+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-13810 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human 17S U2 snRNP HEAT repeats | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
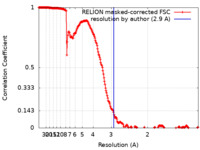
| Method | single particle reconstruction / cryo EM / Resolution: 2.9 Å | |||||||||
 Authors Authors | Tholen J / Galej WP | |||||||||
| Funding support | European Union, 1 items
| |||||||||
 Citation Citation |  Journal: Science / Year: 2022 Journal: Science / Year: 2022Title: Structural basis of branch site recognition by the human spliceosome. Authors: Jonas Tholen / Michal Razew / Felix Weis / Wojciech P Galej /   Abstract: Recognition of the intron branch site (BS) by the U2 small nuclear ribonucleoprotein (snRNP) is a critical event during spliceosome assembly. In mammals, BS sequences are poorly conserved, and ...Recognition of the intron branch site (BS) by the U2 small nuclear ribonucleoprotein (snRNP) is a critical event during spliceosome assembly. In mammals, BS sequences are poorly conserved, and unambiguous intron recognition cannot be achieved solely through a base-pairing mechanism. We isolated human 17 U2 snRNP and reconstituted in vitro its adenosine 5´-triphosphate (ATP)–dependent remodeling and binding to the pre–messenger RNA substrate. We determined a series of high-resolution (2.0 to 2.2 angstrom) structures providing snapshots of the BS selection process. The substrate-bound U2 snRNP shows that SF3B6 stabilizes the BS:U2 snRNA duplex, which could aid binding of introns with poor sequence complementarity. ATP-dependent remodeling uncoupled from substrate binding captures U2 snRNA in a conformation that competes with BS recognition, providing a selection mechanism based on branch helix stability. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_13810.map.gz emd_13810.map.gz | 708.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-13810-v30.xml emd-13810-v30.xml emd-13810.xml emd-13810.xml | 18.9 KB 18.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_13810_fsc.xml emd_13810_fsc.xml | 21.3 KB | Display |  FSC data file FSC data file |
| Images |  emd_13810.png emd_13810.png | 41.6 KB | ||
| Masks |  emd_13810_msk_1.map emd_13810_msk_1.map | 1000 MB |  Mask map Mask map | |
| Others |  emd_13810_half_map_1.map.gz emd_13810_half_map_1.map.gz emd_13810_half_map_2.map.gz emd_13810_half_map_2.map.gz | 712.4 MB 712.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-13810 http://ftp.pdbj.org/pub/emdb/structures/EMD-13810 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13810 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-13810 | HTTPS FTP |
-Related structure data
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_13810.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_13810.map.gz / Format: CCP4 / Size: 1000 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.64 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_13810_msk_1.map emd_13810_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_13810_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_13810_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : 17S U2 snRNP
| Entire | Name: 17S U2 snRNP |
|---|---|
| Components |
|
-Supramolecule #1: 17S U2 snRNP
| Supramolecule | Name: 17S U2 snRNP / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#9 Details: 17S U2 snRNP isolated from nuclear extract of HEK293F cells by affinity chromatography. |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 1.08 MDa |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
Details: Sample after desalting may have also contained up to 5% glycerol. | ||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Number grids imaged: 2 / Number real images: 15531 / Average exposure time: 1.0 sec. / Average electron dose: 53.45 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)