[English] 日本語
 Yorodumi
Yorodumi- EMDB-1236: Type IV pilus structure by cryo-electron microscopy and crystallo... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-1236 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Type IV pilus structure by cryo-electron microscopy and crystallography: implications for pilus assembly and functions. | |||||||||
 Map data Map data | CryoEM map for Neisseria gonorrhoeae Type IV pilus | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationprotein secretion by the type II secretion system / type II protein secretion system complex / pilus / cell adhesion / membrane Similarity search - Function | |||||||||
| Biological species |  Neisseria gonorrhoeae (bacteria) Neisseria gonorrhoeae (bacteria) | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 12.5 Å | |||||||||
 Authors Authors | Craig L / Egelman EH / Volkmann N / Tainer JA | |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2006 Journal: Mol Cell / Year: 2006Title: Type IV pilus structure by cryo-electron microscopy and crystallography: implications for pilus assembly and functions. Authors: Lisa Craig / Niels Volkmann / Andrew S Arvai / Michael E Pique / Mark Yeager / Edward H Egelman / John A Tainer /  Abstract: Type IV pili (T4P) are long, thin, flexible filaments on bacteria that undergo assembly-disassembly from inner membrane pilin subunits and exhibit astonishing multifunctionality. Neisseria ...Type IV pili (T4P) are long, thin, flexible filaments on bacteria that undergo assembly-disassembly from inner membrane pilin subunits and exhibit astonishing multifunctionality. Neisseria gonorrhoeae (gonococcal or GC) T4P are prototypic virulence factors and immune targets for increasingly antibiotic-resistant human pathogens, yet detailed structures are unavailable for any T4P. Here, we determined a detailed experimental GC-T4P structure by quantitative fitting of a 2.3 A full-length pilin crystal structure into a 12.5 A resolution native GC-T4P reconstruction solved by cryo-electron microscopy (cryo-EM) and iterative helical real space reconstruction. Spiraling three-helix bundles form the filament core, anchor the globular heads, and provide strength and flexibility. Protruding hypervariable loops and posttranslational modifications in the globular head shield conserved functional residues in pronounced grooves, creating a surprisingly corrugated pilus surface. These results clarify T4P multifunctionality and assembly-disassembly while suggesting unified assembly mechanisms for T4P, archaeal flagella, and type II secretion system filaments. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_1236.map.gz emd_1236.map.gz | 207.4 KB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-1236-v30.xml emd-1236-v30.xml emd-1236.xml emd-1236.xml | 12.4 KB 12.4 KB | Display Display |  EMDB header EMDB header |
| Images |  1236.gif 1236.gif | 51.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-1236 http://ftp.pdbj.org/pub/emdb/structures/EMD-1236 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1236 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-1236 | HTTPS FTP |
-Validation report
| Summary document |  emd_1236_validation.pdf.gz emd_1236_validation.pdf.gz | 299.3 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_1236_full_validation.pdf.gz emd_1236_full_validation.pdf.gz | 298.9 KB | Display | |
| Data in XML |  emd_1236_validation.xml.gz emd_1236_validation.xml.gz | 4.4 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1236 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1236 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1236 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-1236 | HTTPS FTP |
-Related structure data
| Related structure data |  2hilMC  2hi2C M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_1236.map.gz / Format: CCP4 / Size: 283.2 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_1236.map.gz / Format: CCP4 / Size: 283.2 KB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | CryoEM map for Neisseria gonorrhoeae Type IV pilus | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. generated in cubic-lattice coordinate | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 2.54 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
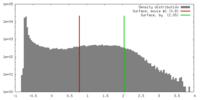
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Type IV pilus filament from Neisseria gonorrhoeae
| Entire | Name: Type IV pilus filament from Neisseria gonorrhoeae |
|---|---|
| Components |
|
-Supramolecule #1000: Type IV pilus filament from Neisseria gonorrhoeae
| Supramolecule | Name: Type IV pilus filament from Neisseria gonorrhoeae / type: sample / ID: 1000 / Details: filaments are variable lengths - 1 to 4 microns Oligomeric state: thousands of pilin subunits form a helical filament Number unique components: 1 |
|---|
-Supramolecule #1: N. gonorrhoeae Type IV pilus
| Supramolecule | Name: N. gonorrhoeae Type IV pilus / type: organelle_or_cellular_component / ID: 1 / Name.synonym: pilus, GC-T4P Details: GC-T4P are variable in length and contail thousands of copies of the pilin subunit. A 1-micron length would contain approx. 952 subunits, with a molecular weight of 17.7 kDa/subunit, giving ...Details: GC-T4P are variable in length and contail thousands of copies of the pilin subunit. A 1-micron length would contain approx. 952 subunits, with a molecular weight of 17.7 kDa/subunit, giving a total molecular weight of approx. 16.8 mDa. Number of copies: 1 / Oligomeric state: helical filament / Recombinant expression: Yes |
|---|---|
| Source (natural) | Organism:  Neisseria gonorrhoeae (bacteria) / Strain: C30 / synonym: bacteria / Cell: N. gonorrhoeae / Organelle: pilus / Location in cell: outer membrane Neisseria gonorrhoeae (bacteria) / Strain: C30 / synonym: bacteria / Cell: N. gonorrhoeae / Organelle: pilus / Location in cell: outer membrane |
| Recombinant expression | Organism: N. gonorrhoeae |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | filament |
- Sample preparation
Sample preparation
| Concentration | 0.3 mg/mL |
|---|---|
| Buffer | pH: 9.5 / Details: 50 mM CHES, pH 9.5 |
| Grid | Details: Quantifoil holey carbon grids, glow-discharged w/ amyl amine |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 4.2 K / Instrument: OTHER / Details: Vitrification instrument: Vitrobot / Method: Blot for 2.5 sec. before plunging |
- Electron microscopy
Electron microscopy
| Microscope | FEI/PHILIPS CM200FEG |
|---|---|
| Temperature | Min: 4.5 K / Max: 4.8 K / Average: 4.5 K |
| Alignment procedure | Legacy - Astigmatism: corrected at 135,000X magnification |
| Details | Images were collected in low dose mode. |
| Date | Jun 23, 2004 |
| Image recording | Category: FILM / Film or detector model: KODAK SO-163 FILM / Digitization - Scanner: OTHER / Digitization - Sampling interval: 12.7 µm / Number real images: 12 / Average electron dose: 10 e/Å2 / Bits/pixel: 14 |
| Electron beam | Acceleration voltage: 120 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Calibrated magnification: 50000 / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.1 µm / Nominal magnification: 50000 |
| Sample stage | Specimen holder: Side entry liquid nitrogen-cooled cryo specimen holder Specimen holder model: GATAN LIQUID NITROGEN |
- Image processing
Image processing
| Details | Native Type IV pili were isoated by shearing from N. gonorrhoeae cells. |
|---|---|
| Final reconstruction | Applied symmetry - Helical parameters - Δz: 10.5 Å Applied symmetry - Helical parameters - Δ&Phi: 100.8 ° Resolution.type: BY AUTHOR / Resolution: 12.5 Å / Resolution method: OTHER / Software - Name: IHRSR Details: The final map was calculated from ~25,000 overlapping particles (508 Angstrom segments with 95% overlap) |
| CTF correction | Details: each micrograph |
-Atomic model buiding 1
| Initial model | PDB ID: |
|---|---|
| Software | Name: CoAn |
| Details | Protocol: rigid body. A single asymmetric unit was isolated from the filament reconstruction using the watershed transform (Volkmann, J Struct Biol 138, 123-129, 2002) and used for unconstrained docking of a single pilin molecule. After fitting the subunit and builiding the filament model (PDB code 2HIL) the density map was corrected by scaling the amplitudes to the spherically averaged molecular transform of the filament model to minimize artifacts from amplitude distortion caused by the CTF and other experimental factors. This correction only affects visual appearance and not the subsequent automated docking process, which automatically corrects for amplitude distortions. |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT Target criteria: real-space density correlation coefficient between the experimental density and the density calculated from the search model |
| Output model |  PDB-2hil: |
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)