[English] 日本語
 Yorodumi
Yorodumi- EMDB-20000: Cdc48-Ufd1/Npl4 complex processing poly-ubiquitinated substrate i... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-20000 | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cdc48-Ufd1/Npl4 complex processing poly-ubiquitinated substrate in the presence of ADP-BeFx, state 2 | ||||||||||||
 Map data Map data | Map sharpened with a B factor of -100 | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | ATPase / ATPase complex / ubiquitin / quality control / MOTOR PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationEGAD pathway / SCF complex disassembly in response to cadmium stress / mitotic DNA replication termination / Ovarian tumor domain proteases / Cdc48p-Npl4p-Vms1p AAA ATPase complex / Doa10p ubiquitin ligase complex / stress-induced homeostatically regulated protein degradation pathway / endoplasmic reticulum membrane fusion / sister chromatid biorientation / Hrd1p ubiquitin ligase ERAD-L complex ...EGAD pathway / SCF complex disassembly in response to cadmium stress / mitotic DNA replication termination / Ovarian tumor domain proteases / Cdc48p-Npl4p-Vms1p AAA ATPase complex / Doa10p ubiquitin ligase complex / stress-induced homeostatically regulated protein degradation pathway / endoplasmic reticulum membrane fusion / sister chromatid biorientation / Hrd1p ubiquitin ligase ERAD-L complex / DNA replication termination / RQC complex / mitochondria-associated ubiquitin-dependent protein catabolic process / positive regulation of mitochondrial fusion / cytoplasm protein quality control by the ubiquitin-proteasome system / HSF1 activation / protein-containing complex disassembly / protein transport to vacuole involved in ubiquitin-dependent protein catabolic process via the multivesicular body sorting pathway / nuclear protein quality control by the ubiquitin-proteasome system / endosome to plasma membrane protein transport / ribophagy / : / protein phosphatase regulator activity / piecemeal microautophagy of the nucleus / mating projection tip / Protein methylation / replisome / mitotic spindle disassembly / VCP-NPL4-UFD1 AAA ATPase complex / ribosome-associated ubiquitin-dependent protein catabolic process / vesicle-fusing ATPase / nonfunctional rRNA decay / retrograde protein transport, ER to cytosol / KEAP1-NFE2L2 pathway / Neddylation / protein quality control for misfolded or incompletely synthesized proteins / autophagosome maturation / polyubiquitin modification-dependent protein binding / ATP metabolic process / ERAD pathway / Neutrophil degranulation / rescue of stalled cytosolic ribosome / macroautophagy / ubiquitin binding / positive regulation of protein localization to nucleus / proteasome-mediated ubiquitin-dependent protein catabolic process / endoplasmic reticulum membrane / ATP hydrolysis activity / mitochondrion / ATP binding / identical protein binding / nucleus / cytosol Similarity search - Function | ||||||||||||
| Biological species |  | ||||||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.6 Å | ||||||||||||
 Authors Authors | Twomey EC / Ji Z | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: Science / Year: 2019 Journal: Science / Year: 2019Title: Substrate processing by the Cdc48 ATPase complex is initiated by ubiquitin unfolding. Authors: Edward C Twomey / Zhejian Ji / Thomas E Wales / Nicholas O Bodnar / Scott B Ficarro / Jarrod A Marto / John R Engen / Tom A Rapoport /  Abstract: The Cdc48 adenosine triphosphatase (ATPase) (p97 or valosin-containing protein in mammals) and its cofactor Ufd1/Npl4 extract polyubiquitinated proteins from membranes or macromolecular complexes for ...The Cdc48 adenosine triphosphatase (ATPase) (p97 or valosin-containing protein in mammals) and its cofactor Ufd1/Npl4 extract polyubiquitinated proteins from membranes or macromolecular complexes for subsequent degradation by the proteasome. How Cdc48 processes its diverse and often well-folded substrates is unclear. Here, we report cryo-electron microscopy structures of the Cdc48 ATPase in complex with Ufd1/Npl4 and polyubiquitinated substrate. The structures show that the Cdc48 complex initiates substrate processing by unfolding a ubiquitin molecule. The unfolded ubiquitin molecule binds to Npl4 and projects its N-terminal segment through both hexameric ATPase rings. Pore loops of the second ring form a staircase that acts as a conveyer belt to move the polypeptide through the central pore. Inducing the unfolding of ubiquitin allows the Cdc48 ATPase complex to process a broad range of substrates. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_20000.map.gz emd_20000.map.gz | 4.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-20000-v30.xml emd-20000-v30.xml emd-20000.xml emd-20000.xml | 21.8 KB 21.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_20000.png emd_20000.png | 125.7 KB | ||
| Filedesc metadata |  emd-20000.cif.gz emd-20000.cif.gz | 6.1 KB | ||
| Others |  emd_20000_additional_1.map.gz emd_20000_additional_1.map.gz emd_20000_additional_2.map.gz emd_20000_additional_2.map.gz emd_20000_half_map_1.map.gz emd_20000_half_map_1.map.gz emd_20000_half_map_2.map.gz emd_20000_half_map_2.map.gz | 4.6 MB 24.4 MB 45.4 MB 45.4 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-20000 http://ftp.pdbj.org/pub/emdb/structures/EMD-20000 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20000 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20000 | HTTPS FTP |
-Related structure data
| Related structure data |  6oabMC  0665C  0666C  6oa9C  6oaaC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_20000.map.gz / Format: CCP4 / Size: 48.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_20000.map.gz / Format: CCP4 / Size: 48.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map sharpened with a B factor of -100 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.15 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
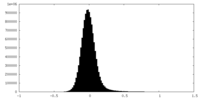
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Additional map: Map sharpened with a B factor of -150
| File | emd_20000_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map sharpened with a B factor of -150 | ||||||||||||
| Projections & Slices |
| ||||||||||||
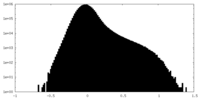
| Density Histograms |
-Additional map: Unsharpened map from refinement
| File | emd_20000_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map from refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
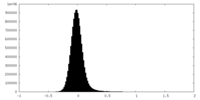
| Density Histograms |
-Half map: Half map 2 from refinement
| File | emd_20000_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 2 from refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
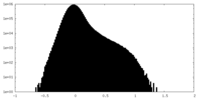
| Density Histograms |
-Half map: Half map 1 from refinement
| File | emd_20000_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 from refinement | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cdc48-Npl4 complex processing poly-ubiquitinated substrate in the...
| Entire | Name: Cdc48-Npl4 complex processing poly-ubiquitinated substrate in the presence of ADP-BeFx, state 2 |
|---|---|
| Components |
|
-Supramolecule #1: Cdc48-Npl4 complex processing poly-ubiquitinated substrate in the...
| Supramolecule | Name: Cdc48-Npl4 complex processing poly-ubiquitinated substrate in the presence of ADP-BeFx, state 2 type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Cell division control protein 48
| Macromolecule | Name: Cell division control protein 48 / type: protein_or_peptide / ID: 1 / Number of copies: 5 / Enantiomer: LEVO / EC number: vesicle-fusing ATPase |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 92.106914 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MGEEHKPLLD ASGVDPREED KTATAILRRK KKDNMLLVDD AINDDNSVIA INSNTMDKLE LFRGDTVLVK GKKRKDTVLI VLIDDELED GACRINRVVR NNLRIRLGDL VTIHPCPDIK YATRISVLPI ADTIEGITGN LFDVFLKPYF VEAYRPVRKG D HFVVRGGM ...String: MGEEHKPLLD ASGVDPREED KTATAILRRK KKDNMLLVDD AINDDNSVIA INSNTMDKLE LFRGDTVLVK GKKRKDTVLI VLIDDELED GACRINRVVR NNLRIRLGDL VTIHPCPDIK YATRISVLPI ADTIEGITGN LFDVFLKPYF VEAYRPVRKG D HFVVRGGM RQVEFKVVDV EPEEYAVVAQ DTIIHWEGEP INREDEENNM NEVGYDDIGG CRKQMAQIRE MVELPLRHPQ LF KAIGIKP PRGVLMYGPP GTGKTLMARA VANETGAFFF LINGPEVMSK MAGESESNLR KAFEEAEKNA PAIIFIDEID SIA PKRDKT NGEVERRVVS QLLTLMDGMK ARSNVVVIAA TNRPNSIDPA LRRFGRFDRE VDIGIPDATG RLEVLRIHTK NMKL ADDVD LEALAAETHG YVGADIASLC SEAAMQQIRE KMDLIDLDED EIDAEVLDSL GVTMDNFRFA LGNSNPSALR ETVVE SVNV TWDDVGGLDE IKEELKETVE YPVLHPDQYT KFGLSPSKGV LFYGPPGTGK TLLAKAVATE VSANFISVKG PELLSM WYG ESESNIRDIF DKARAAAPTV VFLDELDSIA KARGGSLGDA GGASDRVVNQ LLTEMDGMNA KKNVFVIGAT NRPDQID PA ILRPGRLDQL IYVPLPDENA RLSILNAQLR KTPLEPGLEL TAIAKATQGF SGADLLYIVQ RAAKYAIKDS IEAHRQHE A EKEVKVEGED VEMTDEGAKA EQEPEVDPVP YITKEHFAEA MKTAKRSVSD AELRRYEAYS QQMKASRGQF SNFNFNDAP LGTTATDNAN SNNSAPSGAG AAFGSNAEED DDLYS UniProtKB: AAA family ATPase involved in ubiquitin-mediated protein degradation CDC48 |
-Macromolecule #2: poly(alanine) substrate
| Macromolecule | Name: poly(alanine) substrate / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.723883 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: AAAAAAAAAA AAAAAAAAAA AAAA |
-Macromolecule #3: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 3 / Number of copies: 10 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information |  ChemComp-ADP: |
-Macromolecule #4: BERYLLIUM TRIFLUORIDE ION
| Macromolecule | Name: BERYLLIUM TRIFLUORIDE ION / type: ligand / ID: 4 / Number of copies: 8 / Formula: BEF |
|---|---|
| Molecular weight | Theoretical: 66.007 Da |
| Chemical component information |  ChemComp-BEF: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Details: unspecified |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 295 K / Instrument: GATAN CRYOPLUNGE 3 Details: Waited 20 seconds before blotting for 2.5-3 seconds.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 44.7 e/Å2 |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 3.6 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 93395 |
| Initial angle assignment | Type: RANDOM ASSIGNMENT |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC (ver. 2) |
 Movie
Movie Controller
Controller



























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)