+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-4496 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Ovine respiratory supercomplex I+III2 open class 3 | |||||||||
 Map data Map data | Ovine mitochondrial respiratory supercomplex I III2 open class 3. | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | supercomplex / cellular respiration / mitochondria / ELECTRON TRANSPORT | |||||||||
| Function / homology |  Function and homology information Function and homology information: / : / : / : / respiratory chain complex III / quinol-cytochrome-c reductase / quinol-cytochrome-c reductase activity / mitochondrial electron transport, ubiquinol to cytochrome c / ubiquinone binding / NADH:ubiquinone reductase (H+-translocating) ...: / : / : / : / respiratory chain complex III / quinol-cytochrome-c reductase / quinol-cytochrome-c reductase activity / mitochondrial electron transport, ubiquinol to cytochrome c / ubiquinone binding / NADH:ubiquinone reductase (H+-translocating) / apoptotic mitochondrial changes / electron transport coupled proton transport / mitochondrial electron transport, NADH to ubiquinone / acyl binding / mitochondrial respiratory chain complex I assembly / oxidoreductase activity, acting on NAD(P)H / NADH dehydrogenase activity / membrane => GO:0016020 / respiratory chain complex I / NADH dehydrogenase (ubiquinone) activity / acyl carrier activity / ATP metabolic process / ATP synthesis coupled electron transport / reactive oxygen species metabolic process / regulation of mitochondrial membrane potential / respiratory electron transport chain / electron transport chain / circadian rhythm / metalloendopeptidase activity / mitochondrial intermembrane space / 2 iron, 2 sulfur cluster binding / mitochondrial membrane / NAD binding / FMN binding / 4 iron, 4 sulfur cluster binding / response to oxidative stress / electron transfer activity / oxidoreductase activity / mitochondrial inner membrane / mitochondrial matrix / heme binding / protein-containing complex binding / mitochondrion / proteolysis / nucleoplasm / membrane / metal ion binding Similarity search - Function | |||||||||
| Biological species |  | |||||||||
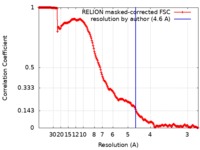
| Method | single particle reconstruction / cryo EM / Resolution: 4.6 Å | |||||||||
 Authors Authors | Letts JA / Sazanov LA | |||||||||
| Funding support |  Austria, 1 items Austria, 1 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2019 Journal: Mol Cell / Year: 2019Title: Structures of Respiratory Supercomplex I+III Reveal Functional and Conformational Crosstalk. Authors: James A Letts / Karol Fiedorczuk / Gianluca Degliesposti / Mark Skehel / Leonid A Sazanov /    Abstract: The mitochondrial electron transport chain complexes are organized into supercomplexes (SCs) of defined stoichiometry, which have been proposed to regulate electron flux via substrate channeling. We ...The mitochondrial electron transport chain complexes are organized into supercomplexes (SCs) of defined stoichiometry, which have been proposed to regulate electron flux via substrate channeling. We demonstrate that CoQ trapping in the isolated SC I+III limits complex (C)I turnover, arguing against channeling. The SC structure, resolved at up to 3.8 Å in four distinct states, suggests that CoQ oxidation may be rate limiting because of unequal access of CoQ to the active sites of CIII. CI shows a transition between "closed" and "open" conformations, accompanied by the striking rotation of a key transmembrane helix. Furthermore, the state of CI affects the conformational flexibility within CIII, demonstrating crosstalk between the enzymes. CoQ was identified at only three of the four binding sites in CIII, suggesting that interaction with CI disrupts CIII symmetry in a functionally relevant manner. Together, these observations indicate a more nuanced functional role for the SCs. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_4496.map.gz emd_4496.map.gz | 480.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-4496-v30.xml emd-4496-v30.xml emd-4496.xml emd-4496.xml | 91.4 KB 91.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_4496_fsc.xml emd_4496_fsc.xml | 18 KB | Display |  FSC data file FSC data file |
| Images |  emd_4496.png emd_4496.png | 83 KB | ||
| Masks |  emd_4496_msk_1.map emd_4496_msk_1.map | 512 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-4496.cif.gz emd-4496.cif.gz | 17.1 KB | ||
| Others |  emd_4496_half_map_1.map.gz emd_4496_half_map_1.map.gz emd_4496_half_map_2.map.gz emd_4496_half_map_2.map.gz | 410.9 MB 410.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-4496 http://ftp.pdbj.org/pub/emdb/structures/EMD-4496 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4496 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-4496 | HTTPS FTP |
-Related structure data
| Related structure data |  6qc4MC  4479C  4480C  4481C  4482C  4493C  4494C  4495C  4497C  4498C  4499C  4500C  4501C  4502C  4505C  4506C  4507C  6q9bC  6q9dC  6q9eC  6qa9C  6qbxC  6qc2C  6qc3C  6qc5C  6qc6C  6qc7C  6qc8C  6qc9C  6qcaC  6qcfC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_4496.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_4496.map.gz / Format: CCP4 / Size: 512 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Ovine mitochondrial respiratory supercomplex I III2 open class 3. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.4 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
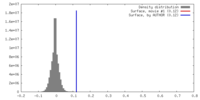
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_4496_msk_1.map emd_4496_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
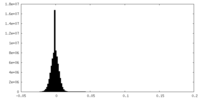
| Density Histograms |
-Half map: Ovine mitochondrial respiratory supercomplex I III2 open class...
| File | emd_4496_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Ovine mitochondrial respiratory supercomplex I III2 open class 3. Half map 1. | ||||||||||||
| Projections & Slices |
| ||||||||||||
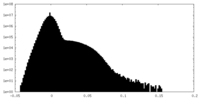
| Density Histograms |
-Half map: Ovine mitochondrial respiratory supercomplex I III2 open class...
| File | emd_4496_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Ovine mitochondrial respiratory supercomplex I III2 open class 3. Half map 2. | ||||||||||||
| Projections & Slices |
| ||||||||||||
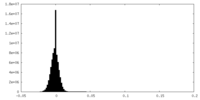
| Density Histograms |
- Sample components
Sample components
+Entire : Ovine mitochondrial respiratory supercomplex I+III2
+Supramolecule #1: Ovine mitochondrial respiratory supercomplex I+III2
+Macromolecule #1: UQCRC1
+Macromolecule #2: Ubiquinol-cytochrome c reductase core protein 2
+Macromolecule #3: Cytochrome b
+Macromolecule #4: Cytochrome c1
+Macromolecule #5: Cytochrome b-c1 complex subunit Rieske, mitochondrial
+Macromolecule #6: UQCRB
+Macromolecule #7: Ubiquinol-cytochrome c reductase complex III subunit VII
+Macromolecule #8: Cytochrome b-c1 complex subunit 6
+Macromolecule #9: UQCRFS1N
+Macromolecule #10: Ubiquinol-cytochrome c reductase, complex III subunit X
+Macromolecule #11: NADH-ubiquinone oxidoreductase chain 3
+Macromolecule #12: NADH-ubiquinone oxidoreductase chain 1
+Macromolecule #13: NADH-ubiquinone oxidoreductase chain 6
+Macromolecule #14: NADH-ubiquinone oxidoreductase chain 4L
+Macromolecule #15: NADH-ubiquinone oxidoreductase chain 5
+Macromolecule #16: NADH-ubiquinone oxidoreductase chain 4
+Macromolecule #17: NADH-ubiquinone oxidoreductase chain 2
+Macromolecule #18: NDUFA11
+Macromolecule #19: NADH:ubiquinone oxidoreductase subunit B5
+Macromolecule #20: Acyl carrier protein
+Macromolecule #21: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 8
+Macromolecule #22: NDUFB10
+Macromolecule #23: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 10, mi...
+Macromolecule #24: NADH:ubiquinone oxidoreductase subunit S5
+Macromolecule #25: NADH:ubiquinone oxidoreductase subunit A3
+Macromolecule #26: NADH:ubiquinone oxidoreductase subunit B3
+Macromolecule #27: NADH dehydrogenase [ubiquinone] 1 subunit C2
+Macromolecule #28: NADH:ubiquinone oxidoreductase subunit B4
+Macromolecule #29: NDUFA13
+Macromolecule #30: NDUFB6
+Macromolecule #31: NADH:ubiquinone oxidoreductase subunit B7
+Macromolecule #32: NADH:ubiquinone oxidoreductase subunit B9
+Macromolecule #33: NADH:ubiquinone oxidoreductase subunit B2
+Macromolecule #34: NADH dehydrogenase [ubiquinone] 1 beta subcomplex subunit 8, mito...
+Macromolecule #35: NDUFB11
+Macromolecule #36: NDUFC1
+Macromolecule #37: NDUFB1
+Macromolecule #38: NDUFA1
+Macromolecule #39: NADH dehydrogenase [ubiquinone] flavoprotein 1, mitochondrial
+Macromolecule #40: NDUFV2
+Macromolecule #41: NADH:ubiquinone oxidoreductase core subunit S1
+Macromolecule #42: NDUFS2
+Macromolecule #43: NADH:ubiquinone oxidoreductase core subunit S3
+Macromolecule #44: NDUFS7
+Macromolecule #45: NDUFS8
+Macromolecule #46: NDUFV3
+Macromolecule #47: NDUFS6
+Macromolecule #48: NADH:ubiquinone oxidoreductase subunit S4
+Macromolecule #49: NADH:ubiquinone oxidoreductase subunit A9
+Macromolecule #50: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 2
+Macromolecule #51: NDUFA5
+Macromolecule #52: NADH:ubiquinone oxidoreductase subunit A6
+Macromolecule #53: NDUFA7
+Macromolecule #54: NADH dehydrogenase [ubiquinone] 1 alpha subcomplex subunit 12
+Macromolecule #55: PROTOPORPHYRIN IX CONTAINING FE
+Macromolecule #56: HEME C
+Macromolecule #57: FE2/S2 (INORGANIC) CLUSTER
+Macromolecule #58: 1,2-DIACYL-SN-GLYCERO-3-PHOSPHOCHOLINE
+Macromolecule #59: S-[2-({N-[(2S)-2-hydroxy-3,3-dimethyl-4-(phosphonooxy)butanoyl]-b...
+Macromolecule #60: IRON/SULFUR CLUSTER
+Macromolecule #61: FLAVIN MONONUCLEOTIDE
+Macromolecule #62: ZINC ION
+Macromolecule #63: NADPH DIHYDRO-NICOTINAMIDE-ADENINE-DINUCLEOTIDE PHOSPHATE
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
Details: 250 mM NaCl, 20 mM HEPES, pH 7.7, 0.02% Brij-35 | ||||||||||||
| Vitrification | Cryogen name: PROPANE / Chamber humidity: 95 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: blotting for 30 seconds at 4 degrees Celsius, 95% humidity and flash freezing. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: FEI FALCON II (4k x 4k) / Number grids imaged: 1 / Number real images: 1854 / Average exposure time: 2.0 sec. / Average electron dose: 51.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated defocus max: 3.0 µm / Calibrated defocus min: 1.5 µm / Calibrated magnification: 100000 / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller
























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)