present the cryo-EM structure of the DDB1-DCAF15-DDA1 core ligase complex bound to RBM39 and E7820 at a resolution of 4.4 Å, together with crystal structures of engineered subcomplexes. We show that DCAF15 adopts a new fold stabilized by DDA1, and that extensive protein-protein contacts between the ligase and substrate mitigate low affinity interactions between aryl-sulfonamides and DCAF15. Our data demonstrate how aryl-sulfonamides neo-functionalize a shallow, non-conserved pocket on DCAF15 to selectively bind and degrade RBM39 and the closely related splicing factor RBM23 without the requirement for a high-affinity ligand, which has broad implications for the de novo discovery of molecular glue degraders. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_20554.map.gz emd_20554.map.gz | 66.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-20554-v30.xml emd-20554-v30.xml emd-20554.xml emd-20554.xml | 23.6 KB 23.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_20554_fsc.xml emd_20554_fsc.xml | 10.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_20554.png emd_20554.png | 35.2 KB | ||
| Masks |  emd_20554_msk_1.map emd_20554_msk_1.map | 85.5 MB |  Mask map Mask map | |
| Others |  emd_20554_additional.map.gz emd_20554_additional.map.gz emd_20554_additional_1.map.gz emd_20554_additional_1.map.gz emd_20554_half_map_1.map.gz emd_20554_half_map_1.map.gz emd_20554_half_map_2.map.gz emd_20554_half_map_2.map.gz | 11.7 MB 11.7 MB 67 MB 67.1 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-20554 http://ftp.pdbj.org/pub/emdb/structures/EMD-20554 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20554 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20554 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_20554.map.gz / Format: CCP4 / Size: 85.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_20554.map.gz / Format: CCP4 / Size: 85.5 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.064 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
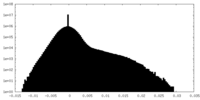
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_20554_msk_1.map emd_20554_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
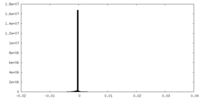
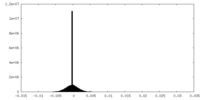
| Density Histograms |
-Additional map: #1
| File | emd_20554_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: #1
| File | emd_20554_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_20554_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_20554_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of DDB1-DCAF15 bound to E7820 and the second RRM of RBM39
| Entire | Name: Complex of DDB1-DCAF15 bound to E7820 and the second RRM of RBM39 |
|---|---|
| Components |
|
-Supramolecule #1: Complex of DDB1-DCAF15 bound to E7820 and the second RRM of RBM39
| Supramolecule | Name: Complex of DDB1-DCAF15 bound to E7820 and the second RRM of RBM39 type: complex / ID: 1 / Parent: 0 / Macromolecule list: all Details: Compound: E7820; Chemical Name: 3-Cyano-N-(3-cyano-4-methyl-1H-indol-7-yl)benzenesulfonamide |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Molecular weight | Theoretical: 211 KDa |
-Macromolecule #1: DNA Damage-Binding Protein 1
| Macromolecule | Name: DNA Damage-Binding Protein 1 / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGSSHHHHHH SAVDENLYFQ GGGRMSYNYV VTAQKPTAVN GCVTGHFTSA EDLNLLIAKN TRLEIYVVTA EGLRPVKEVG MYGKIAVMEL FRPKGESKDL LFILTAKYNA CILEYKQSGE SIDIITRAHG NVQDRIGRPS ETGIIGIIDP ECRMIGLRLY DGLFKVIPLD ...String: MGSSHHHHHH SAVDENLYFQ GGGRMSYNYV VTAQKPTAVN GCVTGHFTSA EDLNLLIAKN TRLEIYVVTA EGLRPVKEVG MYGKIAVMEL FRPKGESKDL LFILTAKYNA CILEYKQSGE SIDIITRAHG NVQDRIGRPS ETGIIGIIDP ECRMIGLRLY DGLFKVIPLD RDNKELKAFN IRLEELHVID VKFLYGCQAP TICFVYQDPQ GRHVKTYEVS LREKEFNKGP WKQENVEAEA SMVIAVPEPF GGAIIIGQES ITYHNGDKYL AIAPPIIKQS TIVCHNRVDP NGSRYLLGDM EGRLFMLLLE KEEQMDGTVT LKDLRVELLG ETSIAECLTY LDNGVVFVGS RLGDSQLVKL NVDSNEQGSY VVAMETFTNL GPIVDMCVVD LERQGQGQLV TCSGAFKEGS LRIIRNGIGI HEHASIDLPG IKGLWPLRSD PNRETDDTLV LSFVGQTRVL MLNGEEVEET ELMGFVDDQQ TFFCGNVAHQ QLIQITSASV RLVSQEPKAL VSEWKEPQAK NISVASCNSS QVVVAVGRAL YYLQIHPQEL RQISHTEMEH EVACLDITPL GDSNGLSPLC AIGLWTDISA RILKLPSFEL LHKEMLGGEI IPRSILMTTF ESSHYLLCAL GDGALFYFGL NIETGLLSDR KKVTLGTQPT VLRTFRSLST TNVFACSDRP TVIYSSNHKL VFSNVNLKEV NYMCPLNSDG YPDSLALANN STLTIGTIDE IQKLHIRTVP LYESPRKICY QEVSQCFGVL SSRIEVQDTS GGTTALRPSA STQALSSSVS SSKLFSSSTA PHETSFGEEV EVHNLLIIDQ HTFEVLHAHQ FLQNEYALSL VSCKLGKDPN TYFIVGTAMV YPEEAEPKQG RIVVFQYSDG KLQTVAEKEV KGAVYSMVEF NGKLLASINS TVRLYEWTTE KELRTECNHY NNIMALYLKT KGDFILVGDL MRSVLLLAYK PMEGNFEEIA RDFNPNWMSA VEILDDDNFL GAENAFNLFV CQKDSAATTD EERQHLQEVG LFHLGEFVNV FCHGSLVMQN LGETSTPTQG SVLFGTVNGM IGLVTSLSES WYNLLLDMQN RLNKVIKSVG KIEHSFWRSF HTERKTEPAT GFIDGDLIES FLDISRPKMQ EVVANLQYDD GSGMKREATA DDLIKVVEEL TRIH |
-Macromolecule #2: DDB1 And CUL4 Associated Factor 15
| Macromolecule | Name: DDB1 And CUL4 Associated Factor 15 / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MDWSHPQFEK SAVDENLYFQ GGCGRMAPSS KSERNSGAGS GGGGPGGAGG KRAAGRRREH VLKQLERVKI SGQLSPRLFR KLPPRVCVSL KNIVDEDFLY AGHIFLGFSK CGRYVLSYTS SSGDDDFSFY IYHLYWWEFN VHSKLKLVRQ VRLFQDEEIY SDLYLTVCEW ...String: MDWSHPQFEK SAVDENLYFQ GGCGRMAPSS KSERNSGAGS GGGGPGGAGG KRAAGRRREH VLKQLERVKI SGQLSPRLFR KLPPRVCVSL KNIVDEDFLY AGHIFLGFSK CGRYVLSYTS SSGDDDFSFY IYHLYWWEFN VHSKLKLVRQ VRLFQDEEIY SDLYLTVCEW PSDASKVIVF GFNTRSANGM LMNMMMMSDE NHRDIYVSTV AVPPPGRCAA CQDASRAHPG DPNAQCLRHG FMLHTKYQVV YPFPTFQPAF QLKKDQVVLL NTSYSLVACA VSVHSAGDRS FCQILYDHST CPLAPASPPE PQSPELPPAL PSFCPEAAPA RSSGSPEPSP AIAKAKEFVA DIFRRAKEAK GGVPEEARPA LCPGPSGSRC RAHSEPLALC GETAPRDSPP ASEAPASEPG YVNYTKLYYV LESGEGTEPE DELEDDKISL PFVVTDLRGR NLRPMRERTA VQGQYLTVEQ LTLDFEYVIN EVIRHDATWG HQFCSFSDYD IVILEVCPET NQVLINIGLL LLAFPSPTEE GQLRPKTYHT SLKVAWDLNT GIFETVSVGD LTEVKGQTSG SVWSSYRKSC VDMVMKWLVP ESSGRYVNRM TNEALHKGCS LKVLADSERY TWIVL |
-Macromolecule #3: RNA Binding Motif Protein 39
| Macromolecule | Name: RNA Binding Motif Protein 39 / type: protein_or_peptide / ID: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGSSHHHHHH SAVDENLYFQ GGCGRGSAGP MRLYVGSLHF NITEDMLRGI FEPFGRIESI QLMMDSETGR SKGYGFITFS DSECAKKALE QLNGFELAGR PMKVGHVTER TDA |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.075 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
Details: Gel filtration buffer was made fresh the day of the purification | |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 60 % / Chamber temperature: 286 K / Instrument: LEICA EM GP / Details: Blotted for 3 seconds before plunging.. | |||||||||||||||
| Details | This sample was monodisperse |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Number grids imaged: 1 / Number real images: 1457 / Average electron dose: 40.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.5 µm / Nominal magnification: 130000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)