[English] 日本語
 Yorodumi
Yorodumi- EMDB-12042: Cryo-EM structure of the human CAK bound to ICEC0942 at 2.5 Angst... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12042 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the human CAK bound to ICEC0942 at 2.5 Angstroms resolution | |||||||||
 Map data Map data | Post-processed (sharpened, filtered) cryo-EM map, re-boxed into a 128 x 128 x 128 pixel box | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Kinase / protein complex / small molecules inhibitor / CDK-activating kinase / TRANSCRIPTION | |||||||||
| Function / homology |  Function and homology information Function and homology informationventricular system development / snRNA transcription by RNA polymerase II / CAK-ERCC2 complex / transcription factor TFIIK complex / adult heart development / transcription factor TFIIH core complex / transcription factor TFIIH holo complex / cyclin-dependent protein serine/threonine kinase activator activity / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity ...ventricular system development / snRNA transcription by RNA polymerase II / CAK-ERCC2 complex / transcription factor TFIIK complex / adult heart development / transcription factor TFIIH core complex / transcription factor TFIIH holo complex / cyclin-dependent protein serine/threonine kinase activator activity / cyclin-dependent kinase / cyclin-dependent protein serine/threonine kinase activity / [RNA-polymerase]-subunit kinase / RNA Polymerase I Transcription Termination / cyclin-dependent protein serine/threonine kinase regulator activity / Cyclin E associated events during G1/S transition / RNA Pol II CTD phosphorylation and interaction with CE during HIV infection / RNA Pol II CTD phosphorylation and interaction with CE / Formation of the Early Elongation Complex / Formation of the HIV-1 Early Elongation Complex / mRNA Capping / Cyclin A/B1/B2 associated events during G2/M transition / HIV Transcription Initiation / RNA Polymerase II HIV Promoter Escape / Transcription of the HIV genome / RNA Polymerase II Promoter Escape / RNA Polymerase II Transcription Pre-Initiation And Promoter Opening / RNA Polymerase II Transcription Initiation / RNA Polymerase II Transcription Initiation And Promoter Clearance / Cyclin A:Cdk2-associated events at S phase entry / cyclin-dependent protein kinase holoenzyme complex / RNA polymerase II CTD heptapeptide repeat kinase activity / regulation of G1/S transition of mitotic cell cycle / RNA Polymerase I Transcription Initiation / RNA polymerase II transcribes snRNA genes / Tat-mediated elongation of the HIV-1 transcript / Formation of HIV-1 elongation complex containing HIV-1 Tat / ATP-dependent activity, acting on DNA / Formation of HIV elongation complex in the absence of HIV Tat / male germ cell nucleus / RNA Polymerase II Transcription Elongation / Formation of RNA Pol II elongation complex / RNA Polymerase II Pre-transcription Events / positive regulation of smooth muscle cell proliferation / RNA Polymerase I Promoter Escape / nucleotide-excision repair / transcription initiation at RNA polymerase II promoter / TP53 Regulates Transcription of DNA Repair Genes / NoRC negatively regulates rRNA expression / response to calcium ion / Cyclin D associated events in G1 / Transcription-Coupled Nucleotide Excision Repair (TC-NER) / G1/S transition of mitotic cell cycle / Formation of TC-NER Pre-Incision Complex / fibrillar center / Formation of Incision Complex in GG-NER / Dual incision in TC-NER / Gap-filling DNA repair synthesis and ligation in TC-NER / RUNX1 regulates transcription of genes involved in differentiation of HSCs / transcription by RNA polymerase II / protein stabilization / regulation of cell cycle / protein kinase activity / cell division / DNA repair / protein serine kinase activity / protein serine/threonine kinase activity / regulation of transcription by RNA polymerase II / negative regulation of apoptotic process / perinuclear region of cytoplasm / positive regulation of transcription by RNA polymerase II / zinc ion binding / nucleoplasm / ATP binding / nucleus / plasma membrane / cytoplasm / cytosol Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 2.5 Å | |||||||||
 Authors Authors | Greber BJ / Remis J | |||||||||
| Funding support |  United States, 1 items United States, 1 items
| |||||||||
 Citation Citation |  Journal: Biophys J / Year: 2021 Journal: Biophys J / Year: 2021Title: 2.5 Å-resolution structure of human CDK-activating kinase bound to the clinical inhibitor ICEC0942. Authors: Basil J Greber / Jonathan Remis / Simak Ali / Eva Nogales /   Abstract: The human CDK-activating kinase (CAK), composed of CDK7, cyclin H, and MAT1, is involved in the control of transcription initiation and the cell cycle. Because of these activities, it has been ...The human CDK-activating kinase (CAK), composed of CDK7, cyclin H, and MAT1, is involved in the control of transcription initiation and the cell cycle. Because of these activities, it has been identified as a promising target for cancer chemotherapy. A number of CDK7 inhibitors have entered clinical trials, among them ICEC0942 (also known as CT7001). Structural information can aid in improving the affinity and specificity of such drugs or drug candidates, reducing side effects in patients. Here, we have determined the structure of the human CAK in complex with ICEC0942 at 2.5 Å-resolution using cryogenic electron microscopy. Our structure reveals conformational differences of ICEC0942 compared with previous X-ray crystal structures of the CDK2-bound complex, and highlights the critical ability of cryogenic electron microscopy to resolve structures of drug-bound protein complexes without the need to crystalize the protein target. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12042.map.gz emd_12042.map.gz | 7.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12042-v30.xml emd-12042-v30.xml emd-12042.xml emd-12042.xml | 23.8 KB 23.8 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_12042.png emd_12042.png | 134.1 KB | ||
| Masks |  emd_12042_msk_1.map emd_12042_msk_1.map | 64 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-12042.cif.gz emd-12042.cif.gz | 7.2 KB | ||
| Others |  emd_12042_half_map_1.map.gz emd_12042_half_map_1.map.gz emd_12042_half_map_2.map.gz emd_12042_half_map_2.map.gz | 49.7 MB 49.6 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12042 http://ftp.pdbj.org/pub/emdb/structures/EMD-12042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12042 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12042 | HTTPS FTP |
-Validation report
| Summary document |  emd_12042_validation.pdf.gz emd_12042_validation.pdf.gz | 923.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_12042_full_validation.pdf.gz emd_12042_full_validation.pdf.gz | 922.7 KB | Display | |
| Data in XML |  emd_12042_validation.xml.gz emd_12042_validation.xml.gz | 11.6 KB | Display | |
| Data in CIF |  emd_12042_validation.cif.gz emd_12042_validation.cif.gz | 13.2 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12042 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12042 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12042 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-12042 | HTTPS FTP |
-Related structure data
| Related structure data |  7b5oMC  7b5qMC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | |
| EM raw data |  EMPIAR-10561 (Title: Cryo-EM structure of the human CDK-activating kinase bound to the clinical inhibitor ICEC0942 EMPIAR-10561 (Title: Cryo-EM structure of the human CDK-activating kinase bound to the clinical inhibitor ICEC0942Data size: 3.8 TB Data #1: Unaligned movies of human CAK-ICEC0942, dataset 1 [micrographs - multiframe] Data #2: Unaligned movies of human CAK-ICEC0942, dataset 3 [micrographs - multiframe] Data #3: Unaligned movies of human CAK-ICEC0942, dataset 2 [micrographs - multiframe]) |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12042.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12042.map.gz / Format: CCP4 / Size: 8 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Post-processed (sharpened, filtered) cryo-EM map, re-boxed into a 128 x 128 x 128 pixel box | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.029 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
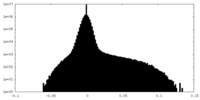
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12042_msk_1.map emd_12042_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Unfiltered half-map
| File | emd_12042_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Unfiltered half-map
| File | emd_12042_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Unfiltered half-map | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : CDK-activating kinase (CAK) in complex with ICEC0942
| Entire | Name: CDK-activating kinase (CAK) in complex with ICEC0942 |
|---|---|
| Components |
|
-Supramolecule #1: CDK-activating kinase (CAK) in complex with ICEC0942
| Supramolecule | Name: CDK-activating kinase (CAK) in complex with ICEC0942 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 120 KDa |
-Macromolecule #1: CDK-activating kinase assembly factor MAT1
| Macromolecule | Name: CDK-activating kinase assembly factor MAT1 / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 38.13234 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MGSSHHHHHH ENLYFQSNAM DDQGCPRCKT TKYRNPSLKL MVNVCGHTLC ESCVDLLFVR GAGNCPECGT PLRKSNFRVQ LFEDPTVDK EVEIRKKVLK IYNKREEDFP SLREYNDFLE EVEEIVFNLT NNVDLDNTKK KMEIYQKENK DVIQKNKLKL T REQEELEE ...String: MGSSHHHHHH ENLYFQSNAM DDQGCPRCKT TKYRNPSLKL MVNVCGHTLC ESCVDLLFVR GAGNCPECGT PLRKSNFRVQ LFEDPTVDK EVEIRKKVLK IYNKREEDFP SLREYNDFLE EVEEIVFNLT NNVDLDNTKK KMEIYQKENK DVIQKNKLKL T REQEELEE ALEVERQENE QRRLFIQKEE QLQQILKRKN KQAFLDELES SDLPVALLLA QHKDRSTQLE MQLEKPKPVK PV TFSTGIK MGQHISLAPI HKLEEALYEY QPLQIETYGP HVPELEMLGR LGYLNHVRAA SPQDLAGGYT SSLACHRALQ DAF SGLFWQ PS UniProtKB: CDK-activating kinase assembly factor MAT1 |
-Macromolecule #2: Cyclin-H
| Macromolecule | Name: Cyclin-H / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 37.695473 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MYHNSSQKRH WTFSSEEQLA RLRADANRKF RCKAVANGKV LPNDPVFLEP HEEMTLCKYY EKRLLEFCSV FKPAMPRSVV GTACMYFKR FYLNNSVMEY HPRIIMLTCA FLACKVDEFN VSSPQFVGNL RESPLGQEKA LEQILEYELL LIQQLNFHLI V HNPYRPFE ...String: MYHNSSQKRH WTFSSEEQLA RLRADANRKF RCKAVANGKV LPNDPVFLEP HEEMTLCKYY EKRLLEFCSV FKPAMPRSVV GTACMYFKR FYLNNSVMEY HPRIIMLTCA FLACKVDEFN VSSPQFVGNL RESPLGQEKA LEQILEYELL LIQQLNFHLI V HNPYRPFE GFLIDLKTRY PILENPEILR KTADDFLNRI ALTDAYLLYT PSQIALTAIL SSASRAGITM ESYLSESLML KE NRTCLSQ LLDIMKSMRN LVKKYEPPRS EEVAVLKQKL ERCHSAELAL NVITKKRKGY EDDDYVSKKS KHEEEEWTDD DLV ESL UniProtKB: Cyclin-H |
-Macromolecule #3: Cyclin-dependent kinase 7
| Macromolecule | Name: Cyclin-dependent kinase 7 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO / EC number: cyclin-dependent kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 43.65107 KDa |
| Recombinant expression | Organism:  Trichoplusia ni (cabbage looper) Trichoplusia ni (cabbage looper) |
| Sequence | String: MASWSHPQFE KGGGSGGGSG GGSWSHPQFE KSGGGSENLY FQSNAMALDV KSRAKRYEKL DFLGEGQFAT VYKARDKNTN QIVAIKKIK LGHRSEAKDG INRTALREIK LLQELSHPNI IGLLDAFGHK SNISLVFDFM ETDLEVIIKD NSLVLTPSHI K AYMLMTLQ ...String: MASWSHPQFE KGGGSGGGSG GGSWSHPQFE KSGGGSENLY FQSNAMALDV KSRAKRYEKL DFLGEGQFAT VYKARDKNTN QIVAIKKIK LGHRSEAKDG INRTALREIK LLQELSHPNI IGLLDAFGHK SNISLVFDFM ETDLEVIIKD NSLVLTPSHI K AYMLMTLQ GLEYLHQHWI LHRDLKPNNL LLDENGVLKL ADFGLAKSFG SPNRAYTHQV VTRWYRAPEL LFGARMYGVG VD MWAVGCI LAELLLRVPF LPGDSDLDQL TRIFETLGTP TEEQWPDMCS LPDYVTFKSF PGIPLHHIFS AAGDDLLDLI QGL FLFNPC ARITATQALK MKYFSNRPGP TPGCQLPRPN CPVETLKEQS NPALAIKRKR TEALEQGGLP KKLIF UniProtKB: Cyclin-dependent kinase 7 |
-Macromolecule #4: (3R,4R)-4-[[[7-[(phenylmethyl)amino]-3-propan-2-yl-pyrazolo[1,5-a...
| Macromolecule | Name: (3R,4R)-4-[[[7-[(phenylmethyl)amino]-3-propan-2-yl-pyrazolo[1,5-a]pyrimidin-5-yl]amino]methyl]piperidin-3-ol type: ligand / ID: 4 / Number of copies: 1 / Formula: I74 |
|---|---|
| Molecular weight | Theoretical: 394.513 Da |
| Chemical component information |  ChemComp-I74: |
-Macromolecule #5: water
| Macromolecule | Name: water / type: ligand / ID: 5 / Number of copies: 73 / Formula: HOH |
|---|---|
| Molecular weight | Theoretical: 18.015 Da |
| Chemical component information |  ChemComp-HOH: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.9 Component:
| ||||||||||||||||||
| Grid | Model: UltrAuFoil R1.2/1.3 / Material: GOLD / Mesh: 300 / Support film - Material: GOLD / Support film - topology: HOLEY / Pretreatment - Type: PLASMA CLEANING / Pretreatment - Time: 30 sec. / Pretreatment - Atmosphere: AIR | ||||||||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 278 K / Instrument: FEI VITROBOT MARK IV | ||||||||||||||||||
| Details | Incubated with 50 uM ICEC0942 for 5 min |
- Electron microscopy
Electron microscopy
| Microscope | FEI TALOS ARCTICA |
|---|---|
| Image recording | #0 - Image recording ID: 1 / #0 - Film or detector model: GATAN K3 (6k x 4k) / #0 - Number grids imaged: 1 / #0 - Number real images: 3571 / #0 - Average exposure time: 2.0 sec. / #0 - Average electron dose: 69.0 e/Å2 #0 - Details: Movies (69 frames) acquired in 3 x 3 pattern by image shift #1 - Image recording ID: 2 / #1 - Film or detector model: GATAN K3 (6k x 4k) / #1 - Number grids imaged: 2 / #1 - Number real images: 5302 / #1 - Average exposure time: 2.0 sec. / #1 - Average electron dose: 69.0 e/Å2 #1 - Details: Movies (70 frames) acquired in 3 x 3 pattern by image shift |
| Electron beam | Acceleration voltage: 200 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 50.0 µm / Calibrated magnification: 72886 / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 1.5 µm / Nominal defocus min: 0.5 µm |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Talos Arctica / Image courtesy: FEI Company |
+ Image processing
Image processing
-Atomic model buiding 1
| Initial model | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| Details | Real space refinement in PHENIX; ligand coordinates from PHENIX-OPLS3e refinement |
| Refinement | Space: REAL / Protocol: RIGID BODY FIT |
| Output model |  PDB-7b5o:  PDB-7b5q: |
 Movie
Movie Controller
Controller






























 Z
Z Y
Y X
X