+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Quis-bound intermediate mGlu5 | |||||||||
 Map data Map data | Quis bound intermediate mGlu5 | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | GPCR / SIGNALING PROTEIN | |||||||||
| Function / homology |  Function and homology information Function and homology informationA2A adenosine receptor binding / neurotransmitter receptor activity involved in regulation of postsynaptic cytosolic calcium ion concentration / G protein-coupled receptor activity involved in regulation of postsynaptic membrane potential / adenylate cyclase inhibiting G protein-coupled glutamate receptor activity / phospholipase C-activating G protein-coupled glutamate receptor signaling pathway / positive regulation of long-term neuronal synaptic plasticity / desensitization of G protein-coupled receptor signaling pathway / G protein-coupled glutamate receptor signaling pathway / astrocyte projection / Class C/3 (Metabotropic glutamate/pheromone receptors) ...A2A adenosine receptor binding / neurotransmitter receptor activity involved in regulation of postsynaptic cytosolic calcium ion concentration / G protein-coupled receptor activity involved in regulation of postsynaptic membrane potential / adenylate cyclase inhibiting G protein-coupled glutamate receptor activity / phospholipase C-activating G protein-coupled glutamate receptor signaling pathway / positive regulation of long-term neuronal synaptic plasticity / desensitization of G protein-coupled receptor signaling pathway / G protein-coupled glutamate receptor signaling pathway / astrocyte projection / Class C/3 (Metabotropic glutamate/pheromone receptors) / glutamate receptor activity / Neurexins and neuroligins / protein tyrosine kinase activator activity / regulation of synaptic transmission, glutamatergic / positive regulation of calcium-mediated signaling / protein tyrosine kinase binding / dendritic shaft / learning / locomotory behavior / G protein-coupled receptor activity / synapse organization / postsynaptic density membrane / Schaffer collateral - CA1 synapse / cognition / cellular response to amyloid-beta / chemical synaptic transmission / G alpha (q) signalling events / dendritic spine / learning or memory / positive regulation of MAPK cascade / neuronal cell body / dendrite / regulation of DNA-templated transcription / glutamatergic synapse / identical protein binding / plasma membrane / cytoplasm Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / Homo sapiens (human) /  | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | |||||||||
 Authors Authors | Krishna Kumar K / Wang H / Kobilka BK | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Step-wise activation of a Family C GPCR. Authors: Kaavya Krishna Kumar / Haoqing Wang / Chris Habrian / Naomi R Latorraca / Jun Xu / Evan S O'Brien / Chensong Zhang / Elizabeth Montabana / Antoine Koehl / Susan Marqusee / Ehud Y Isacoff / Brian K Kobilka /  Abstract: Metabotropic glutamate receptors belong to a family of G protein-coupled receptors that are obligate dimers and possess a large extracellular ligand-binding domain (ECD) that is linked via a cysteine- ...Metabotropic glutamate receptors belong to a family of G protein-coupled receptors that are obligate dimers and possess a large extracellular ligand-binding domain (ECD) that is linked via a cysteine-rich domain (CRDs) to their 7-transmembrane (TM) domain. Upon activation, these receptors undergo a large conformational change to transmit the ligand binding signal from the ECD to the G protein-coupling TM. In this manuscript, we propose a model for a sequential, multistep activation mechanism of metabotropic glutamate receptor subtype 5. We present a series of structures in lipid nanodiscs, from inactive to fully active, including agonist-bound intermediate states. Further, using bulk and single-molecule fluorescence imaging we reveal distinct receptor conformations upon allosteric modulator and G protein binding. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_41092.map.gz emd_41092.map.gz | 141 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-41092-v30.xml emd-41092-v30.xml emd-41092.xml emd-41092.xml | 17.1 KB 17.1 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_41092.png emd_41092.png | 54.1 KB | ||
| Filedesc metadata |  emd-41092.cif.gz emd-41092.cif.gz | 6.8 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41092 http://ftp.pdbj.org/pub/emdb/structures/EMD-41092 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41092 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41092 | HTTPS FTP |
-Validation report
| Summary document |  emd_41092_validation.pdf.gz emd_41092_validation.pdf.gz | 458.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_41092_full_validation.pdf.gz emd_41092_full_validation.pdf.gz | 457.9 KB | Display | |
| Data in XML |  emd_41092_validation.xml.gz emd_41092_validation.xml.gz | 6.6 KB | Display | |
| Data in CIF |  emd_41092_validation.cif.gz emd_41092_validation.cif.gz | 7.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41092 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41092 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41092 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41092 | HTTPS FTP |
-Related structure data
| Related structure data |  8t7hMC  8t6jC  8t8mC  8taoC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_41092.map.gz / Format: CCP4 / Size: 149.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_41092.map.gz / Format: CCP4 / Size: 149.9 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Quis bound intermediate mGlu5 | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.111 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
-Entire : Metabotropic glutamate receptor 5 in complex with Quis
| Entire | Name: Metabotropic glutamate receptor 5 in complex with Quis |
|---|---|
| Components |
|
-Supramolecule #1: Metabotropic glutamate receptor 5 in complex with Quis
| Supramolecule | Name: Metabotropic glutamate receptor 5 in complex with Quis type: organelle_or_cellular_component / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Metabotropic glutamate receptor 5
| Macromolecule | Name: Metabotropic glutamate receptor 5 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 98.868297 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDAAQSSER RVVAHMPGDI IIGALFSVHH QPTVDKVHER KCGAVREQYG IQRVEAMLHT LERINSDPT LLPNITLGCE IRDSCWHSAV ALEQSIEFIR DSLISSEEEE GLVRCVDGSS SSFRSKKPIV GVIGPGSSSV A IQVQNLLQ ...String: MKTIIALSYI FCLVFADYKD DDDAAQSSER RVVAHMPGDI IIGALFSVHH QPTVDKVHER KCGAVREQYG IQRVEAMLHT LERINSDPT LLPNITLGCE IRDSCWHSAV ALEQSIEFIR DSLISSEEEE GLVRCVDGSS SSFRSKKPIV GVIGPGSSSV A IQVQNLLQ LFNIPQIAYS ATSMDLSDKT LFKYFMRVVP SDAQQARAMV DIVKRYNWTY VSAVHTEGNY GESGMEAFKD MS AKEGICI AHSYKIYSNA GEQSFDKLLK KLTSHLPKAR VVACFCEGMT VRGLLMAMRR LGLAGEFLLL GSDGWADRYD VTD GYQREA VGGITIKLQS PDVKWFDDYY LKLRPETNHR NPWFQEFWQH RFQCRLEGFP QENSKYNKTC NSSLTLKTHH VQDS KMGFV INAIYSMAYG LHNMQMSLCP GYAGLCDAMK PIDGRKLLES LMKTNFTGVS GDTILFDENG DSPGRYEIMN FKEMG KDYF DYINVGSWDN GELKMDDDEV WSKKSNIIRS VCSEPCEKGQ IKVIRKGEVS CCWTCTPCKE NEYVFDEYTC KACQLG SWP TDDLTGCDLI PVQYLRWGDP EPIAAVVFAC LGLLATLFVT VVFIIYRDTP VVKSSSRELC YIILAGICLG YLCTFCL IA KPKQIYCYLQ RIGIGLSPAM SYSALVTKTN RIARILAGSK KKICTKKPRF MSACAQLVIA FILICIQLGI IVALFIME P PDIMHDYPSI REVYLICNTT NLGVVTPLGY NGLLILSCTF YAFKTRNVPA NFNEAKYIAF TMYTTCIIWL AFVPIYFGS NYKIITMCFS VSLSATVALG CMFVPKVYII LAKPERNVRS AFTTSTVVRM HVGDGKSSSA ASRSSSLVNL WKRRGSSGET L UniProtKB: Metabotropic glutamate receptor 5 |
-Macromolecule #2: Nanobody 43
| Macromolecule | Name: Nanobody 43 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 13.354672 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: QVQLVESGGG LVQAGGSLRL SCAASGRTFT SYAMGWFRQA PGKERESVAA ISSSGGSTHY ADSVKGRFTI SRDNSKNTVY LQMNSLKPE DTAVYYCAAA MYGSRWPDWE YDYWGQGTQV TVSS |
-Macromolecule #3: (S)-2-AMINO-3-(3,5-DIOXO-[1,2,4]OXADIAZOLIDIN-2-YL)-PROPIONIC ACID
| Macromolecule | Name: (S)-2-AMINO-3-(3,5-DIOXO-[1,2,4]OXADIAZOLIDIN-2-YL)-PROPIONIC ACID type: ligand / ID: 3 / Number of copies: 2 / Formula: QUS |
|---|---|
| Molecular weight | Theoretical: 189.126 Da |
| Chemical component information | 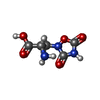 ChemComp-QUS: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.4 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 0.7000000000000001 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller
















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)





















