+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 6p25 | ||||||
|---|---|---|---|---|---|---|---|
| タイトル | Structure of S. cerevisiae protein O-mannosyltransferase Pmt1-Pmt2 complex bound to the sugar donor and a peptide acceptor | ||||||
 要素 要素 |
| ||||||
 キーワード キーワード | TRANSFERASE / complex / glycosylation | ||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報dolichyl-phosphate-mannose-protein mannosyltransferase Pmt1p-Pmt3p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase Pmt1p-Pmt2p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase Pmt5p-Pmt2p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase / dolichyl-phosphate-mannose-protein mannosyltransferase activity / regulation of endoplasmic reticulum unfolded protein response / fungal-type cell wall biogenesis / protein O-linked glycosylation via mannose / protein exit from endoplasmic reticulum / protein O-linked glycosylation ...dolichyl-phosphate-mannose-protein mannosyltransferase Pmt1p-Pmt3p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase Pmt1p-Pmt2p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase Pmt5p-Pmt2p dimer complex / dolichyl-phosphate-mannose-protein mannosyltransferase / dolichyl-phosphate-mannose-protein mannosyltransferase activity / regulation of endoplasmic reticulum unfolded protein response / fungal-type cell wall biogenesis / protein O-linked glycosylation via mannose / protein exit from endoplasmic reticulum / protein O-linked glycosylation / ERAD pathway / endoplasmic reticulum membrane / endoplasmic reticulum 類似検索 - 分子機能 | ||||||
| 生物種 |  | ||||||
| 手法 | 電子顕微鏡法 / 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 3.2 Å | ||||||
 データ登録者 データ登録者 | Bai, L. / Li, H. | ||||||
| 資金援助 |  米国, 1件 米国, 1件
| ||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2019 ジャーナル: Nat Struct Mol Biol / 年: 2019タイトル: Structure of the eukaryotic protein O-mannosyltransferase Pmt1-Pmt2 complex. 著者: Lin Bai / Amanda Kovach / Qinglong You / Alanna Kenny / Huilin Li /  要旨: In eukaryotes, a nascent peptide entering the endoplasmic reticulum (ER) is scanned by two Sec61 translocon-associated large membrane machines for protein N-glycosylation and protein O-mannosylation, ...In eukaryotes, a nascent peptide entering the endoplasmic reticulum (ER) is scanned by two Sec61 translocon-associated large membrane machines for protein N-glycosylation and protein O-mannosylation, respectively. While the structure of the eight-protein oligosaccharyltransferase complex has been determined recently, the structures of mannosyltransferases of the PMT family, which are an integral part of ER protein homeostasis, are still unknown. Here we report cryo-EM structures of the Saccharomyces cerevisiae Pmt1-Pmt2 complex bound to a donor and an acceptor peptide at 3.2-Å resolution, showing that each subunit contains 11 transmembrane helices and a lumenal β-trefoil fold termed the MIR domain. The structures reveal the substrate recognition model and confirm an inverting mannosyl-transferring reaction mechanism by the enzyme complex. Furthermore, we found that the transmembrane domains of Pmt1 and Pmt2 share a structural fold with the catalytic subunits of oligosaccharyltransferases, confirming a previously proposed evolutionary relationship between protein O-mannosylation and protein N-glycosylation. | ||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  6p25.cif.gz 6p25.cif.gz | 273.9 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb6p25.ent.gz pdb6p25.ent.gz | 214.9 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  6p25.json.gz 6p25.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| 文書・要旨 |  6p25_validation.pdf.gz 6p25_validation.pdf.gz | 1013.5 KB | 表示 |  wwPDB検証レポート wwPDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  6p25_full_validation.pdf.gz 6p25_full_validation.pdf.gz | 1 MB | 表示 | |
| XML形式データ |  6p25_validation.xml.gz 6p25_validation.xml.gz | 46.1 KB | 表示 | |
| CIF形式データ |  6p25_validation.cif.gz 6p25_validation.cif.gz | 67.7 KB | 表示 | |
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/p2/6p25 https://data.pdbj.org/pub/pdb/validation_reports/p2/6p25 ftp://data.pdbj.org/pub/pdb/validation_reports/p2/6p25 ftp://data.pdbj.org/pub/pdb/validation_reports/p2/6p25 | HTTPS FTP |
-関連構造データ
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
-Dolichyl-phosphate-mannose--protein mannosyltransferase ... , 2種, 2分子 AB
| #1: タンパク質 | 分子量: 92771.125 Da / 分子数: 1 / 由来タイプ: 組換発現 由来: (組換発現)  株: W303 / 遺伝子: PMT1, YDL095W, D2390 / 発現宿主:  参照: UniProt: P33775, dolichyl-phosphate-mannose-protein mannosyltransferase |
|---|---|
| #2: タンパク質 | 分子量: 86957.422 Da / 分子数: 1 / 由来タイプ: 組換発現 由来: (組換発現)  株: W303 / 遺伝子: PMT2, FUN25, YAL023C / 発現宿主:  参照: UniProt: P31382, dolichyl-phosphate-mannose-protein mannosyltransferase |
-タンパク質・ペプチド / 糖 , 2種, 3分子 D

| #3: タンパク質・ペプチド | 分子量: 478.538 Da / 分子数: 1 / 由来タイプ: 合成 由来: (合成)  |
|---|---|
| #4: 糖 |
-非ポリマー , 2種, 4分子 
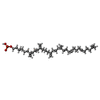

| #5: 化合物 | | #6: 化合物 | ChemComp-NNM / ( | |
|---|
-詳細
| Has protein modification | Y |
|---|
-実験情報
-実験
| 実験 | 手法: 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法: 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: Protein O-mannosyl transferase complex / タイプ: COMPLEX / Entity ID: #1-#3 / 由来: RECOMBINANT |
|---|---|
| 由来(天然) | 生物種:  |
| 由来(組換発現) | 生物種:  |
| 緩衝液 | pH: 7.4 |
| 試料 | 包埋: NO / シャドウイング: NO / 染色: NO / 凍結: YES |
| 試料支持 | 詳細: unspecified |
| 急速凍結 | 凍結剤: ETHANE |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源:  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD |
| 撮影 | 電子線照射量: 2 e/Å2 フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) |
- 解析
解析
| EMソフトウェア |
| ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CTF補正 | タイプ: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||||||||||||||||||||||
| 粒子像の選択 | 選択した粒子像数: 953292 | ||||||||||||||||||||||||||||||||||||||||
| 対称性 | 点対称性: C1 (非対称) | ||||||||||||||||||||||||||||||||||||||||
| 3次元再構成 | 解像度: 3.2 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 521848 / 対称性のタイプ: POINT | ||||||||||||||||||||||||||||||||||||||||
| 原子モデル構築 | プロトコル: FLEXIBLE FIT |
 ムービー
ムービー コントローラー
コントローラー
















 PDBj
PDBj


