[English] 日本語
 Yorodumi
Yorodumi- EMDB-25176: Cryo-EM structure of human ACKR3 in complex with CXCL12, a small ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of human ACKR3 in complex with CXCL12, a small molecule partial agonist CCX662, an extracellular Fab, and an intracellular Fab | ||||||||||||||||||||||||
 Map data Map data | |||||||||||||||||||||||||
 Sample Sample |
| ||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationoculomotor nerve development / chemokine (C-X-C motif) ligand 12 signaling pathway / negative regulation of leukocyte tethering or rolling / C-X-C chemokine binding / positive regulation of mesenchymal stem cell migration / response to ultrasound / telencephalon cell migration / regulation of actin polymerization or depolymerization / C-X-C chemokine receptor activity / CXCL12-activated CXCR4 signaling pathway ...oculomotor nerve development / chemokine (C-X-C motif) ligand 12 signaling pathway / negative regulation of leukocyte tethering or rolling / C-X-C chemokine binding / positive regulation of mesenchymal stem cell migration / response to ultrasound / telencephalon cell migration / regulation of actin polymerization or depolymerization / C-X-C chemokine receptor activity / CXCL12-activated CXCR4 signaling pathway / chemokine receptor binding / positive regulation of vasculature development / CXCR chemokine receptor binding / positive regulation of axon extension involved in axon guidance / positive regulation of dopamine secretion / C-C chemokine receptor activity / scavenger receptor activity / Signaling by ROBO receptors / C-C chemokine binding / induction of positive chemotaxis / integrin activation / cellular response to chemokine / negative regulation of intrinsic apoptotic signaling pathway in response to DNA damage / negative regulation of dendritic cell apoptotic process / positive regulation of monocyte chemotaxis / chemokine-mediated signaling pathway / blood circulation / Chemokine receptors bind chemokines / chemokine activity / positive regulation of calcium ion import / detection of temperature stimulus involved in sensory perception of pain / animal organ regeneration / detection of mechanical stimulus involved in sensory perception of pain / positive regulation of T cell migration / vasculogenesis / coreceptor activity / clathrin-coated pit / Nuclear signaling by ERBB4 / positive regulation of endothelial cell proliferation / positive regulation of neuron differentiation / positive regulation of cell adhesion / cell chemotaxis / adult locomotory behavior / axon guidance / calcium-mediated signaling / growth factor activity / neuron migration / response to virus / receptor internalization / recycling endosome / response to peptide hormone / defense response / intracellular calcium ion homeostasis / chemotaxis / integrin binding / positive regulation of cytosolic calcium ion concentration / G alpha (i) signalling events / angiogenesis / collagen-containing extracellular matrix / Estrogen-dependent gene expression / positive regulation of ERK1 and ERK2 cascade / early endosome / response to hypoxia / cell adhesion / endosome / immune response / G protein-coupled receptor signaling pathway / negative regulation of cell population proliferation / external side of plasma membrane / intracellular membrane-bounded organelle / signaling receptor binding / cell surface / signal transduction / extracellular exosome / extracellular region / plasma membrane Similarity search - Function | ||||||||||||||||||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | ||||||||||||||||||||||||
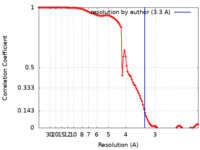
| Method | single particle reconstruction / cryo EM / Resolution: 3.3 Å | ||||||||||||||||||||||||
 Authors Authors | Yen YC / Schafer CT / Gustavsson M / Handel TM / Tesmer JJG | ||||||||||||||||||||||||
| Funding support |  United States, 7 items United States, 7 items
| ||||||||||||||||||||||||
 Citation Citation |  Journal: Sci Adv / Year: 2022 Journal: Sci Adv / Year: 2022Title: Structures of atypical chemokine receptor 3 reveal the basis for its promiscuity and signaling bias. Authors: Yu-Chen Yen / Christopher T Schafer / Martin Gustavsson / Stefanie A Eberle / Pawel K Dominik / Dawid Deneka / Penglie Zhang / Thomas J Schall / Anthony A Kossiakoff / John J G Tesmer / Tracy M Handel /    Abstract: Both CXC chemokine receptor 4 (CXCR4) and atypical chemokine receptor 3 (ACKR3) are activated by the chemokine CXCL12 yet evoke distinct cellular responses. CXCR4 is a canonical G protein-coupled ...Both CXC chemokine receptor 4 (CXCR4) and atypical chemokine receptor 3 (ACKR3) are activated by the chemokine CXCL12 yet evoke distinct cellular responses. CXCR4 is a canonical G protein-coupled receptor (GPCR), whereas ACKR3 is intrinsically biased for arrestin. The molecular basis for this difference is not understood. Here, we describe cryo-EM structures of ACKR3 in complex with CXCL12, a more potent CXCL12 variant, and a small-molecule agonist. The bound chemokines adopt an unexpected pose relative to those established for CXCR4 and observed in other receptor-chemokine complexes. Along with functional studies, these structures provide insight into the ligand-binding promiscuity of ACKR3, why it fails to couple to G proteins, and its bias toward β-arrestin. The results lay the groundwork for understanding the physiological interplay of ACKR3 with other GPCRs. | ||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_25176.map.gz emd_25176.map.gz | 97.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-25176-v30.xml emd-25176-v30.xml emd-25176.xml emd-25176.xml | 21.4 KB 21.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_25176_fsc.xml emd_25176_fsc.xml | 10.4 KB | Display |  FSC data file FSC data file |
| Images |  emd_25176.png emd_25176.png | 83 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-25176 http://ftp.pdbj.org/pub/emdb/structures/EMD-25176 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25176 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-25176 | HTTPS FTP |
-Validation report
| Summary document |  emd_25176_validation.pdf.gz emd_25176_validation.pdf.gz | 425.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_25176_full_validation.pdf.gz emd_25176_full_validation.pdf.gz | 424.8 KB | Display | |
| Data in XML |  emd_25176_validation.xml.gz emd_25176_validation.xml.gz | 11.9 KB | Display | |
| Data in CIF |  emd_25176_validation.cif.gz emd_25176_validation.cif.gz | 15.6 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25176 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25176 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25176 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-25176 | HTTPS FTP |
-Related structure data
| Related structure data |  7sk8MC  7sk3C  7sk4C  7sk5C  7sk6C  7sk7C  7sk9C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_25176.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_25176.map.gz / Format: CCP4 / Size: 103 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.08 Å | ||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
- Sample components
Sample components
+Entire : Complex structure of CID25-ACKR3-CXCL12-CCX662-CID24
+Supramolecule #1: Complex structure of CID25-ACKR3-CXCL12-CCX662-CID24
+Macromolecule #1: Atypical chemokine receptor 3
+Macromolecule #2: Stromal cell-derived factor 1
+Macromolecule #3: CID25 Fab light chain
+Macromolecule #4: CID25 Fab heavy chain
+Macromolecule #5: CID24 Fab light chain
+Macromolecule #6: CID24 Fab heavy chain
+Macromolecule #7: CHOLESTEROL
+Macromolecule #8: Lauryl Maltose Neopentyl Glycol
+Macromolecule #9: (1R)-4-[7-(3-carboxypropoxy)-6-methylquinolin-8-yl]-1-{[2-(4-hydr...
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| |||||||||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Atmosphere: AIR | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 53.8 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 1.0 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller




















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)