[English] 日本語
 Yorodumi
Yorodumi- EMDB-47394: Structure of RyR1 in the primed state in the presence of oxopyricid -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of RyR1 in the primed state in the presence of oxopyricid | |||||||||
 Map data Map data | Structure of RyR1 in the primed state in the presence of oxopyricid | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | calcium channel / TRANSPORT PROTEIN / sarcoplasmic reticulum | |||||||||
| Function / homology |  Function and homology information Function and homology informationcytoplasmic side of membrane / ATP-gated ion channel activity / ryanodine-sensitive calcium-release channel activity / terminal cisterna / ryanodine receptor complex / release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / ossification involved in bone maturation / cellular response to caffeine / skin development / organelle membrane ...cytoplasmic side of membrane / ATP-gated ion channel activity / ryanodine-sensitive calcium-release channel activity / terminal cisterna / ryanodine receptor complex / release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / ossification involved in bone maturation / cellular response to caffeine / skin development / organelle membrane / regulation of ryanodine-sensitive calcium-release channel activity / intracellularly gated calcium channel activity / smooth endoplasmic reticulum / outflow tract morphogenesis / intracellular membrane-bounded organelle / toxic substance binding / voltage-gated calcium channel activity / striated muscle contraction / skeletal muscle fiber development / regulation of release of sequestered calcium ion into cytosol by sarcoplasmic reticulum / release of sequestered calcium ion into cytosol / sarcoplasmic reticulum membrane / cellular response to calcium ion / muscle contraction / sarcoplasmic reticulum / peptidylprolyl isomerase / peptidyl-prolyl cis-trans isomerase activity / sarcolemma / calcium ion transmembrane transport / calcium channel activity / Z disc / intracellular calcium ion homeostasis / disordered domain specific binding / protein homotetramerization / transmembrane transporter binding / calmodulin binding / calcium ion binding / ATP binding / membrane / identical protein binding / cytosol Similarity search - Function | |||||||||
| Biological species |  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.26 Å | |||||||||
 Authors Authors | Miotto MC / Marks AR | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Proc Natl Acad Sci U S A / Year: 2025 Journal: Proc Natl Acad Sci U S A / Year: 2025Title: Targeting ryanodine receptors with allopurinol and xanthine derivatives for the treatment of cardiac and musculoskeletal weakness disorders. Authors: Marco C Miotto / Estefania Luna-Figueroa / Carl Tchagou / Laith Bahlouli / Steven Reiken / Haikel Dridi / Yang Liu / Gunnar Weninger / Andrew R Marks /  Abstract: Ryanodine receptors (RyRs) are intracellular Ca channels essential for muscle contraction. Caffeine, a xanthine derivative, has been known for decades to increase muscle contraction and enhance ...Ryanodine receptors (RyRs) are intracellular Ca channels essential for muscle contraction. Caffeine, a xanthine derivative, has been known for decades to increase muscle contraction and enhance activation of RyRs by increasing the sensitivity to Ca. We previously showed that xanthine, the only physiologically relevant xanthine derivative, also binds to and activates RyR2. Most xanthine derivatives and analogs are safe and widely prescribed, with the most popular being the xanthine oxidoreductase inhibitor allopurinol (~15M yearly prescriptions in USA). We propose that xanthine derivatives and analogs that enhance RyRs activity could be used for lead optimization and eventually for the treatment of the diseases that exhibit decreased muscle contraction and reduced RyRs activity, such as RyR1-related diseases, sarcopenia, and heart failure. Here, we show by cryo-EM that xanthine derivatives, analogs, and other related compounds bind to the xanthine/caffeine binding site and activate RyR1, and identify 4-oxopyrimidine as the minimal motif necessary for such interaction. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_47394.map.gz emd_47394.map.gz | 254.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-47394-v30.xml emd-47394-v30.xml emd-47394.xml emd-47394.xml | 30.7 KB 30.7 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_47394.png emd_47394.png | 133.6 KB | ||
| Filedesc metadata |  emd-47394.cif.gz emd-47394.cif.gz | 9.9 KB | ||
| Others |  emd_47394_additional_1.map.gz emd_47394_additional_1.map.gz emd_47394_half_map_1.map.gz emd_47394_half_map_1.map.gz emd_47394_half_map_2.map.gz emd_47394_half_map_2.map.gz | 257.1 MB 475 MB 475 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-47394 http://ftp.pdbj.org/pub/emdb/structures/EMD-47394 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-47394 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-47394 | HTTPS FTP |
-Related structure data
| Related structure data |  9e1hMC  9e17C  9e18C  9e19C  9e1aC  9e1bC  9e1cC  9e1dC  9e1eC  9e1fC  9e1gC  9e1iC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_47394.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_47394.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of RyR1 in the primed state in the presence of oxopyricid | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.827 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Additional map: Structure of RyR1 in the primed state in...
| File | emd_47394_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Structure of RyR1 in the primed state in the presence of oxopyricid - focused map | ||||||||||||
| Projections & Slices |
| ||||||||||||
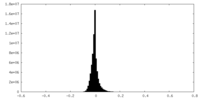
| Density Histograms |
-Half map: #1
| File | emd_47394_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
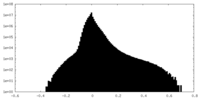
| Density Histograms |
-Half map: #2
| File | emd_47394_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Complex of RyR1 and Calstabin-1
+Supramolecule #1: Complex of RyR1 and Calstabin-1
+Supramolecule #2: Ryanodine Receptor 1
+Supramolecule #3: Calstabin-1
+Macromolecule #1: Ryanodine receptor 1
+Macromolecule #2: Peptidyl-prolyl cis-trans isomerase FKBP1A
+Macromolecule #3: ADENOSINE-5'-TRIPHOSPHATE
+Macromolecule #4: CALCIUM ION
+Macromolecule #5: ZINC ION
+Macromolecule #6: oxopyricid
+Macromolecule #7: water
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 10 mg/mL | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
Details: 2 mM oxopyricid was added to the final sample from a 20 mM stock solution in buffer. | |||||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Temperature | Min: 80.0 K / Max: 100.0 K |
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Average electron dose: 58.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)