+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1190 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Molecular structure of human geminin. | |||||||||
 Map data Map data | 3D map of human Geminin, asymmetrical reconstruction of the tetramer | |||||||||
 Sample Sample |
| |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / negative staining / Resolution: 18.0 Å | |||||||||
 Authors Authors | Okorokov AL / Orlova EV / Kingsbury SR / Bagneris C / Gohlke U / Williams GH / Stoeber K | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2004 Journal: Nat Struct Mol Biol / Year: 2004Title: Molecular structure of human geminin. Authors: Andrei L Okorokov / Elena V Orlova / Sarah R Kingsbury / Claire Bagneris / Ulrich Gohlke / Gareth H Williams / Kai Stoeber /  Abstract: The origin licensing repressor geminin is a unique bifunctional protein providing a molecular link between cellular proliferation, differentiation and genomic stability. Here we report the first ...The origin licensing repressor geminin is a unique bifunctional protein providing a molecular link between cellular proliferation, differentiation and genomic stability. Here we report the first molecular structure of human geminin, determined by EM and image processing at a resolution of 17.5 A. The geminin molecule is a tetramer formed by two dimers with monomers interacting via coiled-coil domains. The unusual structural organization of geminin provides molecular insight into its bifunctional nature. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1190.map.gz emd_1190.map.gz | 2.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1190-v30.xml emd-1190-v30.xml emd-1190.xml emd-1190.xml | 10.1 KB 10.1 KB | Display Display |  EMDB header EMDB header |
| Images |  1190.gif 1190.gif | 10.2 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1190 http://ftp.pdbj.org/pub/emdb/structures/EMD-1190 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1190 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1190 | HTTPS FTP |
-Validation report
| Summary document |  emd_1190_validation.pdf.gz emd_1190_validation.pdf.gz | 186.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_1190_full_validation.pdf.gz emd_1190_full_validation.pdf.gz | 185.7 KB | Display | |
| Data in XML |  emd_1190_validation.xml.gz emd_1190_validation.xml.gz | 5.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1190 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1190 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1190 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1190 | HTTPS FTP |
-Related structure data
| Similar structure data |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_1190.map.gz / Format: CCP4 / Size: 29.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1190.map.gz / Format: CCP4 / Size: 29.8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | 3D map of human Geminin, asymmetrical reconstruction of the tetramer | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
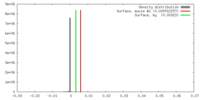
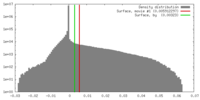
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Human replication licensing factor Geminin
| Entire | Name: Human replication licensing factor Geminin |
|---|---|
| Components |
|
-Supramolecule #1000: Human replication licensing factor Geminin
| Supramolecule | Name: Human replication licensing factor Geminin / type: sample / ID: 1000 Details: sample was produced in E. coli and purified by affinity chromatography followed by the ion-exchange and size exclusion chromatography steps. The resulting sample was homogeneous. Oligomeric state: tetrameric / Number unique components: 1 |
|---|---|
| Molecular weight | Experimental: 103 KDa |
-Macromolecule #1: Geminin
| Macromolecule | Name: Geminin / type: protein_or_peptide / ID: 1 / Name.synonym: Geminin / Details: tetramer / Number of copies: 4 / Oligomeric state: tetramer / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) / synonym: Human / Tissue: Lung and Urinary bladder / Organelle: nucleus Homo sapiens (human) / synonym: Human / Tissue: Lung and Urinary bladder / Organelle: nucleus |
| Molecular weight | Experimental: 90 KDa / Theoretical: 103 KDa |
| Recombinant expression | Organism:  |
-Experimental details
-Structure determination
| Method | negative staining |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.05 mg/mL |
|---|---|
| Buffer | pH: 7 / Details: 10 mM Tris HCl pH 8.0 100 mM NaCl |
| Staining | Type: NEGATIVE Details: negative staining with 2% methylamine vanadate, pH 8.0 or with 2% methylamine tungstate, pH 6.8 |
| Grid | Details: 400 mesh carbon-coated copper grid |
| Vitrification | Cryogen name: NONE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI 12 |
|---|---|
| Details | images were taken on FEI Technai T10 microscope in low dose mode. |
| Date | Jan 1, 2003 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: ZEISS SCAI / Digitization - Sampling interval: 7 µm / Number real images: 4 / Average electron dose: 20 e/Å2 / Camera length: 500 / Od range: 1.5 / Bits/pixel: 8 |
| Electron beam | Acceleration voltage: 100 kV / Electron source: TUNGSTEN HAIRPIN |
| Electron optics | Calibrated magnification: 44000 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.1 mm / Nominal defocus max: 1.6 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 44000 |
| Sample stage | Specimen holder: standard specimen holder / Specimen holder model: OTHER |
- Image processing
Image processing
| Details | selection was done manualy |
|---|---|
| CTF correction | Details: Each particle |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 18.0 Å / Resolution method: FSC 0.5 CUT-OFF / Software - Name: Imagic-5 / Details: Asymmetrical reconstruction / Number images used: 3300 |
| Final two d classification | Number classes: 300 |
-Atomic model buiding 1
| Software | Name: O |
|---|---|
| Details | Protocol: Rigid body. The coiled-coil domains were separately fitted by manual docking using program O |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
 Movie
Movie Controller
Controller



 UCSF Chimera
UCSF Chimera






 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)