+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
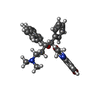
| タイトル | Structure of the human ATP synthase bound to bedaquiline (composite) | ||||||||||||
 マップデータ マップデータ | |||||||||||||
 試料 試料 |
| ||||||||||||
 キーワード キーワード | ATP synthase / Human / cryo-EM / Membrane protein | ||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報mitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / regulation of protein targeting to mitochondrion / negative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / positive regulation of proteolysis involved in protein catabolic process / Cristae formation / positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization / ATP biosynthetic process / angiostatin binding ...mitochondrial proton-transporting ATP synthase complex binding / regulation of ATP metabolic process / regulation of protein targeting to mitochondrion / negative regulation of cell adhesion involved in substrate-bound cell migration / Formation of ATP by chemiosmotic coupling / positive regulation of proteolysis involved in protein catabolic process / Cristae formation / positive regulation of autophagy of mitochondrion in response to mitochondrial depolarization / ATP biosynthetic process / angiostatin binding / positive regulation of mitochondrial outer membrane permeabilization involved in apoptotic signaling pathway / mitochondrial depolarization / Mitochondrial protein import / ATPase inhibitor activity / negative regulation of ATP-dependent activity / mitochondrial proton-transporting ATP synthase complex assembly / negative regulation of hydrolase activity / proton-transporting ATP synthase complex / oxidative phosphorylation / cellular response to interleukin-7 / enzyme inhibitor activity / response to copper ion / response to muscle activity / cellular response to nitric oxide / heme biosynthetic process / : / : / : / : / proton-transporting ATP synthase complex, coupling factor F(o) / mitochondrial nucleoid / : / MHC class I protein binding / negative regulation of endothelial cell proliferation / proton transmembrane transporter activity / proton motive force-driven ATP synthesis / positive regulation of blood vessel endothelial cell migration / response to hyperoxia / generation of precursor metabolites and energy / proton-transporting ATP synthase complex, catalytic core F(1) / cellular response to dexamethasone stimulus / H+-transporting two-sector ATPase / substantia nigra development / proton-transporting ATPase activity, rotational mechanism / proton transmembrane transport / reactive oxygen species metabolic process / proton-transporting ATP synthase activity, rotational mechanism / erythrocyte differentiation / proton motive force-driven mitochondrial ATP synthesis / Mitochondrial protein degradation / lipid metabolic process / regulation of intracellular pH / Transcriptional activation of mitochondrial biogenesis / mitochondrial membrane / aerobic respiration / ADP binding / osteoblast differentiation / ATPase binding / nuclear membrane / protease binding / angiogenesis / response to ethanol / calmodulin binding / mitochondrial inner membrane / mitochondrial matrix / hydrolase activity / membrane raft / lipid binding / enzyme binding / cell surface / ATP hydrolysis activity / protein-containing complex / mitochondrion / RNA binding / extracellular exosome / ATP binding / identical protein binding / membrane / nucleus / plasma membrane 類似検索 - 分子機能 | ||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||||||||
| 手法 | 単粒子再構成法 / クライオ電子顕微鏡法 / 解像度: 2.89 Å | ||||||||||||
 データ登録者 データ登録者 | Lai Y / Zhang Y / Gong H | ||||||||||||
| 資金援助 |  中国, 3件 中国, 3件
| ||||||||||||
 引用 引用 |  ジャーナル: Nature / 年: 2024 ジャーナル: Nature / 年: 2024タイトル: Inhibition of M. tuberculosis and human ATP synthase by BDQ and TBAJ-587. 著者: Yuying Zhang / Yuezheng Lai / Shan Zhou / Ting Ran / Yue Zhang / Ziqing Zhao / Ziyan Feng / Long Yu / Jinxu Xu / Kun Shi / Jianyun Wang / Yu Pang / Liang Li / Hongming Chen / Luke W Guddat / ...著者: Yuying Zhang / Yuezheng Lai / Shan Zhou / Ting Ran / Yue Zhang / Ziqing Zhao / Ziyan Feng / Long Yu / Jinxu Xu / Kun Shi / Jianyun Wang / Yu Pang / Liang Li / Hongming Chen / Luke W Guddat / Yan Gao / Fengjiang Liu / Zihe Rao / Hongri Gong /   要旨: Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation of Mycobacterium tuberculosis by inhibiting ATP synthase. ...Bedaquiline (BDQ), a first-in-class diarylquinoline anti-tuberculosis drug, and its analogue, TBAJ-587, prevent the growth and proliferation of Mycobacterium tuberculosis by inhibiting ATP synthase. However, BDQ also inhibits human ATP synthase. At present, how these compounds interact with either M. tuberculosis ATP synthase or human ATP synthase is unclear. Here we present cryogenic electron microscopy structures of M. tuberculosis ATP synthase with and without BDQ and TBAJ-587 bound, and human ATP synthase bound to BDQ. The two inhibitors interact with subunit a and the c-ring at the leading site, c-only sites and lagging site in M. tuberculosis ATP synthase, showing that BDQ and TBAJ-587 have similar modes of action. The quinolinyl and dimethylamino units of the compounds make extensive contacts with the protein. The structure of human ATP synthase in complex with BDQ reveals that the BDQ-binding site is similar to that observed for the leading site in M. tuberculosis ATP synthase, and that the quinolinyl unit also interacts extensively with the human enzyme. This study will improve researchers' understanding of the similarities and differences between human ATP synthase and M. tuberculosis ATP synthase in terms of the mode of BDQ binding, and will allow the rational design of novel diarylquinolines as anti-tuberculosis drugs. | ||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_37251.map.gz emd_37251.map.gz | 468.4 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-37251-v30.xml emd-37251-v30.xml emd-37251.xml emd-37251.xml | 28.5 KB 28.5 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_37251.png emd_37251.png | 41.8 KB | ||
| Filedesc metadata |  emd-37251.cif.gz emd-37251.cif.gz | 7.9 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37251 http://ftp.pdbj.org/pub/emdb/structures/EMD-37251 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37251 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37251 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  8ki3MC  8j0sC  8j0tC 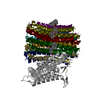 8j57C  8j58C  8jr0C  8jr1C  8khfC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_37251.map.gz / 形式: CCP4 / 大きさ: 512 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_37251.map.gz / 形式: CCP4 / 大きさ: 512 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ボクセルのサイズ | X=Y=Z: 0.73 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
- 試料の構成要素
試料の構成要素
+全体 : human ATP synthase
+超分子 #1: human ATP synthase
+分子 #1: ATP synthase subunit alpha, mitochondrial
+分子 #2: ATP synthase subunit beta, mitochondrial
+分子 #3: ATP synthase subunit gamma, mitochondrial
+分子 #4: ATPase inhibitor, mitochondrial
+分子 #5: ATP synthase subunit O, mitochondrial
+分子 #6: ATP synthase F(0) complex subunit C1, mitochondrial
+分子 #7: ATP synthase subunit delta, mitochondrial
+分子 #8: ATP synthase subunit epsilon, mitochondrial
+分子 #9: ATP synthase F(0) complex subunit B1, mitochondrial
+分子 #10: ATP synthase subunit d, mitochondrial
+分子 #11: ATP synthase subunit a
+分子 #12: ATP synthase subunit ATP5MJ, mitochondrial
+分子 #13: ATP synthase protein 8
+分子 #14: ATP synthase subunit f, mitochondrial
+分子 #15: ATP synthase subunit g, mitochondrial
+分子 #16: ATP synthase subunit e, mitochondrial
+分子 #17: ATP synthase-coupling factor 6, mitochondrial
+分子 #18: ADENOSINE-5'-TRIPHOSPHATE
+分子 #19: MAGNESIUM ION
+分子 #20: ADENOSINE-5'-DIPHOSPHATE
+分子 #21: Bedaquiline
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 撮影 | フィルム・検出器のモデル: FEI FALCON IV (4k x 4k) 平均電子線量: 50.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD / 最大 デフォーカス(公称値): 2.4 µm / 最小 デフォーカス(公称値): 1.2 µm |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 初期モデル | モデルのタイプ: PDB ENTRY PDBモデル - PDB ID: |
|---|---|
| 最終 再構成 | 解像度のタイプ: BY AUTHOR / 解像度: 2.89 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 使用した粒子像数: 84037 |
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
 ムービー
ムービー コントローラー
コントローラー