+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-21143 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of octameric chicken CALHM1 | |||||||||
 マップデータ マップデータ | sharpened map of chicken calcium homeostasis modulator protein 1 | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報voltage-gated monoatomic ion channel activity / calcium-activated cation channel activity / monoatomic cation channel activity / bioluminescence / generation of precursor metabolites and energy / plasma membrane 類似検索 - 分子機能 | |||||||||
| 生物種 |  | |||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.63 Å | |||||||||
 データ登録者 データ登録者 | Syrjanen JL / Chou TH / Furukawa H | |||||||||
| 資金援助 |  米国, 2件 米国, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2020 ジャーナル: Nat Struct Mol Biol / 年: 2020タイトル: Structure and assembly of calcium homeostasis modulator proteins. 著者: Johanna L Syrjanen / Kevin Michalski / Tsung-Han Chou / Timothy Grant / Shanlin Rao / Noriko Simorowski / Stephen J Tucker / Nikolaus Grigorieff / Hiro Furukawa /   要旨: The biological membranes of many cell types contain large-pore channels through which a wide variety of ions and metabolites permeate. Examples include connexin, innexin and pannexin, which form gap ...The biological membranes of many cell types contain large-pore channels through which a wide variety of ions and metabolites permeate. Examples include connexin, innexin and pannexin, which form gap junctions and/or bona fide cell surface channels. The most recently identified large-pore channels are the calcium homeostasis modulators (CALHMs), through which ions and ATP permeate in a voltage-dependent manner to control neuronal excitability, taste signaling and pathologies of depression and Alzheimer's disease. Despite such critical biological roles, the structures and patterns of their oligomeric assembly remain unclear. Here, we reveal the structures of two CALHMs, chicken CALHM1 and human CALHM2, by single-particle cryo-electron microscopy (cryo-EM), which show novel assembly of the four transmembrane helices into channels of octamers and undecamers, respectively. Furthermore, molecular dynamics simulations suggest that lipids can favorably assemble into a bilayer within the larger CALHM2 pore, but not within CALHM1, demonstrating the potential correlation between pore size, lipid accommodation and channel activity. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_21143.map.gz emd_21143.map.gz | 59.4 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-21143-v30.xml emd-21143-v30.xml emd-21143.xml emd-21143.xml | 13.3 KB 13.3 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_21143.png emd_21143.png | 105 KB | ||
| その他 |  emd_21143_additional.map.gz emd_21143_additional.map.gz emd_21143_additional_1.map.gz emd_21143_additional_1.map.gz | 58.5 MB 58.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-21143 http://ftp.pdbj.org/pub/emdb/structures/EMD-21143 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21143 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-21143 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_21143_validation.pdf.gz emd_21143_validation.pdf.gz | 523.7 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_21143_full_validation.pdf.gz emd_21143_full_validation.pdf.gz | 523.2 KB | 表示 | |
| XML形式データ |  emd_21143_validation.xml.gz emd_21143_validation.xml.gz | 6.4 KB | 表示 | |
| CIF形式データ |  emd_21143_validation.cif.gz emd_21143_validation.cif.gz | 7.3 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21143 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21143 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21143 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-21143 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  6vamMC  6vaiC  6vakC  6valC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10485 (タイトル: Chicken CALHM1, 1 mM EDTA / Data size: 2.4 TB EMPIAR-10485 (タイトル: Chicken CALHM1, 1 mM EDTA / Data size: 2.4 TBData #1: Unaligned movies (.tif) for chicken CALHM1 in nanodisc [micrographs - multiframe]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_21143.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_21143.map.gz / 形式: CCP4 / 大きさ: 64 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | sharpened map of chicken calcium homeostasis modulator protein 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.06 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-追加マップ: unsharpened map of chicken calcium homeostasis modulator protein 1
| ファイル | emd_21143_additional.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | unsharpened map of chicken calcium homeostasis modulator protein 1 | ||||||||||||
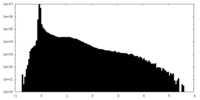
| 投影像・断面図 |
| ||||||||||||
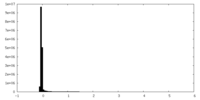
| 密度ヒストグラム |
-追加マップ: unsharpened map of chicken calcium homeostasis modulator protein 1
| ファイル | emd_21143_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | unsharpened map of chicken calcium homeostasis modulator protein 1 | ||||||||||||
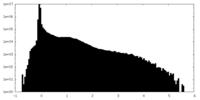
| 投影像・断面図 |
| ||||||||||||
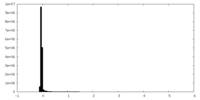
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Octameric chicken CALHM1 in EDTA
| 全体 | 名称: Octameric chicken CALHM1 in EDTA |
|---|---|
| 要素 |
|
-超分子 #1: Octameric chicken CALHM1 in EDTA
| 超分子 | 名称: Octameric chicken CALHM1 in EDTA / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  |
| 組換発現 | 生物種:  |
-分子 #1: Green fluorescent protein,CALHM1 chimera
| 分子 | 名称: Green fluorescent protein,CALHM1 chimera / タイプ: protein_or_peptide / ID: 1 / コピー数: 8 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  |
| 分子量 | 理論値: 71.65782 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MWSHPQFEKG GGSGGGSGGS AWSHPQFEKG AHHHHHHHHA AAMVSKGEEL FTGVVPILVE LDGDVNGHKF SVSGEGEGDA TYGKLTLKF ICTTGKLPVP WPTLVTTLTY GVQCFSRYPD HMKQHDFFKS AMPEGYVQER TIFFKDDGNY KTRAEVKFEG D TLVNRIEL ...文字列: MWSHPQFEKG GGSGGGSGGS AWSHPQFEKG AHHHHHHHHA AAMVSKGEEL FTGVVPILVE LDGDVNGHKF SVSGEGEGDA TYGKLTLKF ICTTGKLPVP WPTLVTTLTY GVQCFSRYPD HMKQHDFFKS AMPEGYVQER TIFFKDDGNY KTRAEVKFEG D TLVNRIEL KGIDFKEDGN ILGHKLEYNY NSHNVYIMAD KQKNGIKVNF KIRHNIEDGS VQLADHYQQN TPIGDGPVLL PD NHYLSTQ SKLSKDPNEK RDHMVLLEFV TAAGITLGMD ELYKSGLRSG LEVLFQGPEF DKFRMVFQFL QSNQESFMSG ICG IMALAS AQLYSAFDFN CPCLPRYNLA YGLGVLLVPP LILFLLGFVL NNNVSMLAEE WRRPQGQRQK DAAVLRYMFC SMVQ RAMIA PAVWVSVTLL DGKCITCAFC TSLPVEALGN ASHHGLPQGE VKRVLARIPC KEIYDGQELI ANEVAVRYLR CISQA LGWC FVLLMTTLAF LVRSLRPCFT QAVFLKSRYW SHYIDIERKL FDETCAEHAK SFAKVCIQQF FQGMSKDLTA THCHPP RKA PTDAGEASEK LLGITDWDTM NIALKSWHRC KPPLHLHPDA PHSSNGWAGE WQPHTQPHTQ PPAPRREAVA YYSGV |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.5 |
|---|---|
| グリッド | 詳細: unspecified |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 85 % / チャンバー内温度: 288.15 K / 装置: FEI VITROBOT MARK IV / 詳細: Blot for 4 sec before plunging. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 平均電子線量: 70.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 最終 再構成 | 想定した対称性 - 点群: C8 (8回回転対称) / 解像度のタイプ: BY AUTHOR / 解像度: 3.63 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: cisTEM / 使用した粒子像数: 308916 |
|---|---|
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
 ムービー
ムービー コントローラー
コントローラー















 Z
Z Y
Y X
X