+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0968 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of human BAF Base module | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of myeloid progenitor cell differentiation / single stranded viral RNA replication via double stranded DNA intermediate / Positive Regulation of CDH1 Gene Transcription / positive regulation of glucose mediated signaling pathway / positive regulation of transcription of nucleolar large rRNA by RNA polymerase I / Formation of the embryonic stem cell BAF (esBAF) complex / bBAF complex / neural retina development / npBAF complex / blastocyst hatching ...negative regulation of myeloid progenitor cell differentiation / single stranded viral RNA replication via double stranded DNA intermediate / Positive Regulation of CDH1 Gene Transcription / positive regulation of glucose mediated signaling pathway / positive regulation of transcription of nucleolar large rRNA by RNA polymerase I / Formation of the embryonic stem cell BAF (esBAF) complex / bBAF complex / neural retina development / npBAF complex / blastocyst hatching / brahma complex / nBAF complex / negative regulation of androgen receptor signaling pathway / N-acetyltransferase activity / hepatocyte differentiation / EGR2 and SOX10-mediated initiation of Schwann cell myelination / Formation of the canonical BAF (cBAF) complex / Formation of neuronal progenitor and neuronal BAF (npBAF and nBAF) / Formation of the polybromo-BAF (pBAF) complex / Formation of the non-canonical BAF (ncBAF) complex / GBAF complex / regulation of G0 to G1 transition / histone H3K14ac reader activity / nucleosome array spacer activity / Tat protein binding / RNA polymerase I preinitiation complex assembly / RSC-type complex / cellular response to fatty acid / XY body / host-mediated activation of viral transcription / regulation of nucleotide-excision repair / germ cell nucleus / nucleosome disassembly / ATP-dependent chromatin remodeler activity / SWI/SNF complex / regulation of mitotic metaphase/anaphase transition / positive regulation of T cell differentiation / nuclear androgen receptor binding / positive regulation of double-strand break repair / nuclear chromosome / positive regulation of stem cell population maintenance / RUNX1 interacts with co-factors whose precise effect on RUNX1 targets is not known / Regulation of MITF-M-dependent genes involved in pigmentation / regulation of G1/S transition of mitotic cell cycle / histone H4K16ac reader activity / negative regulation of cell differentiation / positive regulation of myoblast differentiation / positive regulation of signal transduction by p53 class mediator / ATP-dependent activity, acting on DNA / positive regulation of Wnt signaling pathway / Chromatin modifying enzymes / DNA polymerase binding / neurogenesis / Interleukin-7 signaling / transcription initiation-coupled chromatin remodeling / nuclear receptor binding / transcription coregulator binding / helicase activity / transcription coregulator activity / positive regulation of cell differentiation / Regulation of endogenous retroelements by Piwi-interacting RNAs (piRNAs) / apoptotic signaling pathway / Formation of the beta-catenin:TCF transactivating complex / DNA integration / negative regulation of cell growth / Negative Regulation of CDH1 Gene Transcription / positive regulation of miRNA transcription / kinetochore / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / fibrillar center / RMTs methylate histone arginines / nuclear matrix / p53 binding / transcription corepressor activity / nervous system development / heterochromatin formation / positive regulation of cold-induced thermogenesis / histone binding / molecular adaptor activity / transcription coactivator activity / chromatin remodeling / signaling receptor binding / negative regulation of cell population proliferation / negative regulation of DNA-templated transcription / apoptotic process / positive regulation of cell population proliferation / chromatin binding / regulation of transcription by RNA polymerase II / centrosome / positive regulation of DNA-templated transcription / chromatin / nucleolus / negative regulation of transcription by RNA polymerase II / ATP hydrolysis activity / positive regulation of transcription by RNA polymerase II / protein-containing complex / : / DNA binding / RNA binding / zinc ion binding Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.0 Å | |||||||||
 Authors Authors | Shuang H / Zihan W / Yuan T / Zishuo Y / Jiali Y / Xinxin W / Jie L / Bijun L / Yanhui X | |||||||||
 Citation Citation |  Journal: Science / Year: 2020 Journal: Science / Year: 2020Title: Structure of nucleosome-bound human BAF complex. Authors: Shuang He / Zihan Wu / Yuan Tian / Zishuo Yu / Jiali Yu / Xinxin Wang / Jie Li / Bijun Liu / Yanhui Xu /  Abstract: Mammalian SWI/SNF family chromatin remodelers, BRG1/BRM-associated factor (BAF) and polybromo-associated BAF (PBAF), regulate chromatin structure and transcription, and their mutations are linked to ...Mammalian SWI/SNF family chromatin remodelers, BRG1/BRM-associated factor (BAF) and polybromo-associated BAF (PBAF), regulate chromatin structure and transcription, and their mutations are linked to cancers. The 3.7-angstrom-resolution cryo-electron microscopy structure of human BAF bound to the nucleosome reveals that the nucleosome is sandwiched by the base and the adenosine triphosphatase (ATPase) modules, which are bridged by the actin-related protein (ARP) module. The ATPase motor is positioned proximal to nucleosomal DNA and, upon ATP hydrolysis, engages with and pumps DNA along the nucleosome. The C-terminal α helix of SMARCB1, enriched in positively charged residues frequently mutated in cancers, mediates interactions with an acidic patch of the nucleosome. AT-rich interactive domain-containing protein 1A (ARID1A) and the SWI/SNF complex subunit SMARCC serve as a structural core and scaffold in the base module organization, respectively. Our study provides structural insights into subunit organization and nucleosome recognition of human BAF complex. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0968.map.gz emd_0968.map.gz | 25.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0968-v30.xml emd-0968-v30.xml emd-0968.xml emd-0968.xml | 14.4 KB 14.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_0968.png emd_0968.png | 5.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0968 http://ftp.pdbj.org/pub/emdb/structures/EMD-0968 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0968 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0968 | HTTPS FTP |
-Related structure data
| Related structure data |  6lthMC  0969C  0970C  0971C  0972C  0973C  0974C  6ltjC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0968.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0968.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.04 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
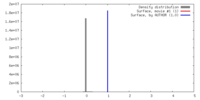
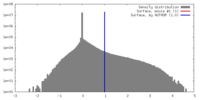
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Structure of nucleosome-bound human BAF Base module
| Entire | Name: Structure of nucleosome-bound human BAF Base module |
|---|---|
| Components |
|
-Supramolecule #1: Structure of nucleosome-bound human BAF Base module
| Supramolecule | Name: Structure of nucleosome-bound human BAF Base module / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#20 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Recombinant expression | Organism:  Homo sapiens (human) / Recombinant cell: HEK293T Homo sapiens (human) / Recombinant cell: HEK293T |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Specialist optics | Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Digitization - Frames/image: 1-32 / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: OTHER |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| CTF correction | Software - Name: Gctf (ver. 1.06) |
|---|---|
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Resolution.type: BY AUTHOR / Resolution: 3.0 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: cryoSPARC (ver. 2.12.4) / Number images used: 197606 |
| Initial angle assignment | Type: OTHER / Software - Name: cryoSPARC (ver. 2.12.4) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION (ver. 3.0.8) |
-Atomic model buiding 1
| Refinement | Space: REAL / Protocol: AB INITIO MODEL |
|---|---|
| Output model |  PDB-6lth: |
 Movie
Movie Controller
Controller

























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)