[English] 日本語
 Yorodumi
Yorodumi- PDB-7bz2: Cryo-EM structure of the formoterol-bound beta2 adrenergic recept... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7bz2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the formoterol-bound beta2 adrenergic receptor-Gs protein complex. | ||||||
 Components Components |
| ||||||
 Keywords Keywords | SIGNALING PROTEIN / Receptor / Formoterol / Complex | ||||||
| Function / homology |  Function and homology information Function and homology informationOlfactory Signaling Pathway / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Activation of the phototransduction cascade / positive regulation of mini excitatory postsynaptic potential / beta2-adrenergic receptor activity / negative regulation of smooth muscle contraction / AMPA selective glutamate receptor signaling pathway / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / positive regulation of autophagosome maturation ...Olfactory Signaling Pathway / Sensory perception of sweet, bitter, and umami (glutamate) taste / Synthesis, secretion, and inactivation of Glucagon-like Peptide-1 (GLP-1) / Activation of the phototransduction cascade / positive regulation of mini excitatory postsynaptic potential / beta2-adrenergic receptor activity / negative regulation of smooth muscle contraction / AMPA selective glutamate receptor signaling pathway / norepinephrine-epinephrine-mediated vasodilation involved in regulation of systemic arterial blood pressure / positive regulation of autophagosome maturation / heat generation / norepinephrine binding / Adrenoceptors / positive regulation of lipophagy / Activation of G protein gated Potassium channels / G-protein activation / G beta:gamma signalling through PI3Kgamma / Prostacyclin signalling through prostacyclin receptor / G beta:gamma signalling through PLC beta / ADP signalling through P2Y purinoceptor 1 / Thromboxane signalling through TP receptor / Presynaptic function of Kainate receptors / G beta:gamma signalling through CDC42 / Inhibition of voltage gated Ca2+ channels via Gbeta/gamma subunits / G alpha (12/13) signalling events / Glucagon-type ligand receptors / G beta:gamma signalling through BTK / ADP signalling through P2Y purinoceptor 12 / Adrenaline,noradrenaline inhibits insulin secretion / negative regulation of G protein-coupled receptor signaling pathway / negative regulation of multicellular organism growth / Cooperation of PDCL (PhLP1) and TRiC/CCT in G-protein beta folding / Ca2+ pathway / Thrombin signalling through proteinase activated receptors (PARs) / G alpha (z) signalling events / Extra-nuclear estrogen signaling / G alpha (s) signalling events / G alpha (q) signalling events / response to psychosocial stress / adrenergic receptor signaling pathway / endosome to lysosome transport / G alpha (i) signalling events / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / diet induced thermogenesis / High laminar flow shear stress activates signaling by PIEZO1 and PECAM1:CDH5:KDR in endothelial cells / Vasopressin regulates renal water homeostasis via Aquaporins / positive regulation of cAMP/PKA signal transduction / adenylate cyclase binding / smooth muscle contraction / PKA activation in glucagon signalling / developmental growth / hair follicle placode formation / bone resorption / potassium channel regulator activity / positive regulation of bone mineralization / D1 dopamine receptor binding / intracellular transport / neuronal dense core vesicle / vascular endothelial cell response to laminar fluid shear stress / renal water homeostasis / activation of adenylate cyclase activity / Hedgehog 'off' state / intercellular bridge / regulation of sodium ion transport / adenylate cyclase-activating adrenergic receptor signaling pathway / brown fat cell differentiation / regulation of insulin secretion / cellular response to glucagon stimulus / receptor-mediated endocytosis / response to cold / adenylate cyclase activator activity / trans-Golgi network membrane / negative regulation of inflammatory response to antigenic stimulus / clathrin-coated endocytic vesicle membrane / bone development / platelet aggregation / cognition / adenylate cyclase-modulating G protein-coupled receptor signaling pathway / G-protein beta/gamma-subunit complex binding / cellular response to amyloid-beta / adenylate cyclase-activating G protein-coupled receptor signaling pathway / Prostacyclin signalling through prostacyclin receptor / Glucagon signaling in metabolic regulation / photoreceptor disc membrane / mitotic spindle / Glucagon-type ligand receptors / sensory perception of smell / Vasopressin regulates renal water homeostasis via Aquaporins / Glucagon-like Peptide-1 (GLP1) regulates insulin secretion / G alpha (z) signalling events / cellular response to catecholamine stimulus / ADORA2B mediated anti-inflammatory cytokines production / adenylate cyclase-activating dopamine receptor signaling pathway / Cargo recognition for clathrin-mediated endocytosis / GPER1 signaling / G-protein beta-subunit binding / cellular response to prostaglandin E stimulus / heterotrimeric G-protein complex / sensory perception of taste / amyloid-beta binding Similarity search - Function | ||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human)  | ||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.82 Å | ||||||
 Authors Authors | Zhang, Y.N. / Yang, F. / Ling, S.L. / Lv, P. / Zhou, Y.X. / Fang, W. / Sun, W. / Shi, P. / Tian, C.L. | ||||||
 Citation Citation |  Journal: Cell Discov / Year: 2020 Journal: Cell Discov / Year: 2020Title: Single-particle cryo-EM structural studies of the βAR-Gs complex bound with a full agonist formoterol. Authors: Yanan Zhang / Fan Yang / Shenglong Ling / Pei Lv / Yingxin Zhou / Wei Fang / Wenjing Sun / Longhua Zhang / Pan Shi / Changlin Tian /  | ||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7bz2.cif.gz 7bz2.cif.gz | 194.4 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7bz2.ent.gz pdb7bz2.ent.gz | 140.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7bz2.json.gz 7bz2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/bz/7bz2 https://data.pdbj.org/pub/pdb/validation_reports/bz/7bz2 ftp://data.pdbj.org/pub/pdb/validation_reports/bz/7bz2 ftp://data.pdbj.org/pub/pdb/validation_reports/bz/7bz2 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  30249MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Guanine nucleotide-binding protein ... , 3 types, 3 molecules ABG
| #1: Protein | Mass: 45812.594 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Gene: GNAS, GNAS1, GSP / Production host: Homo sapiens (human) / Gene: GNAS, GNAS1, GSP / Production host:  |
|---|---|
| #2: Protein | Mass: 37346.812 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
| #3: Protein | Mass: 8805.155 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
-Antibody / Protein / Non-polymers , 3 types, 3 molecules NR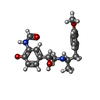

| #4: Antibody | Mass: 17483.693 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   |
|---|---|
| #5: Protein | Mass: 42010.844 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)  Homo sapiens (human) / Production host: Homo sapiens (human) / Production host:  |
| #6: Chemical | ChemComp-H98 / ~{ |
-Details
| Has ligand of interest | Y |
|---|---|
| Has protein modification | Y |
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: Beta2 adrenergic receptor-Gs trimer complex with formoterol Type: COMPLEX / Entity ID: #1-#5 / Source: RECOMBINANT |
|---|---|
| Molecular weight | Value: 151.40 kDa/nm / Experimental value: YES |
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Source (recombinant) | Organism:  |
| Buffer solution | pH: 7.5 |
| Specimen | Conc.: 0.5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES / Details: This sample was monodisperse. |
| Vitrification | Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 4 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: DARK FIELD / Nominal magnification: 29000 X |
| Image recording | Electron dose: 58.7 e/Å2 / Detector mode: COUNTING / Film or detector model: GATAN K3 (6k x 4k) / Num. of real images: 1625 |
| Image scans | Movie frames/image: 30 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.82 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 219254 / Symmetry type: POINT | ||||||||||||||||||||||||
| Refinement | Cross valid method: NONE Stereochemistry target values: GeoStd + Monomer Library + CDL v1.2 | ||||||||||||||||||||||||
| Displacement parameters | Biso mean: 34.36 Å2 | ||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller












 PDBj
PDBj
















