+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Cryo-EM structure of the PP2A:B55-ARPP19 complex | ||||||||||||
 マップデータ マップデータ | Relion 3D auto refine full map. This map was used for Phenix real-space refinement of model coordinates and ADPs. | ||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | Protein Phosphatase 2A:B55 holoenzyme / ARPP19 inhibitor / cell cycle regulation / SIGNALING PROTEIN / HYDROLASE | ||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報phosphatase inhibitor activity / : / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors / regulation of microtubule binding / mitotic sister chromatid separation / MASTL Facilitates Mitotic Progression / protein serine/threonine phosphatase complex / regulation of meiotic cell cycle process involved in oocyte maturation ...phosphatase inhibitor activity / : / meiotic spindle elongation / Integration of energy metabolism / PP2A-mediated dephosphorylation of key metabolic factors / regulation of microtubule binding / mitotic sister chromatid separation / MASTL Facilitates Mitotic Progression / protein serine/threonine phosphatase complex / regulation of meiotic cell cycle process involved in oocyte maturation / protein phosphatase type 2A complex / meiotic sister chromatid cohesion, centromeric / peptidyl-serine dephosphorylation / peptidyl-threonine dephosphorylation / FAR/SIN/STRIPAK complex / positive regulation of microtubule binding / Regulation of glycolysis by fructose 2,6-bisphosphate metabolism / female meiotic nuclear division / Inhibition of replication initiation of damaged DNA by RB1/E2F1 / protein antigen binding / protein phosphatase regulator activity / GABA receptor binding / APC truncation mutants have impaired AXIN binding / AXIN missense mutants destabilize the destruction complex / Truncations of AMER1 destabilize the destruction complex / Initiation of Nuclear Envelope (NE) Reformation / ERKs are inactivated / positive regulation of extrinsic apoptotic signaling pathway in absence of ligand / Beta-catenin phosphorylation cascade / Signaling by GSK3beta mutants / CTNNB1 S33 mutants aren't phosphorylated / CTNNB1 S37 mutants aren't phosphorylated / CTNNB1 S45 mutants aren't phosphorylated / CTNNB1 T41 mutants aren't phosphorylated / regulation of growth / Disassembly of the destruction complex and recruitment of AXIN to the membrane / response to morphine / negative regulation of epithelial to mesenchymal transition / protein phosphatase inhibitor activity / negative regulation of glycolytic process through fructose-6-phosphate / positive regulation of NLRP3 inflammasome complex assembly / myosin phosphatase activity / protein serine/threonine phosphatase activity / Platelet sensitization by LDL / CTLA4 inhibitory signaling / protein-serine/threonine phosphatase / regulation of cell differentiation / ERK/MAPK targets / T cell homeostasis / regulation of G1/S transition of mitotic cell cycle / mesoderm development / phosphoprotein phosphatase activity / DARPP-32 events / negative regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction / chromosome, centromeric region / lateral plasma membrane / potassium channel regulator activity / negative regulation of hippo signaling / Nonsense Mediated Decay (NMD) enhanced by the Exon Junction Complex (EJC) / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Cyclin A/B1/B2 associated events during G2/M transition / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of mitotic centrosome proteins and complexes / positive regulation of gluconeogenesis / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / Resolution of Sister Chromatid Cohesion / protein dephosphorylation / AURKA Activation by TPX2 / protein tyrosine phosphatase activity / protein phosphatase 2A binding / meiotic cell cycle / chromosome segregation / positive regulation of D-glucose import / RHO GTPases Activate Formins / response to lead ion / RAF activation / regulation of protein phosphorylation / Spry regulation of FGF signaling / positive regulation of protein serine/threonine kinase activity / Degradation of beta-catenin by the destruction complex / tau protein binding / PKR-mediated signaling / spindle pole / Negative regulation of MAPK pathway / Separation of Sister Chromatids / Cyclin D associated events in G1 / G2/M transition of mitotic cell cycle / microtubule cytoskeleton / Regulation of PLK1 Activity at G2/M Transition / Regulation of TP53 Degradation / mitotic cell cycle / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling / protein-containing complex assembly / intracellular signal transduction / neuron projection / protein heterodimerization activity 類似検索 - 分子機能 | ||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||||||||
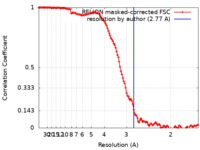
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.77 Å | ||||||||||||
 データ登録者 データ登録者 | Fuller JR / Padi SKR / Peti W / Page R | ||||||||||||
| 資金援助 |  米国, 3件 米国, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nature / 年: 2024 ジャーナル: Nature / 年: 2024タイトル: Cryo-EM structures of PP2A:B55-FAM122A and PP2A:B55-ARPP19. 著者: Sathish K R Padi / Margaret R Vos / Rachel J Godek / James R Fuller / Thomas Kruse / Jamin B Hein / Jakob Nilsson / Matthew S Kelker / Rebecca Page / Wolfgang Peti /   要旨: Progression through the cell cycle is controlled by regulated and abrupt changes in phosphorylation. Mitotic entry is initiated by increased phosphorylation of mitotic proteins, a process driven by ...Progression through the cell cycle is controlled by regulated and abrupt changes in phosphorylation. Mitotic entry is initiated by increased phosphorylation of mitotic proteins, a process driven by kinases, whereas mitotic exit is achieved by counteracting dephosphorylation, a process driven by phosphatases, especially PP2A:B55. Although the role of kinases in mitotic entry is well established, recent data have shown that mitosis is only successfully initiated when the counterbalancing phosphatases are also inhibited. Inhibition of PP2A:B55 is achieved by the intrinsically disordered proteins ARPP19 and FAM122A. Despite their critical roles in mitosis, the mechanisms by which they achieve PP2A:B55 inhibition is unknown. Here, we report the single-particle cryo-electron microscopy structures of PP2A:B55 bound to phosphorylated ARPP19 and FAM122A. Consistent with our complementary NMR spectroscopy studies, both intrinsically disordered proteins bind PP2A:B55, but do so in highly distinct manners, leveraging multiple distinct binding sites on B55. Our extensive structural, biophysical and biochemical data explain how substrates and inhibitors are recruited to PP2A:B55 and provide a molecular roadmap for the development of therapeutic interventions for PP2A:B55-related diseases. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_41604.map.gz emd_41604.map.gz | 131.2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-41604-v30.xml emd-41604-v30.xml emd-41604.xml emd-41604.xml | 27 KB 27 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_41604_fsc.xml emd_41604_fsc.xml | 12.5 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_41604.png emd_41604.png | 167.1 KB | ||
| マスクデータ |  emd_41604_msk_1.map emd_41604_msk_1.map | 166.4 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-41604.cif.gz emd-41604.cif.gz | 7.8 KB | ||
| その他 |  emd_41604_additional_1.map.gz emd_41604_additional_1.map.gz emd_41604_half_map_1.map.gz emd_41604_half_map_1.map.gz emd_41604_half_map_2.map.gz emd_41604_half_map_2.map.gz | 101 MB 131.5 MB 131.5 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-41604 http://ftp.pdbj.org/pub/emdb/structures/EMD-41604 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41604 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-41604 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_41604_validation.pdf.gz emd_41604_validation.pdf.gz | 929.3 KB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_41604_full_validation.pdf.gz emd_41604_full_validation.pdf.gz | 928.9 KB | 表示 | |
| XML形式データ |  emd_41604_validation.xml.gz emd_41604_validation.xml.gz | 19.9 KB | 表示 | |
| CIF形式データ |  emd_41604_validation.cif.gz emd_41604_validation.cif.gz | 26.3 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41604 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41604 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41604 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-41604 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_41604.map.gz / 形式: CCP4 / 大きさ: 166.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_41604.map.gz / 形式: CCP4 / 大きさ: 166.4 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Relion 3D auto refine full map. This map was used for Phenix real-space refinement of model coordinates and ADPs. | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.827 Å | ||||||||||||||||||||||||||||||||||||
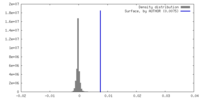
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
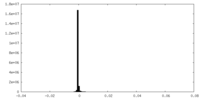
-マスク #1
| ファイル |  emd_41604_msk_1.map emd_41604_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 投影像・断面図 |
| ||||||||||||
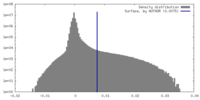
| 密度ヒストグラム |
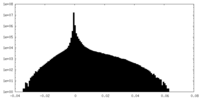
-追加マップ: Map version that was globally sharpened by a...
| ファイル | emd_41604_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Map version that was globally sharpened by a B-factor of -40.0 and filtered to local resolution by the implementation in Relion. This was used to guide manual model building. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
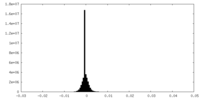
-ハーフマップ: Relion 3D auto refine half map 1
| ファイル | emd_41604_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Relion 3D auto refine half map 1 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
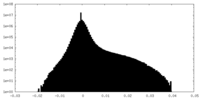
-ハーフマップ: Relion 3D auto refine half map 2
| ファイル | emd_41604_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Relion 3D auto refine half map 2 | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Quadruple complex of PP2A:B55 (PP2Aa:PP2Ac:B55) bound to thiophos...
| 全体 | 名称: Quadruple complex of PP2A:B55 (PP2Aa:PP2Ac:B55) bound to thiophosphorylated ARPP19 |
|---|---|
| 要素 |
|
-超分子 #1: Quadruple complex of PP2A:B55 (PP2Aa:PP2Ac:B55) bound to thiophos...
| 超分子 | 名称: Quadruple complex of PP2A:B55 (PP2Aa:PP2Ac:B55) bound to thiophosphorylated ARPP19 タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: #1-#4 |
|---|---|
| 分子量 | 理論値: 165 KDa |
-分子 #1: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit...
| 分子 | 名称: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform タイプ: protein_or_peptide / ID: 1 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 64.95798 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: GHMSLYPIAV LIDELRNEDV QLRLNSIKKL STIALALGVE RTRSELLPFL TDTIYDEDEV LLALAEQLGT FTTLVGGPEY VHCLLPPLE SLATVEETVV RDKAVESLRA ISHEHSPSDL EAHFVPLVKR LAGGDWFTSR TSACGLFSVC YPRVSSAVKA E LRQYFRNL ...文字列: GHMSLYPIAV LIDELRNEDV QLRLNSIKKL STIALALGVE RTRSELLPFL TDTIYDEDEV LLALAEQLGT FTTLVGGPEY VHCLLPPLE SLATVEETVV RDKAVESLRA ISHEHSPSDL EAHFVPLVKR LAGGDWFTSR TSACGLFSVC YPRVSSAVKA E LRQYFRNL CSDDTPMVRR AAASKLGEFA KVLELDNVKS EIIPMFSNLA SDEQDSVRLL AVEACVNIAQ LLPQEDLEAL VM PTLRQAA EDKSWRVRYM VADKFTELQK AVGPEITKTD LVPAFQNLMK DCEAEVRAAA SHKVKEFCEN LSADCRENVI MSQ ILPCIK ELVSDANQHV KSALASVIMG LSPILGKDNT IEHLLPLFLA QLKDECPEVR LNIISNLDCV NEVIGIRQLS QSLL PAIVE LAEDAKWRVR LAIIEYMPLL AGQLGVEFFD EKLNSLCMAW LVDHVYAIRE AATSNLKKLV EKFGKEWAHA TIIPK VLAM SGDPNYLHRM TTLFCINVLS EVCGQDITTK HMLPTVLRMA GDPVANVRFN VAKSLQKIGP ILDNSTLQSE VKPILE KLT QDQDVDVKYF AQEALTVLSL A UniProtKB: Serine/threonine-protein phosphatase 2A 65 kDa regulatory subunit A alpha isoform |
-分子 #2: Serine/threonine-protein phosphatase 2A 55 kDa regulatory subunit...
| 分子 | 名称: Serine/threonine-protein phosphatase 2A 55 kDa regulatory subunit B alpha isoform タイプ: protein_or_peptide / ID: 2 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 52.044289 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: HMGSAGAGGG NDIQWCFSQV KGAVDDDVAE ADIISTVEFN HSGELLATGD KGGRVVIFQQ EQENKIQSHS RGEYNVYSTF QSHEPEFDY LKSLEIEEKI NKIRWLPQKN AAQFLLSTND KTIKLWKISE RDKRPEGYNL KEEDGRYRDP TTVTTLRVPV F RPMDLMVE ...文字列: HMGSAGAGGG NDIQWCFSQV KGAVDDDVAE ADIISTVEFN HSGELLATGD KGGRVVIFQQ EQENKIQSHS RGEYNVYSTF QSHEPEFDY LKSLEIEEKI NKIRWLPQKN AAQFLLSTND KTIKLWKISE RDKRPEGYNL KEEDGRYRDP TTVTTLRVPV F RPMDLMVE ASPRRIFANA HTYHINSISI NSDYETYLSA DDLRINLWHL EITDRSFNIV DIKPANMEEL TEVITAAEFH PN SCNTFVY SSSKGTIRLC DMRASALCDR HSKLFEEPED PSNRSFFSEI ISSISDVKFS HSGRYMMTRD YLSVKIWDLN MEN RPVETY QVHEYLRSKL CSLYENDCIF DKFECCWNGS DSVVMTGSYN NFFRMFDRNT KRDITLEASR ENNKPRTVLK PRKV CASGK RKKDEISVDS LDFNKKILHT AWHPKENIIA VATTNNLYIF QDKVN UniProtKB: Serine/threonine-protein phosphatase 2A 55 kDa regulatory subunit B alpha isoform |
-分子 #3: Serine/threonine-protein phosphatase 2A catalytic subunit alpha i...
| 分子 | 名称: Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoform タイプ: protein_or_peptide / ID: 3 / コピー数: 1 / 光学異性体: LEVO / EC番号: protein-serine/threonine phosphatase |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 35.845375 KDa |
| 組換発現 | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 配列 | 文字列: GHMDEKVFTK ELDQWIEQLN ECKQLSESQV KSLCEKAKEI LTKESNVQEV RCPVTVCGDV HGQFHDLMEL FRIGGKSPDT NYLFMGDYV DRGYYSVETV TLLVALKVRY RERITILRGN HESRQITQVY GFYDECLRKY GNANVWKYFT DLFDYLPLTA L VDGQIFCL ...文字列: GHMDEKVFTK ELDQWIEQLN ECKQLSESQV KSLCEKAKEI LTKESNVQEV RCPVTVCGDV HGQFHDLMEL FRIGGKSPDT NYLFMGDYV DRGYYSVETV TLLVALKVRY RERITILRGN HESRQITQVY GFYDECLRKY GNANVWKYFT DLFDYLPLTA L VDGQIFCL HGGLSPSIDT LDHIRALDRL QEVPHEGPMC DLLWSDPDDR GGWGISPRGA GYTFGQDISE TFNHANGLTL VS RAHQLVM EGYNWCHDRN VVTIFSAPNY CYRCGNQAAI MELDDTLKYS FLQFDPAPRR GEPHVTRRTP DYF(MLL) UniProtKB: Serine/threonine-protein phosphatase 2A catalytic subunit alpha isoform |
-分子 #4: cAMP-regulated phosphoprotein 19
| 分子 | 名称: cAMP-regulated phosphoprotein 19 / タイプ: protein_or_peptide / ID: 4 / コピー数: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 12.620241 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: GHMSAEVPEA ASAEEQKEME DKVTSPEKAE EAKLKARYPH LGQKPGGSDF LRKRLQKGQK YFD(2RX)GDYNMA KAKMKN KQL PTAAPDKTEV TGDHIPTPQD LPQRKPALVA SKLAG UniProtKB: cAMP-regulated phosphoprotein 19 |
-分子 #5: FE (III) ION
| 分子 | 名称: FE (III) ION / タイプ: ligand / ID: 5 / コピー数: 1 / 式: FE |
|---|---|
| 分子量 | 理論値: 55.845 Da |
-分子 #6: ZINC ION
| 分子 | 名称: ZINC ION / タイプ: ligand / ID: 6 / コピー数: 1 / 式: ZN |
|---|---|
| 分子量 | 理論値: 65.409 Da |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 1.2 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 8 構成要素:
詳細: CHAPSO was added only immediately prior to vitrification | ||||||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 291 K / 装置: FEI VITROBOT MARK IV |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 特殊光学系 | エネルギーフィルター - 名称: GIF Bioquantum / エネルギーフィルター - スリット幅: 20 eV |
| 撮影 | フィルム・検出器のモデル: GATAN K3 BIOQUANTUM (6k x 4k) 撮影したグリッド数: 1 / 平均電子線量: 70.0 e/Å2 詳細: Camera was operated in CDS mode, with hardware binning of super-resolution pixels, writing movies with 62 frames |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 50.0 µm / 最大 デフォーカス(補正後): 2.6 µm / 最小 デフォーカス(補正後): 0.39 µm / 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / Cs: 2.7 mm 最大 デフォーカス(公称値): 1.9000000000000001 µm 最小 デフォーカス(公称値): 0.7000000000000001 µm 倍率(公称値): 105000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
+ 画像解析
画像解析
-原子モデル構築 1
| 初期モデル |
| ||||||
|---|---|---|---|---|---|---|---|
| 詳細 | Iterating between manual refinement in Coot and automated real-space refinement in Phenix | ||||||
| 精密化 | 空間: REAL / プロトコル: FLEXIBLE FIT / 当てはまり具合の基準: Cross-correlation | ||||||
| 得られたモデル |  PDB-8ttb: |
 ムービー
ムービー コントローラー
コントローラー




























 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)