+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-2937 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Electron cryo-microscopy structure of PB1-p62 type T filaments | |||||||||
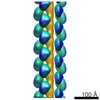
 マップデータ マップデータ | 3D reconstruction of PB1(1-122) type T | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード | Selective autophagy / autophagy receptor / autophagy scaffold / p62/SQSTM1 / single-particle helical reconstruction | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報: / protein binding / protein localization to perinuclear region of cytoplasm / brown fat cell proliferation / protein targeting to vacuole involved in autophagy / regulation of Ras protein signal transduction / intracellular membraneless organelle / aggrephagy / response to mitochondrial depolarisation / negative regulation of toll-like receptor 4 signaling pathway ...: / protein binding / protein localization to perinuclear region of cytoplasm / brown fat cell proliferation / protein targeting to vacuole involved in autophagy / regulation of Ras protein signal transduction / intracellular membraneless organelle / aggrephagy / response to mitochondrial depolarisation / negative regulation of toll-like receptor 4 signaling pathway / regulation of autophagy of mitochondrion / amphisome / protein heterooligomerization / regulation of protein complex stability / endosome organization / autophagy of mitochondrion / pexophagy / membraneless organelle assembly / phagophore assembly site / regulation of mitochondrion organization / ubiquitin-modified protein reader activity / regulation of canonical NF-kappaB signal transduction / Nuclear events mediated by NFE2L2 / aggresome / endosomal transport / K63-linked polyubiquitin modification-dependent protein binding / cellular response to stress / neurotrophin TRK receptor signaling pathway / Lewy body / temperature homeostasis / autolysosome / negative regulation of ferroptosis / response to stress / positive regulation of macroautophagy / molecular sequestering activity / immune system process / mitophagy / energy homeostasis / inclusion body / signaling adaptor activity / negative regulation of protein ubiquitination / positive regulation of autophagy / autophagosome / ionotropic glutamate receptor binding / p75NTR recruits signalling complexes / SH2 domain binding / NF-kB is activated and signals survival / Pexophagy / NRIF signals cell death from the nucleus / response to ischemia / protein kinase C binding / positive regulation of long-term synaptic potentiation / PINK1-PRKN Mediated Mitophagy / sarcomere / protein catabolic process / positive regulation of protein localization to plasma membrane / ubiquitin binding / macroautophagy / apoptotic signaling pathway / P-body / protein sequestering activity / molecular condensate scaffold activity / positive regulation of protein phosphorylation / PML body / receptor tyrosine kinase binding / autophagy / Interleukin-1 signaling / protein import into nucleus / Signaling by ALK fusions and activated point mutants / intracellular protein localization / KEAP1-NFE2L2 pathway / late endosome / Neddylation / signaling receptor activity / cytoplasmic vesicle / sperm midpiece / transcription by RNA polymerase II / ubiquitin-dependent protein catabolic process / protein phosphorylation / protein-macromolecule adaptor activity / cell differentiation / lysosome / endosome / intracellular signal transduction / positive regulation of apoptotic process / protein serine/threonine kinase activity / apoptotic process / ubiquitin protein ligase binding / protein kinase binding / negative regulation of apoptotic process / protein-containing complex binding / glutamatergic synapse / enzyme binding / negative regulation of transcription by RNA polymerase II / endoplasmic reticulum / protein homodimerization activity / positive regulation of transcription by RNA polymerase II / mitochondrion / extracellular exosome / zinc ion binding 類似検索 - 分子機能 | |||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | |||||||||
| 手法 | らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 10.3 Å | |||||||||
 データ登録者 データ登録者 | Ciuffa R / Lamark T / Tarafder A / Guesdon A / Rybina S / Hagen WJH / Johansen T / Sachse C | |||||||||
 引用 引用 |  ジャーナル: Cell Rep / 年: 2015 ジャーナル: Cell Rep / 年: 2015タイトル: The selective autophagy receptor p62 forms a flexible filamentous helical scaffold. 著者: Rodolfo Ciuffa / Trond Lamark / Abul K Tarafder / Audrey Guesdon / Sofia Rybina / Wim J H Hagen / Terje Johansen / Carsten Sachse /   要旨: The scaffold protein p62/SQSTM1 is involved in protein turnover and signaling and is commonly found in dense protein bodies in eukaryotic cells. In autophagy, p62 acts as a selective autophagy ...The scaffold protein p62/SQSTM1 is involved in protein turnover and signaling and is commonly found in dense protein bodies in eukaryotic cells. In autophagy, p62 acts as a selective autophagy receptor that recognizes and shuttles ubiquitinated proteins to the autophagosome for degradation. The structural organization of p62 in cellular bodies and the interplay of these assemblies with ubiquitin and the autophagic marker LC3 remain to be elucidated. Here, we present a cryo-EM structural analysis of p62. Together with structures of assemblies from the PB1 domain, we show that p62 is organized in flexible polymers with the PB1 domain constituting a helical scaffold. Filamentous p62 is capable of binding LC3 and addition of long ubiquitin chains induces disassembly and shortening of filaments. These studies explain how p62 assemblies provide a large molecular scaffold for the nascent autophagosome and reveal how they can bind ubiquitinated cargo. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_2937.map.gz emd_2937.map.gz | 12.2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-2937-v30.xml emd-2937-v30.xml emd-2937.xml emd-2937.xml | 23.3 KB 23.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_2937.png emd_2937.png | 443 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-2937 http://ftp.pdbj.org/pub/emdb/structures/EMD-2937 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2937 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-2937 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_2937.map.gz / 形式: CCP4 / 大きさ: 18.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_2937.map.gz / 形式: CCP4 / 大きさ: 18.3 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | 3D reconstruction of PB1(1-122) type T | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 これらの図は立方格子座標系で作成されたものです | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.372 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
- 試料の構成要素
試料の構成要素
-全体 : PB1(1-122) domain of p62/Sqstm1
| 全体 | 名称: PB1(1-122) domain of p62/Sqstm1 |
|---|---|
| 要素 |
|
-超分子 #1000: PB1(1-122) domain of p62/Sqstm1
| 超分子 | 名称: PB1(1-122) domain of p62/Sqstm1 / タイプ: sample / ID: 1000 / 詳細: Helical polymer / 集合状態: Helical / Number unique components: 1 |
|---|---|
| 分子量 | 実験値: 13.7 KDa / 理論値: 13.7 KDa / 手法: Theoretical weight of construct |
-分子 #1: Sequestosome-1
| 分子 | 名称: Sequestosome-1 / タイプ: protein_or_peptide / ID: 1 / Name.synonym: p62/SQSTM1 / 集合状態: Helical / 組換発現: Yes |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) / 別称: Human Homo sapiens (ヒト) / 別称: Human |
| 分子量 | 実験値: 13.7 KDa / 理論値: 13.7 KDa |
| 組換発現 | 生物種:  |
| 配列 | UniProtKB: Sequestosome-1 GO: phagophore assembly site, autophagy of mitochondrion, P-body, P-body, positive regulation of protein phosphorylation, immune system process, protein serine/threonine kinase activity, protein ...GO: phagophore assembly site, autophagy of mitochondrion, P-body, P-body, positive regulation of protein phosphorylation, immune system process, protein serine/threonine kinase activity, protein kinase C binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, protein binding, nucleus, nucleus, nucleoplasm, cytoplasm, cytoplasm, cytoplasm, lysosome, lysosome, endosome, late endosome, autophagosome, autophagosome, autophagosome, endoplasmic reticulum, endoplasmic reticulum, cytosol, cytosol, cytosol, cytosol, cytosol, cytosol, cytosol, cytosol, cytosol, cytosol, protein phosphorylation, ubiquitin-dependent protein catabolic process, autophagy, autophagy, autophagy, apoptotic process, response to stress, intracellular protein localization, zinc ion binding, regulation of mitochondrion organization, endosomal transport, inclusion body, aggresome, aggresome, macroautophagy, positive regulation of macroautophagy, PML body, protein kinase binding, cell differentiation, receptor tyrosine kinase binding, cytoplasmic vesicle, intracellular signal transduction, SH2 domain binding, identical protein binding, identical protein binding, identical protein binding, identical protein binding, identical protein binding, protein homodimerization activity, positive regulation of apoptotic process, negative regulation of apoptotic process, regulation of canonical NF-kappaB signal transduction, ubiquitin binding, positive regulation of transcription by RNA polymerase II, regulation of Ras protein signal transduction, metal ion binding, neurotrophin TRK receptor signaling pathway, neurotrophin TRK receptor signaling pathway, protein heterooligomerization, extracellular exosome, K63-linked polyubiquitin modification-dependent protein binding, apoptotic signaling pathway, GO: 0098779, regulation of autophagy of mitochondrion InterPro: PB1 domain, UBA-like superfamily, Ubiquitin-associated domain, Zinc finger, ZZ-type |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | helical array |
- 試料調製
試料調製
| 濃度 | 0.25 mg/mL |
|---|---|
| 緩衝液 | pH: 7.5 / 詳細: 50 mM Tris pH 7.5, 100 mM NaCl, DTT 4 mM |
| グリッド | 詳細: glow-discharged C-flat 1.2/1.3 and 200 mesh Quantifoil multi-A grids |
| 凍結 | 凍結剤: ETHANE / チャンバー内温度: 77 K / 装置: HOMEMADE PLUNGER / 手法: Backside blotting |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 日付 | 2012年10月9日 |
| 撮影 | カテゴリ: CCD フィルム・検出器のモデル: GATAN ULTRASCAN 4000 (4k x 4k) 実像数: 443 / 平均電子線量: 10 e/Å2 |
| 電子線 | 加速電圧: 120 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm / 最大 デフォーカス(公称値): 3.5 µm / 最小 デフォーカス(公称値): 1.5 µm / 倍率(公称値): 59000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 詳細 | All of the image processing was carried using the SPRING package. |
|---|---|
| 最終 再構成 | 想定した対称性 - らせんパラメータ - Δz: 10.09 Å 想定した対称性 - らせんパラメータ - ΔΦ: 26.71 ° 想定した対称性 - らせんパラメータ - 軸対称性: C1 (非対称) アルゴリズム: OTHER / 解像度のタイプ: BY AUTHOR / 解像度: 10.3 Å / 解像度の算出法: OTHER / ソフトウェア - 名称: SPRING |
| CTF補正 | 詳細: CTFFIND, convolution images, Wiener filter reconstruction |
| 最終 角度割当 | 詳細: SPIDER |
 ムービー
ムービー コントローラー
コントローラー


















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)






















