[English] 日本語
 Yorodumi
Yorodumi- PDB-6om6: Structure of trans-translation inhibitor bound to E. coli 70S rib... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6om6 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of trans-translation inhibitor bound to E. coli 70S ribosome with P site tRNA | |||||||||
 Components Components |
| |||||||||
 Keywords Keywords | RIBOSOME / trans-translation / translation / antibiotic | |||||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of ribosome biogenesis / DnaA-L2 complex / negative regulation of DNA-templated DNA replication initiation / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / regulation of cell growth / ribosomal large subunit assembly / mRNA 5'-UTR binding / large ribosomal subunit / ribosome binding ...positive regulation of ribosome biogenesis / DnaA-L2 complex / negative regulation of DNA-templated DNA replication initiation / assembly of large subunit precursor of preribosome / cytosolic ribosome assembly / regulation of cell growth / ribosomal large subunit assembly / mRNA 5'-UTR binding / large ribosomal subunit / ribosome binding / small ribosomal subunit / small ribosomal subunit rRNA binding / transferase activity / 5S rRNA binding / cytosolic small ribosomal subunit / cytosolic large ribosomal subunit / tRNA binding / cytoplasmic translation / rRNA binding / ribosome / structural constituent of ribosome / ribonucleoprotein complex / translation / mRNA binding / RNA binding / zinc ion binding / metal ion binding / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | ELECTRON MICROSCOPY / single particle reconstruction / cryo EM / Resolution: 3.1 Å | |||||||||
 Authors Authors | Hoffer, E.D. / Mehrani, A. / Keiler, K.C. / Stagg, S.M. / Dunham, C.M. | |||||||||
| Funding support |  United States, 2items United States, 2items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: trans-Translation inhibitors bind to a novel site on the ribosome and clear Neisseria gonorrhoeae in vivo. Authors: Zachary D Aron / Atousa Mehrani / Eric D Hoffer / Kristie L Connolly / Pooja Srinivas / Matthew C Torhan / John N Alumasa / Mynthia Cabrera / Divya Hosangadi / Jay S Barbor / Steven C ...Authors: Zachary D Aron / Atousa Mehrani / Eric D Hoffer / Kristie L Connolly / Pooja Srinivas / Matthew C Torhan / John N Alumasa / Mynthia Cabrera / Divya Hosangadi / Jay S Barbor / Steven C Cardinale / Steven M Kwasny / Lucas R Morin / Michelle M Butler / Timothy J Opperman / Terry L Bowlin / Ann Jerse / Scott M Stagg / Christine M Dunham / Kenneth C Keiler /  Abstract: Bacterial ribosome rescue pathways that remove ribosomes stalled on mRNAs during translation have been proposed as novel antibiotic targets because they are essential in bacteria and are not ...Bacterial ribosome rescue pathways that remove ribosomes stalled on mRNAs during translation have been proposed as novel antibiotic targets because they are essential in bacteria and are not conserved in humans. We previously reported the discovery of a family of acylaminooxadiazoles that selectively inhibit trans-translation, the main ribosome rescue pathway in bacteria. Here, we report optimization of the pharmacokinetic and antibiotic properties of the acylaminooxadiazoles, producing MBX-4132, which clears multiple-drug resistant Neisseria gonorrhoeae infection in mice after a single oral dose. Single particle cryogenic-EM studies of non-stop ribosomes show that acylaminooxadiazoles bind to a unique site near the peptidyl-transfer center and significantly alter the conformation of ribosomal protein bL27, suggesting a novel mechanism for specific inhibition of trans-translation by these molecules. These results show that trans-translation is a viable therapeutic target and reveal a new conformation within the bacterial ribosome that may be critical for ribosome rescue pathways. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6om6.cif.gz 6om6.cif.gz | 3.1 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6om6.ent.gz pdb6om6.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  6om6.json.gz 6om6.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Summary document |  6om6_validation.pdf.gz 6om6_validation.pdf.gz | 1.8 MB | Display |  wwPDB validaton report wwPDB validaton report |
|---|---|---|---|---|
| Full document |  6om6_full_validation.pdf.gz 6om6_full_validation.pdf.gz | 1.9 MB | Display | |
| Data in XML |  6om6_validation.xml.gz 6om6_validation.xml.gz | 220.2 KB | Display | |
| Data in CIF |  6om6_validation.cif.gz 6om6_validation.cif.gz | 376.5 KB | Display | |
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/om/6om6 https://data.pdbj.org/pub/pdb/validation_reports/om/6om6 ftp://data.pdbj.org/pub/pdb/validation_reports/om/6om6 ftp://data.pdbj.org/pub/pdb/validation_reports/om/6om6 | HTTPS FTP |
-Related structure data
| Related structure data |  20121MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-RNA chain , 5 types, 5 molecules 12345
| #1: RNA chain | Mass: 941804.625 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #2: RNA chain | Mass: 497405.969 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #3: RNA chain | Mass: 38790.090 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #4: RNA chain | Mass: 1609.048 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #5: RNA chain | Mass: 24453.486 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
+50S ribosomal protein ... , 29 types, 29 molecules BCDEFGJKLMNOPQRSTUVXYZabcdefW
-30S ribosomal protein ... , 20 types, 20 molecules ghijklmnopqrstuvwxyz
| #34: Protein | Mass: 26781.670 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
|---|---|
| #35: Protein | Mass: 26031.316 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #36: Protein | Mass: 23514.199 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #37: Protein | Mass: 17629.398 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #38: Protein | Mass: 15727.512 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #39: Protein | Mass: 20055.156 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #40: Protein | Mass: 14146.557 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #41: Protein | Mass: 14886.270 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #42: Protein | Mass: 11755.597 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #43: Protein | Mass: 13870.975 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #44: Protein | Mass: 13814.249 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #45: Protein | Mass: 13128.467 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #46: Protein | Mass: 11606.560 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #47: Protein | Mass: 10290.816 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #48: Protein | Mass: 9207.572 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #49: Protein | Mass: 9724.491 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #50: Protein | Mass: 9005.472 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #51: Protein | Mass: 10455.355 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #52: Protein | Mass: 9708.464 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
| #53: Protein | Mass: 8524.039 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)  |
-Non-polymers , 1 types, 1 molecules 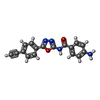
| #55: Chemical | ChemComp-KKL / |
|---|
-Experimental details
-Experiment
| Experiment | Method: ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method: single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight |
| |||||||||||||||||||||||||
| Source (natural) |
| |||||||||||||||||||||||||
| Buffer solution | pH: 7.6 | |||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied: NO / Vitrification applied: YES | |||||||||||||||||||||||||
| Specimen support | Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: UltrAuFoil | |||||||||||||||||||||||||
| Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 281.15 K / Details: Blot time: 3.5 seconds |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source:  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD / Nominal magnification: 59000 X / Nominal defocus max: 3000 nm / Nominal defocus min: 1500 nm / Cs: 2.7 mm / C2 aperture diameter: 70 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 19.302 sec. / Electron dose: 58 e/Å2 / Detector mode: COUNTING / Film or detector model: DIRECT ELECTRON DE-64 (8k x 8k) / Num. of grids imaged: 2 / Num. of real images: 2197 Details: Two separate datesets were combined. The imaging parameter was the same for both datasets. Firth data-set had 1752 micrographs. Second data-set had 445 micrographs. |
| Image scans | Sampling size: 6.5 µm / Width: 4096 / Height: 4096 / Movie frames/image: 78 / Used frames/image: 1-78 |
- Processing
Processing
| Software | Name: PHENIX / Classification: refinement | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image processing | Details: Counting mode was used. The 8k x 8k pixels were binned by two using hardware binning to increase the frame rate. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 474382 Details: 100,537 particles were selected from first dataset and 373,845 partciles from the second dataset. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry: C1 (asymmetric) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3D reconstruction | Resolution: 3.1 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 28121 / Algorithm: BACK PROJECTION / Num. of class averages: 1 / Symmetry type: POINT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | Protocol: RIGID BODY FIT / Space: REAL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 5MDV | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso max: 897.78 Å2 / Biso mean: 169.8625 Å2 / Biso min: 96.44 Å2 |
 Movie
Movie Controller
Controller






















 PDBj
PDBj































