[English] 日本語
 Yorodumi
Yorodumi- EMDB-5954: Protruding Knob-like Proteins Violate Local Symmetries in an Icos... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-5954 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Protruding Knob-like Proteins Violate Local Symmetries in an Icosahedral Marine Virus | |||||||||
 Map data Map data | Icosahedral cryo-EM reconstruction of Syn5 marine virus | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Syn5 / marine / cyanophage / capsid proteins / symmetry break | |||||||||
| Function / homology | : / Major capsid protein / Major capsid protein Function and homology information Function and homology information | |||||||||
| Biological species |  Synechococcus phage Syn5 (virus) Synechococcus phage Syn5 (virus) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 4.7 Å | |||||||||
 Authors Authors | Gipson P / Baker ML / Raytcheva D / Haase-Pettingell C / Piret J / King JA / Chiu W | |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2014 Journal: Nat Commun / Year: 2014Title: Protruding knob-like proteins violate local symmetries in an icosahedral marine virus. Authors: Preeti Gipson / Matthew L Baker / Desislava Raytcheva / Cameron Haase-Pettingell / Jacqueline Piret / Jonathan A King / Wah Chiu /  Abstract: Marine viruses play crucial roles in shaping the dynamics of oceanic microbial communities and in the carbon cycle on Earth. Here we report a 4.7-Å structure of a cyanobacterial virus, Syn5, by ...Marine viruses play crucial roles in shaping the dynamics of oceanic microbial communities and in the carbon cycle on Earth. Here we report a 4.7-Å structure of a cyanobacterial virus, Syn5, by electron cryo-microscopy and modelling. A Cα backbone trace of the major capsid protein (gp39) reveals a classic phage protein fold. In addition, two knob-like proteins protruding from the capsid surface are also observed. Using bioinformatics and structure analysis tools, these proteins are identified to correspond to gp55 and gp58 (each with two copies per asymmetric unit). The non 1:1 stoichiometric distribution of gp55/58 to gp39 breaks all expected local symmetries and leads to non-quasi-equivalence of the capsid subunits, suggesting a role in capsid stabilization. Such a structural arrangement has not yet been observed in any known virus structures. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_5954.map.gz emd_5954.map.gz | 68.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-5954-v30.xml emd-5954-v30.xml emd-5954.xml emd-5954.xml | 8.7 KB 8.7 KB | Display Display |  EMDB header EMDB header |
| Images |  400_5954.gif 400_5954.gif 80_5954.gif 80_5954.gif | 129.3 KB 6.5 KB | ||
| Filedesc structureFactors |  emd_5954_sf.cif.gz emd_5954_sf.cif.gz | 2.5 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-5954 http://ftp.pdbj.org/pub/emdb/structures/EMD-5954 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5954 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-5954 | HTTPS FTP |
-Related structure data
| Related structure data |  4bmlMC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_5954.map.gz / Format: CCP4 / Size: 599.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_5954.map.gz / Format: CCP4 / Size: 599.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Icosahedral cryo-EM reconstruction of Syn5 marine virus | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.32 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
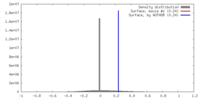
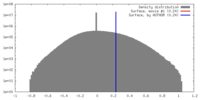
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Syn5 marine virus, also known as horned cyanophage Syn5
| Entire | Name: Syn5 marine virus, also known as horned cyanophage Syn5 |
|---|---|
| Components |
|
-Supramolecule #1000: Syn5 marine virus, also known as horned cyanophage Syn5
| Supramolecule | Name: Syn5 marine virus, also known as horned cyanophage Syn5 type: sample / ID: 1000 / Number unique components: 1 |
|---|
-Supramolecule #1: Synechococcus phage Syn5
| Supramolecule | Name: Synechococcus phage Syn5 / type: virus / ID: 1 / Name.synonym: Horned marine virus / NCBI-ID: 438482 / Sci species name: Synechococcus phage Syn5 / Sci species strain: Syn5 / Database: NCBI / Virus type: VIRION / Virus isolate: STRAIN / Virus enveloped: No / Virus empty: No / Syn species name: Horned marine virus |
|---|---|
| Host (natural) | Organism:  Synechococcus sp. WH 8109 (bacteria) / Strain: WH 8109 / synonym: ALGAE Synechococcus sp. WH 8109 (bacteria) / Strain: WH 8109 / synonym: ALGAE |
| Virus shell | Shell ID: 1 / Diameter: 655 Å / T number (triangulation number): 7 |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Grid | Details: R1.2/1.3 Quantifoil grids |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Instrument: FEI VITROBOT MARK IV / Method: 3 uL sample blotted once for 2.7 seconds |
- Electron microscopy
Electron microscopy
| Microscope | JEOL 3200FSC |
|---|---|
| Specialist optics | Energy filter - Name: Omega |
| Date | Oct 10, 2010 |
| Image recording | Category: CCD / Film or detector model: GATAN ULTRASCAN 10000 (10k x 10k) |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.7 µm / Nominal magnification: 80000 |
| Sample stage | Specimen holder model: JEOL 3200FSC CRYOHOLDER |
- Image processing
Image processing
| Final reconstruction | Algorithm: OTHER / Resolution.type: BY AUTHOR / Resolution: 4.7 Å / Resolution method: OTHER / Software - Name: MPSA / Number images used: 12000 |
|---|
 Movie
Movie Controller
Controller












 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)