[English] 日本語
 Yorodumi
Yorodumi- PDB-8ki3: Structure of the human ATP synthase bound to bedaquiline (composite) -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ki3 | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the human ATP synthase bound to bedaquiline (composite) | |||||||||||||||||||||
 Components Components |
| |||||||||||||||||||||
 Keywords Keywords | MEMBRANE PROTEIN / ATP synthase / Human / cryo-EM | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationmitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / negative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / Cristae formation / estradiol binding / : / : / negative regulation of mitochondrial ATP synthesis coupled proton transport / angiostatin binding ...mitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / negative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / Cristae formation / estradiol binding / : / : / negative regulation of mitochondrial ATP synthesis coupled proton transport / angiostatin binding / negative regulation of hydrolase activity / ATP biosynthetic process / mitochondrial depolarization / ATPase inhibitor activity / positive regulation of type 2 mitophagy / mitochondrial proton-transporting ATP synthase complex assembly / Mitochondrial protein import / positive regulation of mitochondrial outer membrane permeabilization involved in apoptotic signaling pathway / cellular response to interleukin-7 / proton channel activity / oxidative phosphorylation / heme biosynthetic process / response to copper ion / response to muscle activity / negative regulation of endothelial cell proliferation / proton transmembrane transporter activity / cellular response to cytokine stimulus / proton motive force-driven ATP synthesis / proton-transporting two-sector ATPase complex, proton-transporting domain / MHC class I protein binding / proton motive force-driven mitochondrial ATP synthesis / mitochondrial nucleoid / positive regulation of blood vessel endothelial cell migration / proton-transporting ATPase activity, rotational mechanism / response to hyperoxia / cellular response to dexamethasone stimulus / H+-transporting two-sector ATPase / proton-transporting ATP synthase complex / proton-transporting ATP synthase activity, rotational mechanism / cellular response to nitric oxide / proton transmembrane transport / substantia nigra development / enzyme inhibitor activity / Mitochondrial protein degradation / reactive oxygen species metabolic process / cellular response to cAMP / aerobic respiration / regulation of intracellular pH / erythrocyte differentiation / generation of precursor metabolites and energy / lipid metabolic process / Transcriptional activation of mitochondrial biogenesis / ADP binding / mitochondrial membrane / fibrillar center / osteoblast differentiation / ATPase binding / protease binding / angiogenesis / nuclear membrane / response to ethanol / calmodulin binding / mitochondrial inner membrane / membrane raft / mitochondrial matrix / hydrolase activity / lipid binding / protein-containing complex binding / structural molecule activity / enzyme binding / cell surface / ATP hydrolysis activity / protein-containing complex / mitochondrion / RNA binding / extracellular exosome / ATP binding / identical protein binding / membrane / nucleus / plasma membrane / cytoplasm Similarity search - Function | |||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 2.89 Å | |||||||||||||||||||||
 Authors Authors | Lai, Y. / Zhang, Y. / Gong, H. | |||||||||||||||||||||
| Funding support |  China, 3items China, 3items
| |||||||||||||||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Inhibition of M. tuberculosis and human ATP synthase by BDQ and TBAJ-587. Authors: Yuying Zhang / Yuezheng Lai / Shan Zhou / Ting Ran / Yue Zhang / Ziqing Zhao / Ziyan Feng / Long Yu / Jinxu Xu / Kun Shi / Jianyun Wang / Yu Pang / Liang Li / Hongming Chen / Luke W Guddat / ...Authors: Yuying Zhang / Yuezheng Lai / Shan Zhou / Ting Ran / Yue Zhang / Ziqing Zhao / Ziyan Feng / Long Yu / Jinxu Xu / Kun Shi / Jianyun Wang / Yu Pang / Liang Li / Hongming Chen / Luke W Guddat / Yan Gao / Fengjiang Liu / Zihe Rao / Hongri Gong /   Abstract: Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation of Mycobacterium tuberculosis by inhibiting ATP synthase. ...Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation of Mycobacterium tuberculosis by inhibiting ATP synthase. However, BDQ also inhibits human ATP synthase. At present, how these compounds interact with either M. tuberculosis ATP synthase or human ATP synthase is unclear. Here we present cryogenic electron microscopy structures of M. tuberculosis ATP synthase with and without BDQ and TBAJ-587 bound, and human ATP synthase bound to BDQ. The two inhibitors interact with subunit a and the c-ring at the leading site, c-only sites and lagging site in M. tuberculosis ATP synthase, showing that BDQ and TBAJ-587 have similar modes of action. The quinolinyl and dimethylamino units of the compounds make extensive contacts with the protein. The structure of human ATP synthase in complex with BDQ reveals that the BDQ-binding site is similar to that observed for the leading site in M. tuberculosis ATP synthase, and that the quinolinyl unit also interacts extensively with the human enzyme. This study will improve researchers' understanding of the similarities and differences between human ATP synthase and M. tuberculosis ATP synthase in terms of the mode of BDQ binding, and will allow the rational design of novel diarylquinolines as anti-tuberculosis drugs. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ki3.cif.gz 8ki3.cif.gz | 846 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ki3.ent.gz pdb8ki3.ent.gz | 696.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ki3.json.gz 8ki3.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ki/8ki3 https://data.pdbj.org/pub/pdb/validation_reports/ki/8ki3 ftp://data.pdbj.org/pub/pdb/validation_reports/ki/8ki3 ftp://data.pdbj.org/pub/pdb/validation_reports/ki/8ki3 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  37251MC  8j0sC  8j0tC  8j57C  8j58C  8jr0C  8jr1C  8khfC C: citing same article ( M: map data used to model this data |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-ATP synthase subunit ... , 12 types, 16 molecules ABCDEFGOHIMNPRST
| #1: Protein | Mass: 55276.160 Da / Num. of mol.: 3 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P25705 Homo sapiens (human) / References: UniProt: P25705#2: Protein | Mass: 51821.965 Da / Num. of mol.: 3 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P06576 Homo sapiens (human) / References: UniProt: P06576#3: Protein | | Mass: 30207.752 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P36542 Homo sapiens (human) / References: UniProt: P36542#5: Protein | | Mass: 20904.488 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P48047 Homo sapiens (human) / References: UniProt: P48047#7: Protein | | Mass: 15029.817 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P30049 Homo sapiens (human) / References: UniProt: P30049#8: Protein | | Mass: 5790.779 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P56381 Homo sapiens (human) / References: UniProt: P56381#10: Protein | | Mass: 18383.982 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: O75947 Homo sapiens (human) / References: UniProt: O75947#11: Protein | | Mass: 24833.102 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P00846 Homo sapiens (human) / References: UniProt: P00846#12: Protein | | Mass: 6673.053 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P56378 Homo sapiens (human) / References: UniProt: P56378#14: Protein | | Mass: 10804.686 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P56134 Homo sapiens (human) / References: UniProt: P56134#15: Protein | | Mass: 11309.226 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: O75964 Homo sapiens (human) / References: UniProt: O75964#16: Protein | | Mass: 7947.215 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P56385 Homo sapiens (human) / References: UniProt: P56385 |
|---|
-Protein , 3 types, 3 molecules JQL
| #4: Protein | Mass: 9540.627 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: Q9UII2 Homo sapiens (human) / References: UniProt: Q9UII2 |
|---|---|
| #13: Protein | Mass: 8000.634 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P03928 Homo sapiens (human) / References: UniProt: P03928 |
| #17: Protein | Mass: 12606.499 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P18859 Homo sapiens (human) / References: UniProt: P18859 |
-ATP synthase F(0) complex subunit ... , 2 types, 9 molecules 12345678K
| #6: Protein | Mass: 7610.954 Da / Num. of mol.: 8 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P05496 Homo sapiens (human) / References: UniProt: P05496#9: Protein | | Mass: 24658.586 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  Homo sapiens (human) / References: UniProt: P24539 Homo sapiens (human) / References: UniProt: P24539 |
|---|
-Non-polymers , 4 types, 11 molecules 


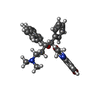



| #18: Chemical | | #19: Chemical | ChemComp-MG / #20: Chemical | #21: Chemical | ChemComp-BQ1 / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | N |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: human ATP synthase / Type: COMPLEX / Entity ID: #1-#17 / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Buffer solution | pH: 7.4 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES |
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal defocus max: 2400 nm / Nominal defocus min: 1200 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: FEI FALCON IV (4k x 4k) |
- Processing
Processing
| EM software | Name: PHENIX / Category: model refinement | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 2.89 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 84037 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller

















 PDBj
PDBj






