[English] 日本語
 Yorodumi
Yorodumi- SASDFW3: Cytokine-like nuclear factor dehydrogenase domain, NPAC DH, plus ... -
+ Open data
Open data
- Basic information
Basic information
| Entry | 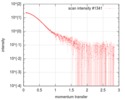 |
|---|---|
 Sample Sample | Cytokine-like nuclear factor dehydrogenase domain, NPAC DH, plus native linker (NPAC delta-205)
|
| Function / homology |  Function and homology information Function and homology informationtranscription elongation-coupled chromatin remodeling / chromatin-protein adaptor activity / nucleosome binding / transcription initiation-coupled chromatin remodeling / NAD binding / NADP binding / nucleosome / histone binding / chromatin binding / chromatin ...transcription elongation-coupled chromatin remodeling / chromatin-protein adaptor activity / nucleosome binding / transcription initiation-coupled chromatin remodeling / NAD binding / NADP binding / nucleosome / histone binding / chromatin binding / chromatin / DNA binding / nucleoplasm / cytosol Similarity search - Function |
| Biological species |  Homo sapiens (human) Homo sapiens (human) |
 Citation Citation |  Journal: Cell Rep / Year: 2019 Journal: Cell Rep / Year: 2019Title: A Tail-Based Mechanism Drives Nucleosome Demethylation by the LSD2/NPAC Multimeric Complex. Authors: Chiara Marabelli / Biagina Marrocco / Simona Pilotto / Sagar Chittori / Sarah Picaud / Sara Marchese / Giuseppe Ciossani / Federico Forneris / Panagis Filippakopoulos / Guy Schoehn / Daniela ...Authors: Chiara Marabelli / Biagina Marrocco / Simona Pilotto / Sagar Chittori / Sarah Picaud / Sara Marchese / Giuseppe Ciossani / Federico Forneris / Panagis Filippakopoulos / Guy Schoehn / Daniela Rhodes / Sriram Subramaniam / Andrea Mattevi /       Abstract: LSD1 and LSD2 are homologous histone demethylases with opposite biological outcomes related to chromatin silencing and transcription elongation, respectively. Unlike LSD1, LSD2 nucleosome-demethylase ...LSD1 and LSD2 are homologous histone demethylases with opposite biological outcomes related to chromatin silencing and transcription elongation, respectively. Unlike LSD1, LSD2 nucleosome-demethylase activity relies on a specific linker peptide from the multidomain protein NPAC. We used single-particle cryoelectron microscopy (cryo-EM), in combination with kinetic and mutational analysis, to analyze the mechanisms underlying the function of the human LSD2/NPAC-linker/nucleosome complex. Weak interactions between LSD2 and DNA enable multiple binding modes for the association of the demethylase to the nucleosome. The demethylase thereby captures mono- and dimethyl Lys4 of the H3 tail to afford histone demethylation. Our studies also establish that the dehydrogenase domain of NPAC serves as a catalytically inert oligomerization module. While LSD1/CoREST forms a nucleosome docking platform at silenced gene promoters, LSD2/NPAC is a multifunctional enzyme complex with flexible linkers, tailored for rapid chromatin modification, in conjunction with the advance of the RNA polymerase on actively transcribed genes. |
 Contact author Contact author |
|
- Structure visualization
Structure visualization
- Downloads & links
Downloads & links
-Data source
| SASBDB page |  SASDFW3 SASDFW3 |
|---|
-Related structure data
| Related structure data |  4704C  4705C  4710C  4711C  4712C  6r1tC  6r1uC  6r25C C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- External links
External links
| Related items in Molecule of the Month |
|---|
-Models
- Sample
Sample
 Sample Sample | Name: Cytokine-like nuclear factor dehydrogenase domain, NPAC DH, plus native linker (NPAC delta-205) Specimen concentration: 1.25 mg/ml |
|---|---|
| Buffer | Name: 15 mM HEPES, 200 mM NaCl / pH: 7.3 |
| Entity #1519 | Name: NPAC delta205 / Type: protein / Description: NPAC linker+DH (delta-205) / Formula weight: 37.438 / Num. of mol.: 4 / Source: Homo sapiens / References: UniProt: Q49A26 Sequence: TASEPVKDAD PHFHHFLLSQ TEKPAVCYQA ITKKLKICEE ETGSTSIQAA DSTAVNGSIT PTDKKIGFLG LGLMGSGIVS NLLKMGHTVT VWNRTAEKCD LFIQEGARLG RTPAEVVSTC DITFACVSDP KAAKDLVLGP SGVLQGIRPG KCYVDMSTVD ADTVTELAQV ...Sequence: TASEPVKDAD PHFHHFLLSQ TEKPAVCYQA ITKKLKICEE ETGSTSIQAA DSTAVNGSIT PTDKKIGFLG LGLMGSGIVS NLLKMGHTVT VWNRTAEKCD LFIQEGARLG RTPAEVVSTC DITFACVSDP KAAKDLVLGP SGVLQGIRPG KCYVDMSTVD ADTVTELAQV IVSRGGRFLE APVSGNQQLS NDGMLVILAA GDRGLYEDCS SCFQAMGKTS FFLGEVGNAA KMMLIVNMVQ GSFMATIAEG LTLAQVTGQS QQTLLDILNQ GQLASIFLDQ KCQNILQGNF KPDFYLKYIQ KDLRLAIALG DAVNHPTPMA AAANEVYKRA KALDQSDNDM SAVYRAYIH |
-Experimental information
| Beam | Instrument name: ESRF BM29 / City: Grenoble / 国: France  / Type of source: X-ray synchrotron / Wavelength: 0.1 Å / Dist. spec. to detc.: 2.87 mm / Type of source: X-ray synchrotron / Wavelength: 0.1 Å / Dist. spec. to detc.: 2.87 mm | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Detector | Name: Pilatus 1M / Type: Dectris / Pixsize x: 172 mm | ||||||||||||||||||||||||||||||
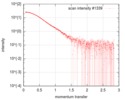
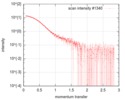
| Scan | Measurement date: Jan 26, 2018 / Storage temperature: 4 °C / Cell temperature: 20 °C / Exposure time: 1 sec. / Number of frames: 222 / Unit: 1/nm /
| ||||||||||||||||||||||||||||||
| Distance distribution function P(R) |
| ||||||||||||||||||||||||||||||
| Result | Comments: The protein construct encompasses the dehydrogenase domain plus an additional upstream native-sequence amino acid linker (compare to the isolated dehydrogenase domain, SASDFV3, NPAC delta-261).
|
 Movie
Movie Controller
Controller