[English] 日本語
 Yorodumi
Yorodumi- EMDB-25539: Structure of the delta dII IRES eIF2-containing 48S initiation co... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Structure of the delta dII IRES eIF2-containing 48S initiation complex, closed conformation. Structure 12(delta dII). | ||||||||||||
 Map data Map data | Post processed map | ||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords | HCV / IRES / 40S / RIBOSOME | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationtranslation initiation ternary complex / regulation of translation in response to endoplasmic reticulum stress / glial limiting end-foot / HRI-mediated signaling / response to manganese-induced endoplasmic reticulum stress / Cellular response to mitochondrial stress / positive regulation of type B pancreatic cell apoptotic process / Response of EIF2AK1 (HRI) to heme deficiency / Recycling of eIF2:GDP / negative regulation of translational initiation in response to stress ...translation initiation ternary complex / regulation of translation in response to endoplasmic reticulum stress / glial limiting end-foot / HRI-mediated signaling / response to manganese-induced endoplasmic reticulum stress / Cellular response to mitochondrial stress / positive regulation of type B pancreatic cell apoptotic process / Response of EIF2AK1 (HRI) to heme deficiency / Recycling of eIF2:GDP / negative regulation of translational initiation in response to stress / PERK-mediated unfolded protein response / PERK regulates gene expression / response to kainic acid / eukaryotic translation initiation factor 2 complex / multi-eIF complex / regulation of translational initiation in response to stress / eukaryotic 43S preinitiation complex / translation factor activity, RNA binding / eukaryotic 48S preinitiation complex / Formation of the ternary complex, and subsequently, the 43S complex / laminin receptor activity / Ribosomal scanning and start codon recognition / Translation initiation complex formation / Formation of a pool of free 40S subunits / 90S preribosome / ubiquitin ligase inhibitor activity / Response of EIF2AK4 (GCN2) to amino acid deficiency / positive regulation of signal transduction by p53 class mediator / GTP hydrolysis and joining of the 60S ribosomal subunit / L13a-mediated translational silencing of Ceruloplasmin expression / mitophagy / phagocytic cup / translation regulator activity / rough endoplasmic reticulum / ribosomal small subunit export from nucleus / laminin binding / translation initiation factor activity / gastrulation / MDM2/MDM4 family protein binding / cytosolic ribosome / class I DNA-(apurinic or apyrimidinic site) endonuclease activity / response to endoplasmic reticulum stress / cellular response to amino acid starvation / stress granule assembly / DNA-(apurinic or apyrimidinic site) lyase / ribosome assembly / positive regulation of apoptotic signaling pathway / maturation of SSU-rRNA from tricistronic rRNA transcript (SSU-rRNA, 5.8S rRNA, LSU-rRNA) / maturation of SSU-rRNA / translational initiation / small-subunit processome / PKR-mediated signaling / ABC-family protein mediated transport / spindle / cytoplasmic stress granule / rRNA processing / cellular response to UV / regulation of translation / positive regulation of canonical Wnt signaling pathway / rhythmic process / cellular response to heat / ribosomal small subunit assembly / virus receptor activity / ribosome binding / ribosomal small subunit biogenesis / cellular response to oxidative stress / small ribosomal subunit / small ribosomal subunit rRNA binding / cytosolic small ribosomal subunit / perikaryon / cell differentiation / cytoplasmic translation / tRNA binding / mitochondrial inner membrane / postsynaptic density / rRNA binding / structural constituent of ribosome / ribosome / translation / ribonucleoprotein complex / cell division / DNA repair / mRNA binding / apoptotic process / centrosome / synapse / dendrite / nucleolus / perinuclear region of cytoplasm / Golgi apparatus / mitochondrion / DNA binding / RNA binding / extracellular exosome / zinc ion binding / membrane / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | ||||||||||||
| Biological species |   Hepatitis C virus (isolate 1) / Hepatitis C virus (isolate 1) /  Homo sapiens (human) Homo sapiens (human) | ||||||||||||
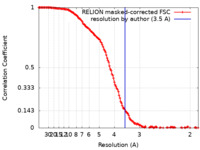
| Method | single particle reconstruction / cryo EM / Resolution: 3.5 Å | ||||||||||||
 Authors Authors | Brown ZP / Abaeva IS / De S / Hellen CUT / Pestova TV / Frank J | ||||||||||||
| Funding support |  United States, 3 items United States, 3 items
| ||||||||||||
 Citation Citation |  Journal: EMBO J / Year: 2022 Journal: EMBO J / Year: 2022Title: Molecular architecture of 40S translation initiation complexes on the hepatitis C virus IRES. Authors: Zuben P Brown / Irina S Abaeva / Swastik De / Christopher U T Hellen / Tatyana V Pestova / Joachim Frank /  Abstract: Hepatitis C virus mRNA contains an internal ribosome entry site (IRES) that mediates end-independent translation initiation, requiring a subset of eukaryotic initiation factors (eIFs). Biochemical ...Hepatitis C virus mRNA contains an internal ribosome entry site (IRES) that mediates end-independent translation initiation, requiring a subset of eukaryotic initiation factors (eIFs). Biochemical studies revealed that direct binding of the IRES to the 40S ribosomal subunit places the initiation codon into the P site, where it base pairs with eIF2-bound Met-tRNAiMet forming a 48S initiation complex. Subsequently, eIF5 and eIF5B mediate subunit joining, yielding an elongation-competent 80S ribosome. Initiation can also proceed without eIF2, in which case Met-tRNAiMet is recruited directly by eIF5B. However, the structures of initiation complexes assembled on the HCV IRES, the transitions between different states, and the accompanying conformational changes have remained unknown. To fill these gaps, we now obtained cryo-EM structures of IRES initiation complexes, at resolutions up to 3.5 Å, that cover all major stages from the initial ribosomal association, through eIF2-containing 48S initiation complexes, to eIF5B-containing complexes immediately prior to subunit joining. These structures provide insights into the dynamic network of 40S/IRES contacts, highlight the role of IRES domain II, and reveal conformational changes that occur during the transition from eIF2- to eIF5B-containing 48S complexes and prepare them for subunit joining. | ||||||||||||
| History |
|
- Structure visualization
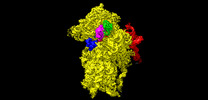
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25539.map.gz emd_25539.map.gz | 228.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25539-v30.xml emd-25539-v30.xml emd-25539.xml emd-25539.xml | 64.3 KB 64.3 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25539_fsc.xml emd_25539_fsc.xml | 14.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_25539.png emd_25539.png | 38.8 KB | ||
| Masks |  emd_25539_msk_1.map emd_25539_msk_1.map | 244.1 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-25539.cif.gz emd-25539.cif.gz | 13.2 KB | ||
| Others |  emd_25539_additional_1.map.gz emd_25539_additional_1.map.gz emd_25539_additional_2.map.gz emd_25539_additional_2.map.gz emd_25539_additional_3.map.gz emd_25539_additional_3.map.gz emd_25539_half_map_1.map.gz emd_25539_half_map_1.map.gz emd_25539_half_map_2.map.gz emd_25539_half_map_2.map.gz | 122.4 MB 143.5 MB 192.2 MB 193.6 MB 193.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25539 http://ftp.pdbj.org/pub/emdb/structures/EMD-25539 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25539 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25539 | HTTPS FTP |
-Related structure data
| Related structure data |  7sysMC  7syiC  7syjC  7sykC  7sylC  7syoC  7sypC  7syqC  7syrC  7sytC  7syuC  7syvC  7sywC  7syxC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_25539.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25539.map.gz / Format: CCP4 / Size: 244.1 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post processed map | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.95 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_25539_msk_1.map emd_25539_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Local resolution values
| File | emd_25539_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Local resolution values | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Local resolution map filtered at local resolution
| File | emd_25539_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Local resolution map filtered at local resolution | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: Unsharpened map
| File | emd_25539_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map
| File | emd_25539_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map
| File | emd_25539_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : 40S ribosomal small subunit with HCV IRES
+Supramolecule #1: 40S ribosomal small subunit with HCV IRES
+Macromolecule #1: 18S rRNA
+Macromolecule #38: HCV IRES
+Macromolecule #39: Met-tRNA-i-Met
+Macromolecule #2: Eukaryotic translation initiation factor 1A, X-chromosomal
+Macromolecule #3: uS2 (SA)
+Macromolecule #4: eS1
+Macromolecule #5: uS5
+Macromolecule #6: uS3
+Macromolecule #7: eS4 (S4 X isoform)
+Macromolecule #8: uS7
+Macromolecule #9: eS6
+Macromolecule #10: eS7
+Macromolecule #11: eS8
+Macromolecule #12: uS4
+Macromolecule #13: eS10
+Macromolecule #14: uS17
+Macromolecule #15: eS12
+Macromolecule #16: uS15
+Macromolecule #17: uS11
+Macromolecule #18: uS19
+Macromolecule #19: uS9
+Macromolecule #20: eS17
+Macromolecule #21: uS13
+Macromolecule #22: eS19
+Macromolecule #23: uS10
+Macromolecule #24: eS21
+Macromolecule #25: uS8
+Macromolecule #26: uS12
+Macromolecule #27: eS24
+Macromolecule #28: eS25
+Macromolecule #29: eS26
+Macromolecule #30: eS27
+Macromolecule #31: eS28
+Macromolecule #32: uS14
+Macromolecule #33: eS30
+Macromolecule #34: eS31
+Macromolecule #35: Receptor for Activated C Kinase 1 (RACK1)
+Macromolecule #36: Eukaryotic translation initiation factor 2 subunit 1
+Macromolecule #37: eL41
+Macromolecule #40: ZINC ION
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.000075 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R0.6/1 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY / Support film - Film thickness: 50 / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 25 sec. / Pretreatment - Atmosphere: OTHER / Details: H2/O2 mixture for 25 seconds at 25W power |
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV / Details: 4 second blot time, force 3. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TECNAI F30 |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average exposure time: 4.0 sec. / Average electron dose: 70.9 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.26 mm / Nominal defocus max: 2.0 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 56000 |
| Sample stage | Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Tecnai F30 / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model |
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Output model |  PDB-7sys: |
 Movie
Movie Controller
Controller
























































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)