+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-8333 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Structural basis for dynamic regulation of the human 26S proteasome | |||||||||
 マップデータ マップデータ | The doubly capped human 26S proteasome reconstruction from 237083 particles. The refinement was focused on the core particle. | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報thyrotropin-releasing hormone receptor binding / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / nuclear proteasome complex / host-mediated perturbation of viral transcription / positive regulation of inclusion body assembly / integrator complex / proteasome accessory complex / meiosis I / purine ribonucleoside triphosphate binding ...thyrotropin-releasing hormone receptor binding / Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / nuclear proteasome complex / host-mediated perturbation of viral transcription / positive regulation of inclusion body assembly / integrator complex / proteasome accessory complex / meiosis I / purine ribonucleoside triphosphate binding / proteasome regulatory particle / cytosolic proteasome complex / positive regulation of proteasomal protein catabolic process / proteasome-activating activity / Antigen processing: Ub, ATP-independent proteasomal degradation / proteasome regulatory particle, lid subcomplex / proteasome regulatory particle, base subcomplex / sperm glycocalyx / protein K63-linked deubiquitination / negative regulation of programmed cell death / metal-dependent deubiquitinase activity / Regulation of ornithine decarboxylase (ODC) / Proteasome assembly / proteasome core complex / perinuclear theca / Cross-presentation of soluble exogenous antigens (endosomes) / Somitogenesis / K63-linked deubiquitinase activity / transcription factor binding / Homologous DNA Pairing and Strand Exchange / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / Resolution of D-loop Structures through Holliday Junction Intermediates / proteasome binding / Impaired BRCA2 binding to RAD51 / myofibril / regulation of protein catabolic process / proteasome storage granule / proteasomal ubiquitin-independent protein catabolic process / sperm head-tail coupling apparatus / Presynaptic phase of homologous DNA pairing and strand exchange / general transcription initiation factor binding / protein deubiquitination / positive regulation of RNA polymerase II transcription preinitiation complex assembly / blastocyst development / polyubiquitin modification-dependent protein binding / immune system process / proteasome endopeptidase complex / NF-kappaB binding / proteasome core complex, beta-subunit complex / endopeptidase activator activity / threonine-type endopeptidase activity / proteasome assembly / mRNA export from nucleus / proteasome core complex, alpha-subunit complex / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / enzyme regulator activity / ERAD pathway / regulation of proteasomal protein catabolic process / inclusion body / ciliary tip / : / TBP-class protein binding / proteasome complex / stem cell differentiation / Regulation of activated PAK-2p34 by proteasome mediated degradation / sarcomere / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / ubiquitin binding / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Asymmetric localization of PCP proteins / Ubiquitin-dependent degradation of Cyclin D / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / TNFR2 non-canonical NF-kB pathway / AUF1 (hnRNP D0) binds and destabilizes mRNA / centriole / sperm end piece / P-body / negative regulation of inflammatory response to antigenic stimulus / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / lipopolysaccharide binding / Degradation of DVL / Dectin-1 mediated noncanonical NF-kB signaling / Degradation of CRY and PER proteins / Degradation of AXIN / Hh mutants are degraded by ERAD / Activation of NF-kappaB in B cells / G2/M Checkpoints / Hedgehog ligand biogenesis / Degradation of GLI1 by the proteasome / Defective CFTR causes cystic fibrosis / Autodegradation of the E3 ubiquitin ligase COP1 / Regulation of RUNX3 expression and activity / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.8 Å | |||||||||
 データ登録者 データ登録者 | Chen S / Wu J / Lu Y / Ma YB / Lee BH / Yu Z / Ouyang Q / Finley D / Kirschner MW / Mao Y | |||||||||
 引用 引用 |  ジャーナル: Proc Natl Acad Sci U S A / 年: 2016 ジャーナル: Proc Natl Acad Sci U S A / 年: 2016タイトル: Structural basis for dynamic regulation of the human 26S proteasome. 著者: Shuobing Chen / Jiayi Wu / Ying Lu / Yong-Bei Ma / Byung-Hoon Lee / Zhou Yu / Qi Ouyang / Daniel J Finley / Marc W Kirschner / Youdong Mao /   要旨: The proteasome is the major engine of protein degradation in all eukaryotic cells. At the heart of this machine is a heterohexameric ring of AAA (ATPases associated with diverse cellular activities) ...The proteasome is the major engine of protein degradation in all eukaryotic cells. At the heart of this machine is a heterohexameric ring of AAA (ATPases associated with diverse cellular activities) proteins that unfolds ubiquitylated target proteins that are concurrently translocated into a proteolytic chamber and degraded into peptides. Using cryoelectron microscopy, we determined a near-atomic-resolution structure of the 2.5-MDa human proteasome in its ground state, as well as subnanometer-resolution structures of the holoenzyme in three alternative conformational states. The substrate-unfolding AAA-ATPase channel is narrowed by 10 inward-facing pore loops arranged into two helices that run in parallel with each other, one hydrophobic in character and the other highly charged. The gate of the core particle was unexpectedly found closed in the ground state and open in only one of the alternative states. Coordinated, stepwise conformational changes of the regulatory particle couple ATP hydrolysis to substrate translocation and regulate gating of the core particle, leading to processive degradation. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_8333.map.gz emd_8333.map.gz | 917.8 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-8333-v30.xml emd-8333-v30.xml emd-8333.xml emd-8333.xml | 14.7 KB 14.7 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_8333.png emd_8333.png | 44 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-8333 http://ftp.pdbj.org/pub/emdb/structures/EMD-8333 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8333 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-8333 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  5t0cMC  8332C  8334C  8335C  8336C  8337C  5t0gC  5t0hC  5t0iC  5t0jC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10072 (タイトル: Structural basis for dynamic regulation of the human 26S proteasome EMPIAR-10072 (タイトル: Structural basis for dynamic regulation of the human 26S proteasomeData size: 2.0 TB Data #1: Drift-corrected micrographs of human 26S proteasome holoenzyme [micrographs - single frame] Data #2: Classified particle datasets for the human proteasome in four conformational states [picked particles - multiframe - processed]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_8333.map.gz / 形式: CCP4 / 大きさ: 1000 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_8333.map.gz / 形式: CCP4 / 大きさ: 1000 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | The doubly capped human 26S proteasome reconstruction from 237083 particles. The refinement was focused on the core particle. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.86 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
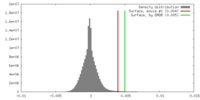
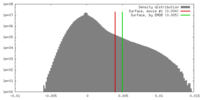
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : 26S proteasome holoenzyme
| 全体 | 名称: 26S proteasome holoenzyme |
|---|---|
| 要素 |
|
-超分子 #1: 26S proteasome holoenzyme
| 超分子 | 名称: 26S proteasome holoenzyme / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1 |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 実験値: 2.5 MDa |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 1.5 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.5 構成要素:
| ||||||||||||||||||
| グリッド | モデル: C-Flat R1/1 / 材質: COPPER / メッシュ: 400 / 支持フィルム - 材質: CARBON / 支持フィルム - トポロジー: HOLEY ARRAY / 支持フィルム - Film thickness: 50.0 nm / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 雰囲気: AIR | ||||||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 277 K / 装置: FEI VITROBOT MARK IV / 詳細: blotted for 2 seconds, blotting force 3. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TECNAI ARCTICA |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: SUPER-RESOLUTION / デジタル化 - サイズ - 横: 7420 pixel / デジタル化 - サイズ - 縦: 7676 pixel / デジタル化 - サンプリング間隔: 5.0 µm / デジタル化 - 画像ごとのフレーム数: 3-20 / 撮影したグリッド数: 12 / 実像数: 10367 / 平均露光時間: 9.0 sec. / 平均電子線量: 30.0 e/Å2 |
| 電子線 | 加速電圧: 200 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 50.0 µm / 倍率(補正後): 28736 / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): -3.0 µm / 最小 デフォーカス(公称値): -1.0 µm / 倍率(公称値): 21000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Talos Arctica / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 精密化 | 空間: RECIPROCAL / プロトコル: AB INITIO MODEL / 温度因子: 80 |
|---|---|
| 得られたモデル |  PDB-5t0c: |
 ムービー
ムービー コントローラー
コントローラー


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)