[English] 日本語
 Yorodumi
Yorodumi- EMDB-11099: Cryo-EM structure of human 80S ribosomes bound to EBP1, eEF2 and ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11099 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human 80S ribosomes bound to EBP1, eEF2 and SERBP1 | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Ribosome / hibernation | |||||||||
| Function / homology |  Function and homology information Function and homology informationSynthesis of diphthamide-EEF2 / translation at postsynapse / ribosome hibernation / translation elongation factor binding / PML body organization / response to folic acid / SUMO binding / translation at presynapse / exit from mitosis / positive regulation of cytoplasmic translation ...Synthesis of diphthamide-EEF2 / translation at postsynapse / ribosome hibernation / translation elongation factor binding / PML body organization / response to folic acid / SUMO binding / translation at presynapse / exit from mitosis / positive regulation of cytoplasmic translation / optic nerve development / response to insecticide / regulation of translation involved in cellular response to UV / eukaryotic 80S initiation complex / negative regulation of formation of translation preinitiation complex / axial mesoderm development / negative regulation of endoplasmic reticulum unfolded protein response / ribosomal protein import into nucleus / regulation of G1 to G0 transition / retinal ganglion cell axon guidance / oxidized pyrimidine DNA binding / response to TNF agonist / positive regulation of base-excision repair / positive regulation of ubiquitin-protein transferase activity / protein-DNA complex disassembly / positive regulation of respiratory burst involved in inflammatory response / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage by p53 class mediator / positive regulation of gastrulation / positive regulation of intrinsic apoptotic signaling pathway in response to DNA damage / protein tyrosine kinase inhibitor activity / 90S preribosome assembly / IRE1-RACK1-PP2A complex / positive regulation of Golgi to plasma membrane protein transport / nucleolus organization / aggresome / positive regulation of DNA-templated transcription initiation / alpha-beta T cell differentiation / TNFR1-mediated ceramide production / positive regulation of DNA damage response, signal transduction by p53 class mediator / GAIT complex / negative regulation of RNA splicing / TORC2 complex binding / neural crest cell differentiation / supercoiled DNA binding / NF-kappaB complex / negative regulation of DNA repair / G1 to G0 transition / cytoplasmic translational initiation / oxidized purine DNA binding / cysteine-type endopeptidase activator activity involved in apoptotic process / middle ear morphogenesis / negative regulation of intrinsic apoptotic signaling pathway in response to hydrogen peroxide / rRNA modification in the nucleus and cytosol / negative regulation of bicellular tight junction assembly / ubiquitin-like protein conjugating enzyme binding / regulation of establishment of cell polarity / negative regulation of phagocytosis / erythrocyte homeostasis / cytoplasmic side of rough endoplasmic reticulum membrane / Formation of the ternary complex, and subsequently, the 43S complex / Uptake and function of diphtheria toxin / lncRNA binding / ion channel inhibitor activity / laminin receptor activity / protein kinase A binding / homeostatic process / pigmentation / Ribosomal scanning and start codon recognition / positive regulation of mitochondrial depolarization / Translation initiation complex formation / macrophage chemotaxis / lung morphogenesis / negative regulation of Wnt signaling pathway / positive regulation of natural killer cell proliferation / male meiosis I / fibroblast growth factor binding / monocyte chemotaxis / BH3 domain binding / Protein hydroxylation / negative regulation of translational frameshifting / regulation of adenylate cyclase-activating G protein-coupled receptor signaling pathway / positive regulation of GTPase activity / TOR signaling / SARS-CoV-1 modulates host translation machinery / mTORC1-mediated signalling / iron-sulfur cluster binding / skeletal muscle cell differentiation / regulation of cell division / Peptide chain elongation / translational elongation / cellular response to ethanol / Selenocysteine synthesis / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / Formation of a pool of free 40S subunits / negative regulation of protein binding / protein serine/threonine kinase inhibitor activity / Eukaryotic Translation Termination / blastocyst development / ubiquitin ligase inhibitor activity / SRP-dependent cotranslational protein targeting to membrane Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
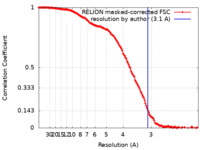
| Method | single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Wells JN / Buschauer R | |||||||||
 Citation Citation |  Journal: PLoS Biol / Year: 2020 Journal: PLoS Biol / Year: 2020Title: Structure and function of yeast Lso2 and human CCDC124 bound to hibernating ribosomes. Authors: Jennifer N Wells / Robert Buschauer / Timur Mackens-Kiani / Katharina Best / Hanna Kratzat / Otto Berninghausen / Thomas Becker / Wendy Gilbert / Jingdong Cheng / Roland Beckmann /   Abstract: Cells adjust to nutrient deprivation by reversible translational shutdown. This is accompanied by maintaining inactive ribosomes in a hibernation state, in which they are bound by proteins with ...Cells adjust to nutrient deprivation by reversible translational shutdown. This is accompanied by maintaining inactive ribosomes in a hibernation state, in which they are bound by proteins with inhibitory and protective functions. In eukaryotes, such a function was attributed to suppressor of target of Myb protein 1 (Stm1; SERPINE1 mRNA-binding protein 1 [SERBP1] in mammals), and recently, late-annotated short open reading frame 2 (Lso2; coiled-coil domain containing short open reading frame 124 [CCDC124] in mammals) was found to be involved in translational recovery after starvation from stationary phase. Here, we present cryo-electron microscopy (cryo-EM) structures of translationally inactive yeast and human ribosomes. We found Lso2/CCDC124 accumulating on idle ribosomes in the nonrotated state, in contrast to Stm1/SERBP1-bound ribosomes, which display a rotated state. Lso2/CCDC124 bridges the decoding sites of the small with the GTPase activating center (GAC) of the large subunit. This position allows accommodation of the duplication of multilocus region 34 protein (Dom34)-dependent ribosome recycling system, which splits Lso2-containing, but not Stm1-containing, ribosomes. We propose a model in which Lso2 facilitates rapid translation reactivation by stabilizing the recycling-competent state of inactive ribosomes. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11099.map.gz emd_11099.map.gz | 144 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11099-v30.xml emd-11099-v30.xml emd-11099.xml emd-11099.xml | 100.8 KB 100.8 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_11099_fsc.xml emd_11099_fsc.xml | 14.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_11099.png emd_11099.png | 239.6 KB | ||
| Filedesc metadata |  emd-11099.cif.gz emd-11099.cif.gz | 20.4 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11099 http://ftp.pdbj.org/pub/emdb/structures/EMD-11099 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11099 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11099 | HTTPS FTP |
-Related structure data
| Related structure data |  6z6mMC  6z6jC  6z6kC  6z6lC  6z6nC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_11099.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11099.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.061 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : 80S ribosome bound to EBP1, eEF2 and SERBP1
+Supramolecule #1: 80S ribosome bound to EBP1, eEF2 and SERBP1
+Macromolecule #1: 28S rRNA
+Macromolecule #2: 5S rRNA
+Macromolecule #3: 5.8S rRNA
+Macromolecule #47: 18S rRNA
+Macromolecule #83: tRNA
+Macromolecule #4: 60S ribosomal protein L8
+Macromolecule #5: 60S ribosomal protein L3
+Macromolecule #6: 60S ribosomal protein L4
+Macromolecule #7: 60S ribosomal protein L5
+Macromolecule #8: 60S ribosomal protein L6
+Macromolecule #9: 60S ribosomal protein L7
+Macromolecule #10: 60S ribosomal protein L7a
+Macromolecule #11: 60S ribosomal protein L9
+Macromolecule #12: 60S ribosomal protein L10-like
+Macromolecule #13: 60S ribosomal protein L11
+Macromolecule #14: 60S ribosomal protein L13
+Macromolecule #15: 60S ribosomal protein L14
+Macromolecule #16: 60S ribosomal protein L15
+Macromolecule #17: 60S ribosomal protein L13a
+Macromolecule #18: 60S ribosomal protein L17
+Macromolecule #19: 60S ribosomal protein L18
+Macromolecule #20: 60S ribosomal protein L19
+Macromolecule #21: 60S ribosomal protein L18a
+Macromolecule #22: 60S ribosomal protein L21
+Macromolecule #23: 60S ribosomal protein L22
+Macromolecule #24: 60S ribosomal protein L23
+Macromolecule #25: 60S ribosomal protein L24
+Macromolecule #26: 60S ribosomal protein L23a
+Macromolecule #27: 60S ribosomal protein L26
+Macromolecule #28: 60S ribosomal protein L27
+Macromolecule #29: 60S ribosomal protein L27a
+Macromolecule #30: 60S ribosomal protein L29
+Macromolecule #31: 60S ribosomal protein L30
+Macromolecule #32: 60S ribosomal protein L31
+Macromolecule #33: 60S ribosomal protein L32
+Macromolecule #34: 60S ribosomal protein L35a
+Macromolecule #35: 60S ribosomal protein L34
+Macromolecule #36: 60S ribosomal protein L35
+Macromolecule #37: 60S ribosomal protein L36
+Macromolecule #38: 60S ribosomal protein L37
+Macromolecule #39: 60S ribosomal protein L38
+Macromolecule #40: 60S ribosomal protein L39
+Macromolecule #41: Ubiquitin-60S ribosomal protein L40
+Macromolecule #42: 60S ribosomal protein L41
+Macromolecule #43: 60S ribosomal protein L36a
+Macromolecule #44: 60S ribosomal protein L37a
+Macromolecule #45: 60S ribosomal protein L28
+Macromolecule #46: 60S ribosomal protein L10a
+Macromolecule #48: 40S ribosomal protein SA
+Macromolecule #49: 40S ribosomal protein S3a
+Macromolecule #50: 40S ribosomal protein S3
+Macromolecule #51: 40S ribosomal protein S4, X isoform
+Macromolecule #52: 40S ribosomal protein S5
+Macromolecule #53: 40S ribosomal protein S7
+Macromolecule #54: 40S ribosomal protein S8
+Macromolecule #55: 40S ribosomal protein S10
+Macromolecule #56: 40S ribosomal protein S11
+Macromolecule #57: 40S ribosomal protein S15
+Macromolecule #58: 40S ribosomal protein S16
+Macromolecule #59: 40S ribosomal protein S17
+Macromolecule #60: 40S ribosomal protein S18
+Macromolecule #61: 40S ribosomal protein S19
+Macromolecule #62: 40S ribosomal protein S20
+Macromolecule #63: 40S ribosomal protein S21
+Macromolecule #64: 40S ribosomal protein S23
+Macromolecule #65: 40S ribosomal protein S26
+Macromolecule #66: 40S ribosomal protein S28
+Macromolecule #67: 40S ribosomal protein S29
+Macromolecule #68: Receptor of activated protein C kinase 1
+Macromolecule #69: 40S ribosomal protein S2
+Macromolecule #70: 40S ribosomal protein S6
+Macromolecule #71: 40S ribosomal protein S9
+Macromolecule #72: 40S ribosomal protein S12
+Macromolecule #73: 40S ribosomal protein S13
+Macromolecule #74: 40S ribosomal protein S14
+Macromolecule #75: 40S ribosomal protein S15a
+Macromolecule #76: 40S ribosomal protein S24
+Macromolecule #77: 40S ribosomal protein S25
+Macromolecule #78: 40S ribosomal protein S27
+Macromolecule #79: 40S ribosomal protein S30
+Macromolecule #80: Ubiquitin-40S ribosomal protein S27a
+Macromolecule #81: Proliferation-associated protein 2G4
+Macromolecule #82: Elongation factor 2
+Macromolecule #84: Plasminogen activator inhibitor 1 RNA-binding protein
+Macromolecule #85: MAGNESIUM ION
+Macromolecule #86: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON III (4k x 4k) / Average electron dose: 28.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller



















































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)