[English] 日本語
 Yorodumi
Yorodumi- PDB-9qkt: Cryo-EM structure of the CDK11B-cyclin L2-SAP30BP bound to OTS964... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 9qkt | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the CDK11B-cyclin L2-SAP30BP bound to OTS964 (conformation 1) | ||||||||||||||||||||||||||||||
 Components Components |
| ||||||||||||||||||||||||||||||
 Keywords Keywords | SPLICING / kinase / cyclin-dependent kinase / inhibitor / transcription / molecular complex | ||||||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmitotic sister chromatid cohesion, centromeric / regulation of mRNA processing / regulation of centrosome cycle / intermediate filament cytoskeleton / [RNA-polymerase]-subunit kinase / cyclin-dependent protein serine/threonine kinase regulator activity / regulation of RNA splicing / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity / cyclin-dependent protein kinase holoenzyme complex ...mitotic sister chromatid cohesion, centromeric / regulation of mRNA processing / regulation of centrosome cycle / intermediate filament cytoskeleton / [RNA-polymerase]-subunit kinase / cyclin-dependent protein serine/threonine kinase regulator activity / regulation of RNA splicing / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity / cyclin-dependent protein kinase holoenzyme complex / Recruitment of mitotic centrosome proteins and complexes / regulation of cell growth / NoRC negatively regulates rRNA expression / response to virus / mitotic cell cycle / regulation of apoptotic process / protein phosphorylation / protein kinase activity / regulation of cell cycle / nuclear speck / protein serine kinase activity / protein serine/threonine kinase activity / apoptotic process / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / RNA binding / nucleoplasm / ATP binding / nucleus / cytoplasm Similarity search - Function | ||||||||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.4 Å | ||||||||||||||||||||||||||||||
 Authors Authors | McGeoch, A.J.S. / Cushing, V.I. / Cronin, N.B. / Alfieri, C. / Greber, B.J. | ||||||||||||||||||||||||||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||||||||||||||||||||||||||
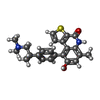
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Cryo-EM structures of the CDK11-cyclin L-SAP30BP complex reveal mechanisms of CDK11 regulation. Authors: Amy J S McGeoch / Victoria I Cushing / Theodoros I Roumeliotis / Nora B Cronin / Stephen J Hearnshaw / Jyoti S Choudhary / Claudio Alfieri / Basil J Greber /  Abstract: The cyclin-dependent kinase CDK11 functions in transcription, mitotic progression, and mRNA splicing. Specifically, spliceosome activation during the B to B transition depends on phosphorylation of ...The cyclin-dependent kinase CDK11 functions in transcription, mitotic progression, and mRNA splicing. Specifically, spliceosome activation during the B to B transition depends on phosphorylation of the U2 snRNP component SF3B1 by the CDK11-cyclin L-SAP30BP complex. Here, we present the structure of this spliceosome-activating CDK-cyclin complex, determined by cryogenic electron microscopy at 2.3 Å resolution. Our structure and biochemical experiments show that SAP30BP forms extensive interactions with cyclin L2, thereby stabilising it, and forms critical interactions with the C-terminal kinase lobe of CDK11 that promote complex assembly. Destabilisation of cyclin L2 in the absence of SAP30BP suggests that these principles are applicable to all CDK11-cyclin L complexes. Furthermore, we identify a pseudo-substrate sequence near the CDK11 C-terminus and provide evidence for a role of this segment in CDK11 auto-regulation. Finally, the structure of the CDK11-cyclin L-SAP30BP complex bound to the clinical high-affinity CDK11 inhibitor OTS964 and a comparison to OTS964-bound off-target complexes provide insight into the mechanism of OTS964 selectivity and specificity. | ||||||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  9qkt.cif.gz 9qkt.cif.gz | 164.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb9qkt.ent.gz pdb9qkt.ent.gz | 118.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  9qkt.json.gz 9qkt.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/qk/9qkt https://data.pdbj.org/pub/pdb/validation_reports/qk/9qkt ftp://data.pdbj.org/pub/pdb/validation_reports/qk/9qkt ftp://data.pdbj.org/pub/pdb/validation_reports/qk/9qkt | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  53221MC  9qjjC  9qjnC  9qkzC  9ql1C C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 34186.633 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: SAP30BP, HCNGP, HTRG, HTRP / Cell line (production host): Hi5 / Production host: Homo sapiens (human) / Gene: SAP30BP, HCNGP, HTRG, HTRP / Cell line (production host): Hi5 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: Q9UHR5 Trichoplusia ni (cabbage looper) / References: UniProt: Q9UHR5 |
|---|---|
| #2: Protein | Mass: 35690.453 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CCNL2, SB138 / Cell line (production host): Hi5 / Production host: Homo sapiens (human) / Gene: CCNL2, SB138 / Cell line (production host): Hi5 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: Q96S94 Trichoplusia ni (cabbage looper) / References: UniProt: Q96S94 |
| #3: Protein | Mass: 50050.590 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: CDK11B, CDC2L1, CDK11, PITSLREA, PK58 / Cell line (production host): Hi5 / Production host: Homo sapiens (human) / Gene: CDK11B, CDC2L1, CDK11, PITSLREA, PK58 / Cell line (production host): Hi5 / Production host:  Trichoplusia ni (cabbage looper) / References: UniProt: P21127, cyclin-dependent kinase Trichoplusia ni (cabbage looper) / References: UniProt: P21127, cyclin-dependent kinase |
| #4: Chemical | ChemComp-NK3 / |
| #5: Water | ChemComp-HOH / |
| Has ligand of interest | Y |
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: CDK11B-cyclin L2-SAP30BP-OTS964 complex / Type: COMPLEX / Entity ID: #1-#3 / Source: RECOMBINANT | ||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.12 MDa / Experimental value: NO | ||||||||||||||||||||||||||||||
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||||||||
| Source (recombinant) | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) | ||||||||||||||||||||||||||||||
| Buffer solution | pH: 7.9 | ||||||||||||||||||||||||||||||
| Buffer component |
| ||||||||||||||||||||||||||||||
| Specimen | Conc.: 0.5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | ||||||||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: UltrAuFoil R1.2/1.3 | ||||||||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 278 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: TFS KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 165000 X / Nominal defocus max: 1800 nm / Nominal defocus min: 800 nm / Cs: 2.7 mm / C2 aperture diameter: 50 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Electron dose: 70 e/Å2 / Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Num. of real images: 13829 |
| EM imaging optics | Energyfilter name: GIF Bioquantum / Energyfilter slit width: 20 eV |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | ||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 286108 / Symmetry type: POINT | ||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL | ||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Details: Coordinates based on prior cryo-EM reconstruction of this complex. Source name: Other / Type: experimental model | ||||||||||||||||||||||||||||||||||||||||||||
| Refinement | Highest resolution: 2.4 Å Stereochemistry target values: REAL-SPACE (WEIGHTED MAP SUM AT ATOM CENTERS) | ||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller





 PDBj
PDBj