+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | native NMDA receptor-GluN1/N2A-S3 in the closed state | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | native / NMDA receptor / inotropic ion channel / excitatory neurotransmitter / MEMBRANE PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationAssembly and cell surface presentation of NMDA receptors / EPHB-mediated forward signaling / Synaptic adhesion-like molecules / Unblocking of NMDA receptors, glutamate binding and activation / neurotransmitter receptor transport, plasma membrane to endosome / RAF/MAP kinase cascade / receptor recycling / directional locomotion / pons maturation / positive regulation of Schwann cell migration ...Assembly and cell surface presentation of NMDA receptors / EPHB-mediated forward signaling / Synaptic adhesion-like molecules / Unblocking of NMDA receptors, glutamate binding and activation / neurotransmitter receptor transport, plasma membrane to endosome / RAF/MAP kinase cascade / receptor recycling / directional locomotion / pons maturation / positive regulation of Schwann cell migration / regulation of cell communication / olfactory learning / serotonin metabolic process / dendritic branch / conditioned taste aversion / protein localization to postsynaptic membrane / regulation of ARF protein signal transduction / transmitter-gated monoatomic ion channel activity / suckling behavior / sleep / regulation of respiratory gaseous exchange / propylene metabolic process / response to glycine / dendritic spine organization / locomotion / positive regulation of inhibitory postsynaptic potential / neurotransmitter receptor complex / regulation of monoatomic cation transmembrane transport / NMDA glutamate receptor activity / NMDA selective glutamate receptor complex / glutamate binding / voltage-gated monoatomic cation channel activity / glutamate receptor signaling pathway / ligand-gated sodium channel activity / transport vesicle membrane / neuromuscular process / regulation of axonogenesis / calcium ion transmembrane import into cytosol / regulation of dendrite morphogenesis / male mating behavior / regulation of synapse assembly / protein heterotetramerization / response to morphine / glycine binding / startle response / dopamine metabolic process / cellular response to zinc ion / positive regulation of reactive oxygen species biosynthetic process / response to lithium ion / parallel fiber to Purkinje cell synapse / monoatomic ion channel complex / regulation of postsynaptic membrane potential / monoatomic cation transmembrane transport / positive regulation of calcium ion transport into cytosol / modulation of excitatory postsynaptic potential / associative learning / positive regulation of dendritic spine maintenance / action potential / positive regulation of protein targeting to membrane / regulation of neuronal synaptic plasticity / monoatomic cation transport / glutamate receptor binding / social behavior / ligand-gated monoatomic ion channel activity / phosphatase binding / long-term memory / prepulse inhibition / postsynaptic density, intracellular component / monoatomic cation channel activity / synaptic cleft / calcium ion homeostasis / glutamate-gated receptor activity / positive regulation of synaptic transmission, glutamatergic / glutamate-gated calcium ion channel activity / presynaptic active zone membrane / cell adhesion molecule binding / neurogenesis / excitatory synapse / ionotropic glutamate receptor signaling pathway / sensory perception of pain / ionotropic glutamate receptor binding / dendrite membrane / cytoplasmic vesicle membrane / regulation of neuron apoptotic process / ligand-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / protein tyrosine kinase binding / positive regulation of excitatory postsynaptic potential / hippocampal mossy fiber to CA3 synapse / learning / sodium ion transmembrane transport / synaptic membrane / response to amphetamine / adult locomotory behavior / cellular response to amino acid stimulus / transmitter-gated monoatomic ion channel activity involved in regulation of postsynaptic membrane potential / synaptic transmission, glutamatergic / regulation of membrane potential / excitatory postsynaptic potential / locomotory behavior / negative regulation of protein catabolic process Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.29 Å | |||||||||
 Authors Authors | Yu J / Xu RS / Ge JP | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2026 Journal: Nature / Year: 2026Title: Conformational diversity and fully opening mechanism of native NMDA receptor. Authors: Ruisheng Xu / Qiqi Jiang / Hongwei Xu / Lu Zhang / Xiangzi Hu / Zizhuo Lu / Huaqin Deng / Haolin Xiong / Sensen Zhang / Zhongwen Chen / Yifan Ge / Zhengjiang Zhu / Yaoyang Zhang / Yelin Chen ...Authors: Ruisheng Xu / Qiqi Jiang / Hongwei Xu / Lu Zhang / Xiangzi Hu / Zizhuo Lu / Huaqin Deng / Haolin Xiong / Sensen Zhang / Zhongwen Chen / Yifan Ge / Zhengjiang Zhu / Yaoyang Zhang / Yelin Chen / Jingpeng Ge / Jie Yu /  Abstract: N-methyl-D-aspartate receptors (NMDARs) are glutamate-gated ion channels that mediate excitatory neurotransmission throughout the brain. As obligate heterotetramers, their activation requires the ...N-methyl-D-aspartate receptors (NMDARs) are glutamate-gated ion channels that mediate excitatory neurotransmission throughout the brain. As obligate heterotetramers, their activation requires the binding of both glycine and glutamate. Although recent structural studies have provided insights into endogenous receptors from select brain regions, most previous work has relied on recombinant receptors and engineered constructs, which limits our understanding of native NMDARs across the whole brain. Here we identify and resolve ten distinct native NMDAR assemblies from the whole-brain tissue of female C57BL/6 mice using immunoaffinity purification, single-molecule total internal reflection fluorescence microscopy and cryo-electron microscopy. Analyses of the GluN1-GluN2A(S1), GluN1-GluN2A(S2), GluN1-GluN2A(S3), GluN1-GluN2B, GluN1-GluN2A-GluN2B(S1), GluN1-GluN2A-GluN2B(S2), GluN1-GluN2A-GluNX(S1), GluN1-GluN2A-GluNX(S2), GluN1-GluN2B-GluNX and GluN1-GluNX structures reveal that GluN2A is the most prevalent subunit across assemblies. Moreover, the substantial conformational flexibility observed in the GluN2A amino-terminal domain may explain its fast kinetics and dominant role in gating. Dynamic movements of S-ketamine were also captured at the channel vestibule, as was pore dilation in both the GluN1 and GluN2B subunits of a native GluN1-GluN2B receptor. The latter observation represents a previously unknown fully open state of NMDAR. Our large collection of heterogeneous NMDAR structures from whole brain reveals previously unrecognized properties of conformational diversity and channel dilation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_64357.map.gz emd_64357.map.gz | 228.5 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-64357-v30.xml emd-64357-v30.xml emd-64357.xml emd-64357.xml | 19.9 KB 19.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_64357.png emd_64357.png | 37.6 KB | ||
| Filedesc metadata |  emd-64357.cif.gz emd-64357.cif.gz | 6.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-64357 http://ftp.pdbj.org/pub/emdb/structures/EMD-64357 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-64357 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-64357 | HTTPS FTP |
-Related structure data
| Related structure data |  9unnMC  9un2C  9un3C  9unjC  9unkC  9unmC  9unoC  9unpC  9unqC  9unrC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_64357.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_64357.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.055 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : native NMDA receptor-GluN1/N2A-S3 in the closed state
| Entire | Name: native NMDA receptor-GluN1/N2A-S3 in the closed state |
|---|---|
| Components |
|
-Supramolecule #1: native NMDA receptor-GluN1/N2A-S3 in the closed state
| Supramolecule | Name: native NMDA receptor-GluN1/N2A-S3 in the closed state / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  |
-Macromolecule #1: Glutamate receptor ionotropic, NMDA 1
| Macromolecule | Name: Glutamate receptor ionotropic, NMDA 1 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 91.838938 KDa |
| Sequence | String: KIVNIGAVLS TRKHEQMFRE AVNQANKRHG SWKIQLNATS VTHKPNAIQM ALSVCEDLIS SQVYAILVSH PPTPNDHFTP TPVSYTAGF YRIPVLGLTT RMSIYSDKSI HLSFLRTVPP YSHQSSVWFE MMRVYNWNHI ILLVSDDHEG RAAQKRLETL L EERESKAE ...String: KIVNIGAVLS TRKHEQMFRE AVNQANKRHG SWKIQLNATS VTHKPNAIQM ALSVCEDLIS SQVYAILVSH PPTPNDHFTP TPVSYTAGF YRIPVLGLTT RMSIYSDKSI HLSFLRTVPP YSHQSSVWFE MMRVYNWNHI ILLVSDDHEG RAAQKRLETL L EERESKAE KVLQFDPGTK NVTALLMEAR DLEARVIILS ASEDDAATVY RAAAMLNMTG SGYVWLVGER EISGNALRYA PD GIIGLQL INGKNESAHI SDAVGVVAQA VHELLEKENI TDPPRGCVGN TNIWKTGPLF KRVLMSSKYA DGVTGRVEFN EDG DRKFAN YSIMNLQNRK LVQVGIYNGT HVIPNDRKII WPGGETEKPR GYQMSTRLKI VTIHQEPFVY VKPTMSDGTC KEEF TVNGD PVKKVICTGP NDTSPGSPRH TVPQCCYGFC VDLLIKLART MNFTYEVHLV ADGKFGTQER VNNSNKKEWN GMMGE LLSG QADMIVAPLT INNERAQYIE FSKPFKYQGL TILVKKEIPR STLDSFMQPF QSTLWLLVGL SVHVVAVMLY LLDRFS PFG RFKVNSEEEE EDALTLSSAM WFSWGVLLNS GIGEGAPRSF SARILGMVWA GFAMIIVASY TANLAAFLVL DRPEERI TG INDPRLRNPS DKFIYATVKQ SSVDIYFRRQ VELSTMYRHM EKHNYESAAE AIQAVRDNKL HAFIWDSAVL EFEASQKC D LVTTGELFFR SGFGIGMRKD SPWKQNVSLS ILKSHENGFM EDLDKTWVRY QECDSRSNAP ATLTFENMAG VFMLVAGGI VAGIFLIFIE IAYKRH UniProtKB: Glutamate receptor ionotropic, NMDA 1 |
-Macromolecule #2: Glutamate receptor ionotropic, NMDA 2A
| Macromolecule | Name: Glutamate receptor ionotropic, NMDA 2A / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 91.226312 KDa |
| Sequence | String: PALNIAVLLG HSHDVTEREL RNLWGPEQAT GLPLDVNVVA LLMNRTDPKS LITHVCDLMS GARIHGLVFG DDTDQEAVAQ MLDFISSQT FIPILGIHGG ASMIMADKDP TSTFFQFGAS IQQQATVMLK IMQDYDWHVF SLVTTIFPGY RDFISFIKTT V DNSFVGWD ...String: PALNIAVLLG HSHDVTEREL RNLWGPEQAT GLPLDVNVVA LLMNRTDPKS LITHVCDLMS GARIHGLVFG DDTDQEAVAQ MLDFISSQT FIPILGIHGG ASMIMADKDP TSTFFQFGAS IQQQATVMLK IMQDYDWHVF SLVTTIFPGY RDFISFIKTT V DNSFVGWD MQNVITLDTS FEDAKTQVQL KKIHSSVILL YCSKDEAVLI LSEARSLGLT GYDFFWIVPS LVSGNTELIP KE FPSGLIS VSYDDWDYSL EARVRDGLGI LTTAASSMLE KFSYIPEAKA SCYGQTEKPE TPLHTLHQFM VNVTWDGKDL SFT EEGYQV HPRLVVIVLN KDREWEKVGK WENQTLSLRH AVWPRYKSFS DCEPDDNHLS IVTLEEAPFV IVEDIDPLTE TCVR NTVPC RKFVKINNST NEGMNVKKCC KGFCIDILKK LSRTVKFTYD LYLVTNGKHG KKVNNVWNGM IGEVVYQRAV MAVGS LTIN EERSEVVDFS VPFVETGISV MVSRSNGTVS PSAFLEPFSA SVWVMMFVML LIVSAIAVFV FEYFSPVGYN RNLAKG KAP HGPSFTIGKA IWLLWGLVFN NSVPVQNPKG TTSKIMVSVW AFFAVIFLAS YTANLAAFMI QEEFVDQVTG LSDKKFQ RP HDYSPPFRFG TVPNGSTERN IRNNYPYMHQ YMTKFNQRGV EDALVSLKTG KLDAFIYDAA VLNYKAGRDE GCKLVTIG S GYIFATTGYG IALQKGSPWK RQIDLALLQF VGDGEMEELE TLWLTGICHN EKNEVMSSQL DIDNMAGVFY MLAAAMALS LITFIWEHLF YW UniProtKB: Glutamate receptor ionotropic, NMDA 2A |
-Macromolecule #3: 2-acetamido-2-deoxy-beta-D-glucopyranose
| Macromolecule | Name: 2-acetamido-2-deoxy-beta-D-glucopyranose / type: ligand / ID: 3 / Number of copies: 25 / Formula: NAG |
|---|---|
| Molecular weight | Theoretical: 221.208 Da |
| Chemical component information |  ChemComp-NAG: |
-Macromolecule #4: GLYCINE
| Macromolecule | Name: GLYCINE / type: ligand / ID: 4 / Number of copies: 2 / Formula: GLY |
|---|---|
| Molecular weight | Theoretical: 75.067 Da |
| Chemical component information |  ChemComp-GLY: |
-Macromolecule #5: GLUTAMIC ACID
| Macromolecule | Name: GLUTAMIC ACID / type: ligand / ID: 5 / Number of copies: 2 / Formula: GLU |
|---|---|
| Molecular weight | Theoretical: 147.129 Da |
| Chemical component information |  ChemComp-GLU: |
-Macromolecule #6: (2~{S})-2-(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one
| Macromolecule | Name: (2~{S})-2-(2-chlorophenyl)-2-(methylamino)cyclohexan-1-one type: ligand / ID: 6 / Number of copies: 1 / Formula: JC9 |
|---|---|
| Molecular weight | Theoretical: 237.725 Da |
| Chemical component information | 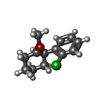 ChemComp-JC9: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




























































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















