[English] 日本語
 Yorodumi
Yorodumi- EMDB-0262: III2-IV2 mitochondrial respiratory supercomplex from S. cerevisiae -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0262 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | III2-IV2 mitochondrial respiratory supercomplex from S. cerevisiae | |||||||||
 Map data Map data | The sharpened map of the III2IV2 supercomplex | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationComplex III assembly / : / matrix side of mitochondrial inner membrane / : / mitochondrial respiratory chain complex IV assembly / Respiratory electron transport / mitochondrial respirasome assembly / Mitochondrial protein degradation / mitochondrial respiratory chain complex III assembly / respiratory chain complex IV ...Complex III assembly / : / matrix side of mitochondrial inner membrane / : / mitochondrial respiratory chain complex IV assembly / Respiratory electron transport / mitochondrial respirasome assembly / Mitochondrial protein degradation / mitochondrial respiratory chain complex III assembly / respiratory chain complex IV / respiratory chain complex / cytochrome-c oxidase / respiratory chain complex III / cellular respiration / quinol-cytochrome-c reductase / mitochondrial electron transport, cytochrome c to oxygen / quinol-cytochrome-c reductase activity / cytochrome-c oxidase activity / mitochondrial electron transport, ubiquinol to cytochrome c / mitochondrial crista / ubiquinone binding / ATP synthesis coupled electron transport / enzyme regulator activity / nuclear periphery / proton transmembrane transport / aerobic respiration / respiratory electron transport chain / metalloendopeptidase activity / mitochondrial intermembrane space / 2 iron, 2 sulfur cluster binding / maintenance of translational fidelity / mitochondrial membrane / oxidoreductase activity / mitochondrial inner membrane / copper ion binding / heme binding / mitochondrion / proteolysis / zinc ion binding / membrane / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
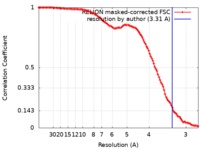
| Method | single particle reconstruction / cryo EM / Resolution: 3.31 Å | |||||||||
 Authors Authors | Hartley AM / Lukoyanova N / Pinotsis N / Marechal A | |||||||||
 Citation Citation |  Journal: Nat Struct Mol Biol / Year: 2019 Journal: Nat Struct Mol Biol / Year: 2019Title: Structure of yeast cytochrome c oxidase in a supercomplex with cytochrome bc. Authors: Andrew M Hartley / Natalya Lukoyanova / Yunyi Zhang / Alfredo Cabrera-Orefice / Susanne Arnold / Brigitte Meunier / Nikos Pinotsis / Amandine Maréchal /    Abstract: Cytochrome c oxidase (complex IV, CIV) is known in mammals to exist independently or in association with other respiratory proteins to form supercomplexes (SCs). In Saccharomyces cerevisiae, CIV is ...Cytochrome c oxidase (complex IV, CIV) is known in mammals to exist independently or in association with other respiratory proteins to form supercomplexes (SCs). In Saccharomyces cerevisiae, CIV is found solely in an SC with cytochrome bc (complex III, CIII). Here, we present the cryogenic electron microscopy (cryo-EM) structure of S. cerevisiae CIV in a IIIIV SC at 3.3 Å resolution. While overall similarity to mammalian homologs is high, we found notable differences in the supernumerary subunits Cox26 and Cox13; the latter exhibits a unique arrangement that precludes CIV dimerization as seen in bovine. A conformational shift in the matrix domain of Cox5A-involved in allosteric inhibition by ATP-may arise from its association with CIII. The CIII-CIV arrangement highlights a conserved interaction interface of CIII, albeit one occupied by complex I in mammalian respirasomes. We discuss our findings in the context of the potential impact of SC formation on CIV regulation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0262.map.gz emd_0262.map.gz | 106.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0262-v30.xml emd-0262-v30.xml emd-0262.xml emd-0262.xml | 30.6 KB 30.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_0262_fsc.xml emd_0262_fsc.xml | 11 KB | Display |  FSC data file FSC data file |
| Images |  emd_0262.png emd_0262.png | 147.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0262 http://ftp.pdbj.org/pub/emdb/structures/EMD-0262 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0262 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0262 | HTTPS FTP |
-Related structure data
| Related structure data |  6hu9MC  0268C  0269C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0262.map.gz / Format: CCP4 / Size: 113.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0262.map.gz / Format: CCP4 / Size: 113.6 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | The sharpened map of the III2IV2 supercomplex | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.3861 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
+Entire : III2-IV2 mitochondrial respiratory supercomplex
+Supramolecule #1: III2-IV2 mitochondrial respiratory supercomplex
+Supramolecule #2: 10-subunit yeast cytochrome bc1
+Supramolecule #3: 12-subunit cytochrome c oxidase with isoform Cox5A
+Macromolecule #1: Cytochrome b-c1 complex subunit 1, mitochondrial
+Macromolecule #2: Cytochrome b-c1 complex subunit 2, mitochondrial
+Macromolecule #3: Cytochrome b
+Macromolecule #4: Cytochrome c1, heme protein, mitochondrial
+Macromolecule #5: Cytochrome b-c1 complex subunit Rieske, mitochondrial
+Macromolecule #6: Cytochrome b-c1 complex subunit 6
+Macromolecule #7: Cytochrome b-c1 complex subunit 7
+Macromolecule #8: Cytochrome b-c1 complex subunit 8
+Macromolecule #9: Cytochrome b-c1 complex subunit 9
+Macromolecule #10: Cytochrome b-c1 complex subunit 10
+Macromolecule #11: Cox1
+Macromolecule #12: Cox2
+Macromolecule #13: Cox3
+Macromolecule #14: Cox4
+Macromolecule #15: Cox5A
+Macromolecule #16: Cox6
+Macromolecule #17: Cox7
+Macromolecule #18: Cox8
+Macromolecule #19: Cox9
+Macromolecule #20: Cox12
+Macromolecule #21: Cox13
+Macromolecule #22: Cox26
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Grid | Model: Quantifoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR / Pretreatment - Pressure: 4.0 kPa |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 92 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV Details: 3 uL of sample applied to negatively glow discharged grid, blot force -10; blotting time 8.5 sec. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 QUANTUM (4k x 4k) / Detector mode: COUNTING / Digitization - Dimensions - Width: 3838 pixel / Digitization - Dimensions - Height: 3710 pixel / Digitization - Sampling interval: 5.0 µm / Average exposure time: 8.0 sec. / Average electron dose: 1.645 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 70.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 130000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)