[English] 日本語
 Yorodumi
Yorodumi- EMDB-27756: Primase of mutant bacteriophage T4 primosome with single strand D... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Primase of mutant bacteriophage T4 primosome with single strand DNA/RNA primer hybrid in primer exiting state | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | phage / complex / helicase / REPLICATION-DNA-RNA complex | |||||||||
| Function / homology | DNA primase, bacteriophage T4 / viral DNA genome replication / Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases / DNA-directed RNA polymerase activity / DNA replication / metal ion binding / DNA primase Function and homology information Function and homology information | |||||||||
| Biological species |  Escherichia phage T4 (virus) Escherichia phage T4 (virus) | |||||||||
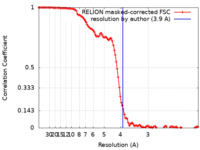
| Method | single particle reconstruction / cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Feng X / Li H | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation | Journal: bioRxiv / Year: 2023 Title: Structural basis of the T4 bacteriophage primosome assembly and primer synthesis. Authors: Xiang Feng / Michelle M Spiering / Ruda de Luna Almeida Santos / Stephen J Benkovic / Huilin Li /  Abstract: The T4 bacteriophage gp41 helicase and gp61 primase assemble into a primosome complex to couple DNA unwinding with RNA primer synthesis for DNA replication. How a primosome is assembled and how the ...The T4 bacteriophage gp41 helicase and gp61 primase assemble into a primosome complex to couple DNA unwinding with RNA primer synthesis for DNA replication. How a primosome is assembled and how the length of the RNA primer is defined in the T4 bacteriophage, or in any model system, are unclear. Here we report a series of cryo-EM structures of T4 primosome assembly intermediates at resolutions up to 2.7 Å. We show that the gp41 helicase is an open spiral in the absence of ssDNA, and ssDNA binding triggers a large-scale scissor-like conformational change that drives the open spiral to a closed ring that activates the helicase. We found that the activation of the gp41 helicase exposes a cryptic hydrophobic primase-binding surface allowing for the recruitment of the gp61 primase. The primase binds the gp41 helicase in a bipartite mode in which the N-terminal Zn-binding domain (ZBD) and the C-terminal RNA polymerase domain (RPD) each contain a helicase-interacting motif (HIM1 and HIM2, respectively) that bind to separate gp41 N-terminal hairpin dimers, leading to the assembly of one primase on the helicase hexamer. Based on two observed primosome conformations - one in a DNA-scanning mode and the other in a post RNA primer-synthesis mode - we suggest that the linker loop between the gp61 ZBD and RPD contributes to the T4 pentaribonucleotide primer. Our study reveals T4 primosome assembly process and sheds light on RNA primer synthesis mechanism. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_27756.map.gz emd_27756.map.gz | 116.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-27756-v30.xml emd-27756-v30.xml emd-27756.xml emd-27756.xml | 19.5 KB 19.5 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_27756_fsc.xml emd_27756_fsc.xml | 11.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_27756.png emd_27756.png | 145.5 KB | ||
| Others |  emd_27756_half_map_1.map.gz emd_27756_half_map_1.map.gz emd_27756_half_map_2.map.gz emd_27756_half_map_2.map.gz | 98.4 MB 98.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-27756 http://ftp.pdbj.org/pub/emdb/structures/EMD-27756 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27756 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-27756 | HTTPS FTP |
-Related structure data
| Related structure data |  8dwjMC  8dtpC  8dueC  8duoC  8dvfC  8dviC  8dw6C  8g0zC  8gaoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_27756.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_27756.map.gz / Format: CCP4 / Size: 125 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.029 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_27756_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_27756_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Complex of T4 bacteriophage primosome including helicase gp41, pr...
| Entire | Name: Complex of T4 bacteriophage primosome including helicase gp41, primase gp61 and ssDNA/RNA hybrid |
|---|---|
| Components |
|
-Supramolecule #1: Complex of T4 bacteriophage primosome including helicase gp41, pr...
| Supramolecule | Name: Complex of T4 bacteriophage primosome including helicase gp41, primase gp61 and ssDNA/RNA hybrid type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Escherichia phage T4 (virus) Escherichia phage T4 (virus) |
-Macromolecule #1: RNA(5'-ppp-GCCGA-3')
| Macromolecule | Name: RNA(5'-ppp-GCCGA-3') / type: rna / ID: 1 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Escherichia phage T4 (virus) Escherichia phage T4 (virus) |
| Molecular weight | Theoretical: 1.744983 KDa |
| Sequence | String: (GTP)CCGA |
-Macromolecule #2: DNA (70-MER)
| Macromolecule | Name: DNA (70-MER) / type: dna / ID: 2 / Number of copies: 1 / Classification: DNA |
|---|---|
| Source (natural) | Organism:  Escherichia phage T4 (virus) Escherichia phage T4 (virus) |
| Molecular weight | Theoretical: 21.900025 KDa |
| Sequence | String: (DG)(DA)(DA)(DT)(DG)(DA)(DG)(DG)(DA)(DG) (DT)(DA)(DG)(DT)(DA)(DG)(DT)(DG)(DA)(DA) (DT)(DG)(DT)(DA)(DG)(DT)(DG)(DA)(DG) (DG)(DT)(DA)(DA)(DT)(DA)(DT)(DC)(DG)(DG) (DC) (DT)(DG)(DG)(DT)(DC)(DT) ...String: (DG)(DA)(DA)(DT)(DG)(DA)(DG)(DG)(DA)(DG) (DT)(DA)(DG)(DT)(DA)(DG)(DT)(DG)(DA)(DA) (DT)(DG)(DT)(DA)(DG)(DT)(DG)(DA)(DG) (DG)(DT)(DA)(DA)(DT)(DA)(DT)(DC)(DG)(DG) (DC) (DT)(DG)(DG)(DT)(DC)(DT)(DG)(DG) (DT)(DC)(DT)(DG)(DT)(DG)(DC)(DC)(DA)(DA) (DG)(DT) (DT)(DG)(DC)(DT)(DG)(DC)(DA) (DA)(DA)(DA) |
-Macromolecule #3: DNA primase
| Macromolecule | Name: DNA primase / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO EC number: Transferases; Transferring phosphorus-containing groups; Nucleotidyltransferases |
|---|---|
| Source (natural) | Organism:  Escherichia phage T4 (virus) Escherichia phage T4 (virus) |
| Molecular weight | Theoretical: 39.842043 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: MSSIPWIDNE FAYRALAHLP KFTQVNNSST FKLRFRCPVC GDSKTDQNKA RGWYYGDNNE GNIHCYNCNY HAPIGIYLKE FEPDLYREY IFEIRKEKGK SRPIEKPKEL PKQPEKKIIK SLPSCVRLDK LAEDHPIIKY VKARCIPKDK WKYLWFTTEW P KLVNSIAP ...String: MSSIPWIDNE FAYRALAHLP KFTQVNNSST FKLRFRCPVC GDSKTDQNKA RGWYYGDNNE GNIHCYNCNY HAPIGIYLKE FEPDLYREY IFEIRKEKGK SRPIEKPKEL PKQPEKKIIK SLPSCVRLDK LAEDHPIIKY VKARCIPKDK WKYLWFTTEW P KLVNSIAP GTYKKEISEP RLVIPIYNAN GKAESFQGRA LKKDAPQKYI TIEAYPEATK IYGVERVKDG DVYVLEGPID SL FIENGIA ITGGQLDLEV VPFKDRRVWV LDNEPRHPDT IKRMTKLVDA GERVMFWDKS PWKSKDVNDM IRKEGATPEQ IME YMKNNI AQGLMAKMRL SKYAKI UniProtKB: DNA primase |
-Macromolecule #4: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 4 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.8 Component:
Details: 20 mM HEPES pH 7.8, 100 mM NaCl, 10 mM MgCl2 and 2 mM DTT | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 66.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller














 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)