[English] 日本語
 Yorodumi
Yorodumi- EMDB-53221: Cryo-EM structure of the CDK11B-cyclin L2-SAP30BP bound to OTS964... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the CDK11B-cyclin L2-SAP30BP bound to OTS964 (conformation 1) | |||||||||
 Map data Map data | Post-processed (sharpened, filtered) cryo-EM map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | kinase / cyclin-dependent kinase / inhibitor / transcription / molecular complex / SPLICING | |||||||||
| Function / homology |  Function and homology information Function and homology informationmitotic sister chromatid cohesion, centromeric / regulation of mRNA processing / regulation of centrosome cycle / intermediate filament cytoskeleton / [RNA-polymerase]-subunit kinase / cyclin-dependent protein serine/threonine kinase regulator activity / regulation of RNA splicing / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity / cyclin-dependent protein kinase holoenzyme complex ...mitotic sister chromatid cohesion, centromeric / regulation of mRNA processing / regulation of centrosome cycle / intermediate filament cytoskeleton / [RNA-polymerase]-subunit kinase / cyclin-dependent protein serine/threonine kinase regulator activity / regulation of RNA splicing / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity / cyclin-dependent protein kinase holoenzyme complex / Recruitment of mitotic centrosome proteins and complexes / regulation of cell growth / NoRC negatively regulates rRNA expression / response to virus / mitotic cell cycle / regulation of apoptotic process / protein phosphorylation / protein kinase activity / regulation of cell cycle / nuclear speck / protein serine kinase activity / protein serine/threonine kinase activity / apoptotic process / regulation of transcription by RNA polymerase II / regulation of DNA-templated transcription / RNA binding / nucleoplasm / ATP binding / nucleus / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.4 Å | |||||||||
 Authors Authors | McGeoch AJS / Cushing VI / Cronin NB / Alfieri C / Greber BJ | |||||||||
| Funding support |  United Kingdom, 1 items United Kingdom, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2026 Journal: Nat Commun / Year: 2026Title: Cryo-EM structures of the CDK11-cyclin L-SAP30BP complex reveal mechanisms of CDK11 regulation. Authors: Amy J S McGeoch / Victoria I Cushing / Theodoros I Roumeliotis / Nora B Cronin / Stephen J Hearnshaw / Jyoti S Choudhary / Claudio Alfieri / Basil J Greber /  Abstract: The cyclin-dependent kinase CDK11 functions in transcription, mitotic progression, and mRNA splicing. Specifically, spliceosome activation during the B to B transition depends on phosphorylation of ...The cyclin-dependent kinase CDK11 functions in transcription, mitotic progression, and mRNA splicing. Specifically, spliceosome activation during the B to B transition depends on phosphorylation of the U2 snRNP component SF3B1 by the CDK11-cyclin L-SAP30BP complex. Here, we present the structure of this spliceosome-activating CDK-cyclin complex, determined by cryogenic electron microscopy at 2.3 Å resolution. Our structure and biochemical experiments show that SAP30BP forms extensive interactions with cyclin L2, thereby stabilising it, and forms critical interactions with the C-terminal kinase lobe of CDK11 that promote complex assembly. Destabilisation of cyclin L2 in the absence of SAP30BP suggests that these principles are applicable to all CDK11-cyclin L complexes. Furthermore, we identify a pseudo-substrate sequence near the CDK11 C-terminus and provide evidence for a role of this segment in CDK11 auto-regulation. Finally, the structure of the CDK11-cyclin L-SAP30BP complex bound to the clinical high-affinity CDK11 inhibitor OTS964 and a comparison to OTS964-bound off-target complexes provide insight into the mechanism of OTS964 selectivity and specificity. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_53221.map.gz emd_53221.map.gz | 25.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-53221-v30.xml emd-53221-v30.xml emd-53221.xml emd-53221.xml | 31.9 KB 31.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_53221.png emd_53221.png | 113.4 KB | ||
| Masks |  emd_53221_msk_1.map emd_53221_msk_1.map | 27 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-53221.cif.gz emd-53221.cif.gz | 8.1 KB | ||
| Others |  emd_53221_additional_1.map.gz emd_53221_additional_1.map.gz emd_53221_additional_2.map.gz emd_53221_additional_2.map.gz emd_53221_half_map_1.map.gz emd_53221_half_map_1.map.gz emd_53221_half_map_2.map.gz emd_53221_half_map_2.map.gz | 25.2 MB 25.2 MB 20.7 MB 20.7 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-53221 http://ftp.pdbj.org/pub/emdb/structures/EMD-53221 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53221 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-53221 | HTTPS FTP |
-Related structure data
| Related structure data |  9qktMC  9ql1MC  9qjjC  9qjnC  9qkzC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_53221.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_53221.map.gz / Format: CCP4 / Size: 27 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed (sharpened, filtered) cryo-EM map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.02 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_53221_msk_1.map emd_53221_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Post-processed cryo-EM map with lower sharpening b-factor (b = -10)
| File | emd_53221_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed cryo-EM map with lower sharpening b-factor (b = -10) | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Post-processed cryo-EM map with lower sharpening b-factor (b...
| File | emd_53221_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed cryo-EM map with lower sharpening b-factor (b = -10) and low-pass filtered to 3.3 Angstroms resolution | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Unfiltered half-map from refinement.
| File | emd_53221_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map from refinement. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Unfiltered half-map from refinement.
| File | emd_53221_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map from refinement. | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : CDK11B-cyclin L2-SAP30BP-OTS964 complex
| Entire | Name: CDK11B-cyclin L2-SAP30BP-OTS964 complex |
|---|---|
| Components |
|
-Supramolecule #1: CDK11B-cyclin L2-SAP30BP-OTS964 complex
| Supramolecule | Name: CDK11B-cyclin L2-SAP30BP-OTS964 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 120 KDa |
-Macromolecule #1: SAP30-binding protein
| Macromolecule | Name: SAP30-binding protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 34.186633 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: SNAMAGKKNV LSSLAVYAED SEPESDGEAG IEAVGSAAEE KGGLVSDAYG EDDFSRLGGD EDGYEEEEDE NSRQSEDDDS ETEKPEADD PKDNTEAEKR DPQELVASFS ERVRNMSPDE IKIPPEPPGR CSNHLQDKIQ KLYERKIKEG MDMNYIIQRK K EFRNPSIY ...String: SNAMAGKKNV LSSLAVYAED SEPESDGEAG IEAVGSAAEE KGGLVSDAYG EDDFSRLGGD EDGYEEEEDE NSRQSEDDDS ETEKPEADD PKDNTEAEKR DPQELVASFS ERVRNMSPDE IKIPPEPPGR CSNHLQDKIQ KLYERKIKEG MDMNYIIQRK K EFRNPSIY EKLIQFCAID ELGTNYPKDM FDPHGWSEDS YYEALAKAQK IEMDKLEKAK KERTKIEFVT GTKKGTTTNA TS TTTTTAS TAVADAQKRK SKWDSAIPVT TIAQPTILTT TATLPAVVTV TTSASGSKTT VISAVGTIVK KAKQ UniProtKB: SAP30-binding protein |
-Macromolecule #2: Cyclin-L2
| Macromolecule | Name: Cyclin-L2 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 35.690453 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: SNAMAAAAAA AGAAGSAAPA AAAGAPGSGG APSGSQGVLI GDRLYSGVLI TLENCLLPDD KLRFTPSMSS GLDTDTETDL RVVGCELIQ AAGILLRLPQ VAMATGQVLF QRFFYTKSFV KHSMEHVSMA CVHLASKIEE APRRIRDVIN VFHRLRQLRD K KKPVPLLL ...String: SNAMAAAAAA AGAAGSAAPA AAAGAPGSGG APSGSQGVLI GDRLYSGVLI TLENCLLPDD KLRFTPSMSS GLDTDTETDL RVVGCELIQ AAGILLRLPQ VAMATGQVLF QRFFYTKSFV KHSMEHVSMA CVHLASKIEE APRRIRDVIN VFHRLRQLRD K KKPVPLLL DQDYVNLKNQ IIKAERRVLK ELGFCVHVKH PHKIIVMYLQ VLECERNQHL VQTSWNYMND SLRTDVFVRF QP ESIACAC IYLAARTLEI PLPNRPHWFL LFGATEEEIQ EICLKILQLY ARKKVDLTHL EGEVEKRKHA IEEAKAQARG LLP GG UniProtKB: Cyclin-L2 |
-Macromolecule #3: Cyclin-dependent kinase 11B
| Macromolecule | Name: Cyclin-dependent kinase 11B / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: cyclin-dependent kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 50.05059 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: SNAMSEDEER ENENHLLVVP ESRFDRDSGE SEEAEEEVGE GTPQSSALTE GDYVPDSPAL SPIELKQELP KYLPALQGCR SVEEFQCLN RIEEGTYGVV YRAKDKKTDE IVALKRLKME KEKEGFPITS LREINTILKA QHPNIVTVRE IVVGSNMDKI Y IVMNYVEH ...String: SNAMSEDEER ENENHLLVVP ESRFDRDSGE SEEAEEEVGE GTPQSSALTE GDYVPDSPAL SPIELKQELP KYLPALQGCR SVEEFQCLN RIEEGTYGVV YRAKDKKTDE IVALKRLKME KEKEGFPITS LREINTILKA QHPNIVTVRE IVVGSNMDKI Y IVMNYVEH DLKSLMETMK QPFLPGEVKT LMIQLLRGVK HLHDNWILHR DLKTSNLLLS HAGILKVGDF GLAREYGSPL KA Y(TPO)PVVVT LWYRAPELLL GAKEYSTAVD MWSVGCIFGE LLTQKPLFPG KSEIDQINKV FKDLGTPSEK IWPGYSELP AVKKMTFSEH PYNNLRKRFG ALLSDQGFDL MNKFLTYFPG RRISAEDGLK HEYFRETPLP IDPSMFPTWP AKSEQQRVKR GT(SEP)PRPPEG GLGYSQLGDD DLKETGFHLT TTNQGASAAG PGFSLKF UniProtKB: Cyclin-dependent kinase 11B |
-Macromolecule #4: OTS964
| Macromolecule | Name: OTS964 / type: ligand / ID: 4 / Number of copies: 1 / Formula: NK3 |
|---|---|
| Molecular weight | Theoretical: 392.514 Da |
| Chemical component information | 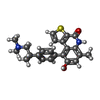 ChemComp-NK3: |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 80 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.5 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
| ||||||||||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 50 sec. | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 278 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number real images: 13829 / Average electron dose: 70.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.8 µm / Nominal defocus min: 0.8 µm / Nominal magnification: 165000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | Chain - Source name: Other / Chain - Initial model type: experimental model Details: Coordinates based on prior cryo-EM reconstruction of this complex. |
|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
| Output model |  PDB-9qkt:  PDB-9ql1: |
 Movie
Movie Controller
Controller










 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)




























































