+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | PAPP-A dimer in complex with its inhibitor STC2 (CASP target) | |||||||||
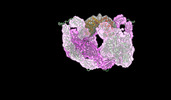
 Map data Map data | Composit map of PAPP-A dimer in complex with its endogenous inhibitor STC2 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Metzincin metalloprotease Inhibitor complex / HYDROLASE | |||||||||
| Function / homology |  Function and homology information Function and homology informationregulation of hormone biosynthetic process / pappalysin-1 / regulation of store-operated calcium entry / response to vitamin D / negative regulation of multicellular organism growth / decidualization / endoplasmic reticulum unfolded protein response / embryo implantation / Post-translational protein phosphorylation / female pregnancy ...regulation of hormone biosynthetic process / pappalysin-1 / regulation of store-operated calcium entry / response to vitamin D / negative regulation of multicellular organism growth / decidualization / endoplasmic reticulum unfolded protein response / embryo implantation / Post-translational protein phosphorylation / female pregnancy / hormone activity / metalloendopeptidase activity / response to peptide hormone / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / intracellular calcium ion homeostasis / metallopeptidase activity / response to oxidative stress / cellular response to hypoxia / cell surface receptor signaling pathway / endoplasmic reticulum lumen / negative regulation of gene expression / heme binding / perinuclear region of cytoplasm / enzyme binding / endoplasmic reticulum / Golgi apparatus / protein homodimerization activity / proteolysis / : / extracellular region / zinc ion binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 5.02 Å | |||||||||
 Authors Authors | Kobbero SD / Gajhede M / Mirza OA / Boesen T / Oxvig C | |||||||||
| Funding support |  Denmark, 1 items Denmark, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structure of the proteolytic enzyme PAPP-A with the endogenous inhibitor stanniocalcin-2 reveals its inhibitory mechanism. Authors: Sara Dam Kobberø / Michael Gajhede / Osman Asghar Mirza / Søren Kløverpris / Troels Rønn Kjær / Jakob Hauge Mikkelsen / Thomas Boesen / Claus Oxvig /  Abstract: The metzincin metalloproteinase PAPP-A plays a key role in the regulation of insulin-like growth factor (IGF) signaling by specific cleavage of inhibitory IGF binding proteins (IGFBPs). Using single- ...The metzincin metalloproteinase PAPP-A plays a key role in the regulation of insulin-like growth factor (IGF) signaling by specific cleavage of inhibitory IGF binding proteins (IGFBPs). Using single-particle cryo-electron microscopy (cryo-EM), we here report the structure of PAPP-A in complex with its endogenous inhibitor, stanniocalcin-2 (STC2), neither of which have been reported before. The highest resolution (3.1 Å) was obtained for the STC2 subunit and the N-terminal approximately 1000 residues of the PAPP-A subunit. The 500 kDa 2:2 PAPP-A·STC2 complex is a flexible multidomain ensemble with numerous interdomain contacts. In particular, a specific disulfide bond between the subunits of STC2 and PAPP-A prevents dissociation, and interactions between STC2 and a module located in the very C-terminal end of the PAPP-A subunit prevent binding of its main substrate, IGFBP-4. While devoid of activity towards IGFBP-4, the active site cleft of the catalytic domain is accessible in the inhibited PAPP-A·STC2 complex, as shown by its ability to hydrolyze a synthetic peptide derived from IGFBP-4. Relevant to multiple human pathologies, this unusual mechanism of proteolytic inhibition may support the development of specific pharmaceutical agents, by which IGF signaling can be indirectly modulated. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_15221.map.gz emd_15221.map.gz | 55.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-15221-v30.xml emd-15221-v30.xml emd-15221.xml emd-15221.xml | 25.9 KB 25.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_15221.png emd_15221.png | 48.4 KB | ||
| Filedesc metadata |  emd-15221.cif.gz emd-15221.cif.gz | 7.4 KB | ||
| Others |  emd_15221_additional_1.map.gz emd_15221_additional_1.map.gz emd_15221_additional_2.map.gz emd_15221_additional_2.map.gz emd_15221_additional_3.map.gz emd_15221_additional_3.map.gz | 59.4 MB 59.3 MB 38.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-15221 http://ftp.pdbj.org/pub/emdb/structures/EMD-15221 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15221 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-15221 | HTTPS FTP |
-Related structure data
| Related structure data |  8a7eMC  8a7dC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_15221.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_15221.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Composit map of PAPP-A dimer in complex with its endogenous inhibitor STC2 | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.18594 Å | ||||||||||||||||||||||||||||||||||||
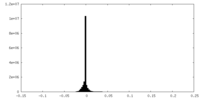
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Submap of composite map EMDB ID: EMD-15217
| File | emd_15221_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Submap of composite map EMDB ID: EMD-15217 | ||||||||||||
| Projections & Slices |
| ||||||||||||
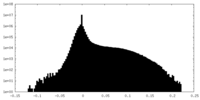
| Density Histograms |
-Additional map: Submap of composite map EMDB ID: EMD-15219
| File | emd_15221_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Submap of composite map EMDB ID: EMD-15219 | ||||||||||||
| Projections & Slices |
| ||||||||||||
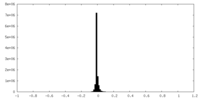
| Density Histograms |
-Additional map: Submap of composite map EMDB ID: EMD-15220
| File | emd_15221_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Submap of composite map EMDB ID: EMD-15220 | ||||||||||||
| Projections & Slices |
| ||||||||||||
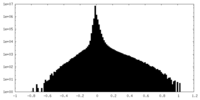
| Density Histograms |
- Sample components
Sample components
-Entire : PAPP-A dimer in complex with its endogenous inhibitor STC2 dimer
| Entire | Name: PAPP-A dimer in complex with its endogenous inhibitor STC2 dimer |
|---|---|
| Components |
|
-Supramolecule #1: PAPP-A dimer in complex with its endogenous inhibitor STC2 dimer
| Supramolecule | Name: PAPP-A dimer in complex with its endogenous inhibitor STC2 dimer type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 Details: Inhibited proteolytic complex generated by harvest of serum media, purifying on a nickel column followed by negative affinity purification and size-exclusion chromatography. |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular |
| Molecular weight | Theoretical: 100 KDa |
-Supramolecule #2: Pregnancy-associated plasma protein A
| Supramolecule | Name: Pregnancy-associated plasma protein A / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #2 / Details: Homodimer |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular |
-Supramolecule #3: Stanniocalcin-2
| Supramolecule | Name: Stanniocalcin-2 / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #1 / Details: Homodimer |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular Homo sapiens (human) / Tissue: ubiquitous / Location in cell: extracellular |
-Macromolecule #1: Stanniocalcin-2
| Macromolecule | Name: Stanniocalcin-2 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Cell: extracellular Homo sapiens (human) / Cell: extracellular |
| Molecular weight | Theoretical: 18.922953 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: RLSLQNTAEI QHCLVNAGDV GCGVFECFEN NSCEIRGLHG ICMTFLHNAG KFDAQGKSFI KDALKCKAHA LRHRFGCISR KCPAIREMV SQLQRECYLK HDLCAAAQEN TRVIVEMIHF KDLLLHEPYV DLVNLLLTCG EEVKEAITHS VQVQCEQNWG S LCSILSFC UniProtKB: Stanniocalcin-2 |
-Macromolecule #2: Pappalysin-1
| Macromolecule | Name: Pappalysin-1 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO / EC number: pappalysin-1 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / Organ: Ubiquitous / Cell: Extracellular Homo sapiens (human) / Organ: Ubiquitous / Cell: Extracellular |
| Molecular weight | Theoretical: 171.143047 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: EARGATEEPS PPSRALYFSG RGEQLRLRAD LELPRDAFTL QVWLRAEGGQ RSPAVITGLY DKCSYISRDR GWVVGIHTIS DQDNKDPRY FFSLKTDRAR QVTTINAHRS YLPGQWVYLA ATYDGQFMKL YVNGAQVATS GEQVGGIFSP LTQKCKVLML G GSALNHNY ...String: EARGATEEPS PPSRALYFSG RGEQLRLRAD LELPRDAFTL QVWLRAEGGQ RSPAVITGLY DKCSYISRDR GWVVGIHTIS DQDNKDPRY FFSLKTDRAR QVTTINAHRS YLPGQWVYLA ATYDGQFMKL YVNGAQVATS GEQVGGIFSP LTQKCKVLML G GSALNHNY RGYIEHFSLW KVARTQREIL SDMETHGAHT ALPQLLLQEN WDNVKHAWSP MKDGSSPKVE FSNAHGFLLD TS LEPPLCG QTLCDNTEVI ASYNQLSSFR QPKVVRYRVV NLYEDDHKNP TVTREQVDFQ HHQLAEAFKQ YNISWELDVL EVS NSSLRR RLILANCDIS KIGDENCDPE CNHTLTGHDG GDCRHLRHPA FVKKQHNGVC DMDCNYERFN FDGGECCDPE ITNV TQTCF DPDSPHRAYL DVNELKNILK LDGSTHLNIF FAKSSEEELA GVATWPWDKE ALMHLGGIVL NPSFYGMPGH THTMI HQIG HSLGLYHVFR GISEIQSCSD PCMETEPSFE TGDLCNDTNP APKHKSCGDP GPGNDTCGFH SFFNTPYNNF MSYADD DCT DSFTPNQVAR MHCYLDLVYQ GWQPSRKPAP VALAPQVLGH TTDSVTLEWF PPIDGHFFER ELGSACHLCL EGRILVQ YA SNASSPMPCS PSGHWSPREA EGHPDVEQPC KSSVRTWSPN SAVNPHTVPP ACPEPQGCYL ELEFLYPLVP ESLTIWVT F VSTDWDSSGA VNDIKLLAVS GKNISLGPQN VFCDVPLTIR LWDVGEEVYG IQIYTLDEHL EIDAAMLTST ADTPLCLQC KPLKYKVVRD PPLQMDVASI LHLNRKFVDM DLNLGSVYQY WVITISGTEE SEPSPAVTYI HGSGYCGDGI IQKDQGEQCD DMNKINGDG CSLFCRQEVS FNCIDEPSRC YFHDGDGVCE EFEQKTSIKD CGVYTPQGFL DQWASNASVS HQDQQCPGWV I IGQPAASQ VCRTKVIDLS EGISQHAWYP CTISYPYSQL AQTTFWLRAY FSQPMVAAAV IVHLVTDGTY YGDQKQETIS VQ LLDTKDQ SHDLGLHVLS CRNNPLIIPV VHDLSQPFYH SQAVRVSFSS PLVAISGVAL RSFDNFDPVT LSSCQRGETY SPA EQSCVH FACEKTDCPE LAVENASLNC SSSDRYHGAQ CTVSCRTGYV LQIRRDDELI KSQTGPSVTV TCTEGKWNKQ VACE PVDCS IPDHHQVYAA SFSCPEGTTF GSQCSFQCRH PAQLKGNNSL LTCMEDGLWS FPEALCELMC LAPPPVPNAD LQTAR CREN KHKVGSFCKY KCKPGYHVPG SSRKSKKRAF KTQCTQDGSW QEGACVPVTC DPPPPKFHGL YQCTNGFQFN SECRIK CED SDASQGLGSN VIHCRKDGTW NGSFHVCQEM QGQCSVPNEL NSNLKLQCPD GYAIGSECAT SCLDHNSESI ILPMNVT VR DIPHWLNPTR VERVVCTAGL KWYPHPALIH CVKGCEPFMG DNYCDAINNR AFCNYDGGDC CTSTVKTKKV TPFPMSCD L QGDCACRDPQ AQEHS UniProtKB: Pappalysin-1 |
-Macromolecule #3: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 3 / Number of copies: 2 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Macromolecule #4: CALCIUM ION
| Macromolecule | Name: CALCIUM ION / type: ligand / ID: 4 / Number of copies: 16 / Formula: CA |
|---|---|
| Molecular weight | Theoretical: 40.078 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.6 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
Details: HEPES buffer 20 mM Hepes pH 7.5 100 mM NaCl, 1mM CaCl | ||||||||||||
| Grid | Model: C-flat-2/2 / Material: COPPER / Mesh: 400 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 45 sec. | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 277.15 K / Instrument: LEICA EM GP / Details: Blot time 4.5 sec. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | #0 - Image recording ID: 1 / #0 - Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / #0 - Number grids imaged: 1 / #0 - Number real images: 10060 / #0 - Average exposure time: 0.8 sec. / #0 - Average electron dose: 58.0 e/Å2 / #1 - Image recording ID: 2 / #1 - Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / #1 - Number grids imaged: 1 / #1 - Number real images: 32144 / #1 - Average exposure time: 0.91 sec. / #1 - Average electron dose: 59.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.6 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Details | Initial model source: alphafold2 models PAPP-A AF-Q13219-F1 STC2 AF-O76061-F1 |
|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
| Output model |  PDB-8a7e: |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)