[English] 日本語
 Yorodumi
Yorodumi- PDB-7nwh: Mammalian pre-termination 80S ribosome with eRF1 and eRF3 bound b... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7nwh | ||||||
|---|---|---|---|---|---|---|---|
| Title | Mammalian pre-termination 80S ribosome with eRF1 and eRF3 bound by Blasticidin S. | ||||||
 Components Components |
| ||||||
 Keywords Keywords | RIBOSOME / Inhibitor 80S Termination Complex Blasticidin S Translation NMD | ||||||
| Function / homology |  Function and homology information Function and homology informationProtein hydroxylation / L13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Major pathway of rRNA processing in the nucleolus and cytosol / Formation of a pool of free 40S subunits / GTP hydrolysis and joining of the 60S ribosomal subunit / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Translation initiation complex formation / Formation of the ternary complex, and subsequently, the 43S complex ...Protein hydroxylation / L13a-mediated translational silencing of Ceruloplasmin expression / SRP-dependent cotranslational protein targeting to membrane / Major pathway of rRNA processing in the nucleolus and cytosol / Formation of a pool of free 40S subunits / GTP hydrolysis and joining of the 60S ribosomal subunit / Nonsense Mediated Decay (NMD) independent of the Exon Junction Complex (EJC) / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Translation initiation complex formation / Formation of the ternary complex, and subsequently, the 43S complex / Ribosomal scanning and start codon recognition / Translesion synthesis by REV1 / Recognition of DNA damage by PCNA-containing replication complex / Translesion Synthesis by POLH / Downregulation of ERBB4 signaling / Spry regulation of FGF signaling / Downregulation of ERBB2:ERBB3 signaling / NOD1/2 Signaling Pathway / APC/C:Cdc20 mediated degradation of Cyclin B / APC-Cdc20 mediated degradation of Nek2A / EGFR downregulation / TCF dependent signaling in response to WNT / NRIF signals cell death from the nucleus / p75NTR recruits signalling complexes / NF-kB is activated and signals survival / Activated NOTCH1 Transmits Signal to the Nucleus / Downregulation of TGF-beta receptor signaling / TGF-beta receptor signaling in EMT (epithelial to mesenchymal transition) / Downregulation of SMAD2/3:SMAD4 transcriptional activity / SMAD2/SMAD3:SMAD4 heterotrimer regulates transcription / Senescence-Associated Secretory Phenotype (SASP) / activated TAK1 mediates p38 MAPK activation / JNK (c-Jun kinases) phosphorylation and activation mediated by activated human TAK1 / Regulation of FZD by ubiquitination / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Regulation of TNFR1 signaling / TNFR1-induced NF-kappa-B signaling pathway / Translesion synthesis by POLK / Translesion synthesis by POLI / Regulation of necroptotic cell death / HDR through Homologous Recombination (HRR) / Josephin domain DUBs / Recruitment and ATM-mediated phosphorylation of repair and signaling proteins at DNA double strand breaks / Processing of DNA double-strand break ends / Gap-filling DNA repair synthesis and ligation in GG-NER / Dual Incision in GG-NER / Fanconi Anemia Pathway / Regulation of TP53 Activity through Phosphorylation / Regulation of TP53 Degradation / Regulation of TP53 Activity through Methylation / Negative regulation of MET activity / PTK6 Regulates RTKs and Their Effectors AKT1 and DOK1 / Downregulation of ERBB2 signaling / E3 ubiquitin ligases ubiquitinate target proteins / Regulation of PTEN localization / ER Quality Control Compartment (ERQC) / Regulation of expression of SLITs and ROBOs / Interferon alpha/beta signaling / Endosomal Sorting Complex Required For Transport (ESCRT) / Activation of IRF3, IRF7 mediated by TBK1, IKKε (IKBKE) / IKK complex recruitment mediated by RIP1 / IRAK2 mediated activation of TAK1 complex / TRAF6-mediated induction of TAK1 complex within TLR4 complex / Alpha-protein kinase 1 signaling pathway / RAS processing / Pexophagy / Negative regulation of FLT3 / IRAK2 mediated activation of TAK1 complex upon TLR7/8 or 9 stimulation / Regulation of NF-kappa B signaling / Regulation of TBK1, IKKε (IKBKE)-mediated activation of IRF3, IRF7 / Regulation of pyruvate metabolism / SCF-beta-TrCP mediated degradation of Emi1 / MAP3K8 (TPL2)-dependent MAPK1/3 activation / Ovarian tumor domain proteases / Cyclin D associated events in G1 / Regulation of BACH1 activity / Regulation of innate immune responses to cytosolic DNA / Negative regulation of FGFR1 signaling / Negative regulation of FGFR2 signaling / Negative regulation of FGFR3 signaling / Negative regulation of FGFR4 signaling / Negative regulation of MAPK pathway / Formation of Incision Complex in GG-NER / Synthesis of active ubiquitin: roles of E1 and E2 enzymes / Inactivation of CSF3 (G-CSF) signaling / Termination of translesion DNA synthesis / Iron uptake and transport / Negative regulators of DDX58/IFIH1 signaling / Deactivation of the beta-catenin transactivating complex / Metalloprotease DUBs / Major pathway of rRNA processing in the nucleolus and cytosol / GTP hydrolysis and joining of the 60S ribosomal subunit / Formation of TC-NER Pre-Incision Complex / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / Autodegradation of the E3 ubiquitin ligase COP1 Similarity search - Function | ||||||
| Biological species |  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 4.1 Å | ||||||
 Authors Authors | Powers, K.T. / Yadav, S.K.N. / Bufton, J.C. / Schaffitzel, C. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: Nucleic Acids Res / Year: 2021 Journal: Nucleic Acids Res / Year: 2021Title: Blasticidin S inhibits mammalian translation and enhances production of protein encoded by nonsense mRNA. Authors: Kyle T Powers / Flint Stevenson-Jones / Sathish K N Yadav / Beate Amthor / Joshua C Bufton / Ufuk Borucu / Dakang Shen / Jonas P Becker / Daria Lavysh / Matthias W Hentze / Andreas E Kulozik ...Authors: Kyle T Powers / Flint Stevenson-Jones / Sathish K N Yadav / Beate Amthor / Joshua C Bufton / Ufuk Borucu / Dakang Shen / Jonas P Becker / Daria Lavysh / Matthias W Hentze / Andreas E Kulozik / Gabriele Neu-Yilik / Christiane Schaffitzel /   Abstract: Deciphering translation is of paramount importance for the understanding of many diseases, and antibiotics played a pivotal role in this endeavour. Blasticidin S (BlaS) targets translation by binding ...Deciphering translation is of paramount importance for the understanding of many diseases, and antibiotics played a pivotal role in this endeavour. Blasticidin S (BlaS) targets translation by binding to the peptidyl transferase center of the large ribosomal subunit. Using biochemical, structural and cellular approaches, we show here that BlaS inhibits both translation elongation and termination in Mammalia. Bound to mammalian terminating ribosomes, BlaS distorts the 3'CCA tail of the P-site tRNA to a larger extent than previously reported for bacterial ribosomes, thus delaying both, peptide bond formation and peptidyl-tRNA hydrolysis. While BlaS does not inhibit stop codon recognition by the eukaryotic release factor 1 (eRF1), it interferes with eRF1's accommodation into the peptidyl transferase center and subsequent peptide release. In human cells, BlaS inhibits nonsense-mediated mRNA decay and, at subinhibitory concentrations, modulates translation dynamics at premature termination codons leading to enhanced protein production. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7nwh.cif.gz 7nwh.cif.gz | 7.9 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7nwh.ent.gz pdb7nwh.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7nwh.json.gz 7nwh.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/nw/7nwh https://data.pdbj.org/pub/pdb/validation_reports/nw/7nwh ftp://data.pdbj.org/pub/pdb/validation_reports/nw/7nwh ftp://data.pdbj.org/pub/pdb/validation_reports/nw/7nwh | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  12632MC  7nwgC  7nwiC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-60S RIBOSOMAL PROTEIN ... , 16 types, 16 molecules ADEgiIJLmnORSWZa
| #1: Protein | Mass: 27129.809 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #6: Protein | Mass: 34350.633 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #11: Protein | Mass: 33028.336 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #16: Protein | Mass: 14210.088 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #26: Protein | Mass: 12317.796 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #29: Protein | Mass: 24643.057 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #33: Protein | Mass: 20086.191 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #36: Protein | Mass: 24331.723 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #40: Protein | Mass: 14695.310 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #43: Protein/peptide | Mass: 3473.451 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #45: Protein | Mass: 23533.299 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #54: Protein | Mass: 23535.281 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #57: Protein | Mass: 20827.561 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #66: Protein | Mass: 15538.256 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #75: Protein | Mass: 15835.831 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #78: Protein | Mass: 16489.363 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
+Protein , 30 types, 30 molecules CdddbfFggGhHHHiijjkloOOPQrstTTTUVXAABc
+40S ribosomal protein ... , 23 types, 23 molecules DDEEeeffBBGGbbIIJJKKLLMMPPRRSSUUVVXXYYZZaaCCcc
-Ribosomal protein ... , 10 types, 10 molecules eFFjMNNNpQQWWY
| #7: Protein | Mass: 15153.217 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #13: Protein | Mass: 22913.453 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #30: Protein | Mass: 11111.032 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #39: Protein | Mass: 23870.549 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #42: Protein | Mass: 24207.285 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #44: Protein | Mass: 17188.309 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #49: Protein | Mass: 12010.391 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #53: Protein | Mass: 17716.932 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #68: Protein | Mass: 15778.584 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #72: Protein | Mass: 15891.787 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-RNA chain , 5 types, 5 molecules hh5789
| #23: RNA chain | Mass: 4753.857 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #67: RNA chain | Mass: 1199695.250 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #70: RNA chain | Mass: 38691.914 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #73: RNA chain | Mass: 48545.672 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #76: RNA chain | Mass: 573842.125 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Non-polymers , 4 types, 291 molecules 


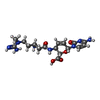



| #85: Chemical | ChemComp-ZN / #86: Chemical | ChemComp-MG / #87: Chemical | ChemComp-GCP / | #88: Chemical | ChemComp-BLS / | |
|---|
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Mammalian pre-termination 80S ribosome with eRF1 and eRF3 bound by Blasticidin S. Type: RIBOSOME Details: Rabbit reticulocyte lysate derived (in vitro transcription-FLAG affinity purified) ribosomal complex. Entity ID: #1-#84 / Source: NATURAL |
|---|---|
| Molecular weight | Value: 3.8 MDa / Experimental value: NO |
| Source (natural) | Organism:  |
| Buffer solution | pH: 7.4 Details: 50 mM HEPES pH 7.4 100 mM KOAc 5 mM Mg(Oac)2 1mM DTT |
| Specimen | Conc.: 0.63 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES / Details: 165nM (OD260nm~10.5) |
| Specimen support | Grid material: COPPER / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R2/2 |
| Vitrification | Instrument: LEICA EM GP / Cryogen name: ETHANE / Humidity: 70 % / Chamber temperature: 288.15 K Details: 30s sample incubation on grid followed by 1.1s blotting time. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TALOS ARCTICA Details: Collected in super-resolution mode (0.675A pixel size) |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: OTHER FIELD EMISSION GUN / Accelerating voltage: 200 kV / Illumination mode: OTHER |
| Electron lens | Mode: BRIGHT FIELD / Calibrated magnification: 79000 X / Calibrated defocus min: 400 nm / Calibrated defocus max: 2000 nm / Cs: 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 8 sec. / Electron dose: 41.92 e/Å2 / Detector mode: SUPER-RESOLUTION / Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Num. of grids imaged: 1 / Num. of real images: 3500 |
| Image scans | Movie frames/image: 40 |
- Processing
Processing
| EM software |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 730463 | ||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 4.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 18937 / Num. of class averages: 1 / Symmetry type: POINT | ||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 5LZT Accession code: 5LZT / Source name: PDB / Type: experimental model |
 Movie
Movie Controller
Controller














 PDBj
PDBj



































