[English] 日本語
 Yorodumi
Yorodumi- EMDB-24883: Human Nuclear Exosome Targeting (NEXT) complex bound to RNA (subs... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Human Nuclear Exosome Targeting (NEXT) complex bound to RNA (substrate 2) | |||||||||
 Map data Map data | NEXT-RNA subtrate 2 complex - composite reconstruction | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Helicase / ATPase / RNA / Exosome / RNA BINDING PROTEIN / RNA BINDING PROTEIN-RNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationsnRNA catabolic process / TRAMP complex / snRNA binding / mRNA 3'-end processing / RNA catabolic process / regulation of alternative mRNA splicing, via spliceosome / maturation of 5.8S rRNA / Major pathway of rRNA processing in the nucleolus and cytosol / RNA processing / pre-mRNA intronic binding ...snRNA catabolic process / TRAMP complex / snRNA binding / mRNA 3'-end processing / RNA catabolic process / regulation of alternative mRNA splicing, via spliceosome / maturation of 5.8S rRNA / Major pathway of rRNA processing in the nucleolus and cytosol / RNA processing / pre-mRNA intronic binding / 14-3-3 protein binding / catalytic step 2 spliceosome / Regulation of endogenous retroelements by the Human Silencing Hub (HUSH) complex / mRNA Splicing - Major Pathway / meiotic cell cycle / mRNA splicing, via spliceosome / rRNA processing / RNA helicase activity / single-stranded RNA binding / nuclear body / RNA helicase / nuclear speck / DNA damage response / nucleolus / ATP hydrolysis activity / RNA binding / zinc ion binding / nucleoplasm / ATP binding / nucleus Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) / synthetic construct (others) Homo sapiens (human) / synthetic construct (others) | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.62 Å | |||||||||
 Authors Authors | Puno MR / Lima CD | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Cell / Year: 2022 Journal: Cell / Year: 2022Title: Structural basis for RNA surveillance by the human nuclear exosome targeting (NEXT) complex. Authors: M Rhyan Puno / Christopher D Lima /  Abstract: RNA quality control relies on co-factors and adaptors to identify and prepare substrates for degradation by ribonucleases such as the 3' to 5' ribonucleolytic RNA exosome. Here, we determined ...RNA quality control relies on co-factors and adaptors to identify and prepare substrates for degradation by ribonucleases such as the 3' to 5' ribonucleolytic RNA exosome. Here, we determined cryogenic electron microscopy structures of human nuclear exosome targeting (NEXT) complexes bound to RNA that reveal mechanistic insights to substrate recognition and early steps that precede RNA handover to the exosome. The structures illuminate ZCCHC8 as a scaffold, mediating homodimerization while embracing the MTR4 helicase and flexibly anchoring RBM7 to the helicase core. All three subunits collaborate to bind the RNA, with RBM7 and ZCCHC8 surveying sequences upstream of the 3' end to facilitate RNA capture by MTR4. ZCCHC8 obscures MTR4 surfaces important for RNA binding and extrusion as well as MPP6-dependent recruitment and docking onto the RNA exosome core, interactions that contribute to RNA surveillance by coordinating RNA capture, translocation, and extrusion from the helicase to the exosome for decay. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_24883.map.gz emd_24883.map.gz | 14.1 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-24883-v30.xml emd-24883-v30.xml emd-24883.xml emd-24883.xml | 52.9 KB 52.9 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_24883.png emd_24883.png | 59.3 KB | ||
| Filedesc metadata |  emd-24883.cif.gz emd-24883.cif.gz | 7.9 KB | ||
| Others |  emd_24883_additional_1.map.gz emd_24883_additional_1.map.gz emd_24883_additional_10.map.gz emd_24883_additional_10.map.gz emd_24883_additional_11.map.gz emd_24883_additional_11.map.gz emd_24883_additional_12.map.gz emd_24883_additional_12.map.gz emd_24883_additional_13.map.gz emd_24883_additional_13.map.gz emd_24883_additional_14.map.gz emd_24883_additional_14.map.gz emd_24883_additional_15.map.gz emd_24883_additional_15.map.gz emd_24883_additional_16.map.gz emd_24883_additional_16.map.gz emd_24883_additional_17.map.gz emd_24883_additional_17.map.gz emd_24883_additional_18.map.gz emd_24883_additional_18.map.gz emd_24883_additional_2.map.gz emd_24883_additional_2.map.gz emd_24883_additional_3.map.gz emd_24883_additional_3.map.gz emd_24883_additional_4.map.gz emd_24883_additional_4.map.gz emd_24883_additional_5.map.gz emd_24883_additional_5.map.gz emd_24883_additional_6.map.gz emd_24883_additional_6.map.gz emd_24883_additional_7.map.gz emd_24883_additional_7.map.gz emd_24883_additional_8.map.gz emd_24883_additional_8.map.gz emd_24883_additional_9.map.gz emd_24883_additional_9.map.gz | 6.1 MB 65.3 MB 65.4 MB 65.4 MB 65.3 MB 65.4 MB 65.4 MB 65.4 MB 65.3 MB 65.4 MB 5.2 MB 5.1 MB 5.9 MB 5.6 MB 4.6 MB 65.4 MB 65.4 MB 65.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-24883 http://ftp.pdbj.org/pub/emdb/structures/EMD-24883 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24883 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-24883 | HTTPS FTP |
-Validation report
| Summary document |  emd_24883_validation.pdf.gz emd_24883_validation.pdf.gz | 358.6 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_24883_full_validation.pdf.gz emd_24883_full_validation.pdf.gz | 358.2 KB | Display | |
| Data in XML |  emd_24883_validation.xml.gz emd_24883_validation.xml.gz | 6.6 KB | Display | |
| Data in CIF |  emd_24883_validation.cif.gz emd_24883_validation.cif.gz | 7.5 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24883 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24883 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24883 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-24883 | HTTPS FTP |
-Related structure data
| Related structure data |  7s7cMC  7s7bC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_24883.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_24883.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | NEXT-RNA subtrate 2 complex - composite reconstruction | ||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.1 Å | ||||||||||||||||||||||||||||||||||||
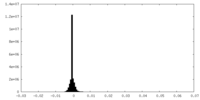
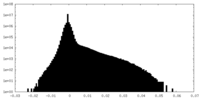
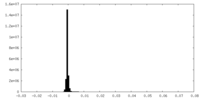
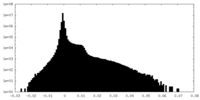
| Density |
| ||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
+Additional map: overall map
+Additional map: focused refinement map of RBM7-ZCCHC8 middle region-RNA - half map 2
+Additional map: overall reconstruction half map
+Additional map: focused refinement map of MTR4 core - half map 2
+Additional map: focused refinement map of RBM7-ZCCHC8 middle region-RNA - half map 1
+Additional map: focused refinement map of MTR4 core - half map 1
+Additional map: focused refinement map of MTR4-ZCCHC8 HD/KID - half map 1
+Additional map: focused refinement map of MTR4-ZCCHC8 HD/KID - half map 2
+Additional map: focused refinement map of MTR4 KOW-ZCCHC8 HD/KID region - half map 1
+Additional map: focused refinement map of MTR4 core-ZCCHC8 middle region-RBM7-RNA...
+Additional map: focused refinement map of MTR4 core - component...
+Additional map: focused refinement map of MTR4 core-ZCCHC8 middle region-RBM7-RNA...
+Additional map: focused refinement map of RBM7-ZCCHC8 middle region-RNA -...
+Additional map: focused refinement map of MTR4-ZCCHC8 HD/KID - component...
+Additional map: focused refinement map of MTR4 KOW-ZCCHC8 HD/KID -...
+Additional map: overall reconstruction half map 1
+Additional map: focused refinement map of MTR4 core-ZCCHC8 middle region-RBM7-RNA...
+Additional map: focused refinement map of MTR4 KOW-ZCCHC8 HD/KID - half map 2
- Sample components
Sample components
-Entire : human Nuclear Exosome Targeting (NEXT) complex bound to RNA (subs...
| Entire | Name: human Nuclear Exosome Targeting (NEXT) complex bound to RNA (substrate 2) |
|---|---|
| Components |
|
-Supramolecule #1: human Nuclear Exosome Targeting (NEXT) complex bound to RNA (subs...
| Supramolecule | Name: human Nuclear Exosome Targeting (NEXT) complex bound to RNA (substrate 2) type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#4 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: Exosome RNA helicase MTR4
| Macromolecule | Name: Exosome RNA helicase MTR4 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO / EC number: RNA helicase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 118.224961 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGDMADAFGD ELFSVFEGDS TTAAGTKKDK EKDKGKWKGP PGSADKAGKR FDGKLQSEST NNGKNKRDVD FEGTDEPIFG KKPRIEESI TEDLSLADLM PRVKVQSVET VEGCTHEVAL PAEEDYLPLK PRVGKAAKEY PFILDAFQRE AIQCVDNNQS V LVSAHTSA ...String: SGDMADAFGD ELFSVFEGDS TTAAGTKKDK EKDKGKWKGP PGSADKAGKR FDGKLQSEST NNGKNKRDVD FEGTDEPIFG KKPRIEESI TEDLSLADLM PRVKVQSVET VEGCTHEVAL PAEEDYLPLK PRVGKAAKEY PFILDAFQRE AIQCVDNNQS V LVSAHTSA GKTVCAEYAI ALALREKQRV IFTSPIKALS NQKYREMYEE FQDVGLMTGD VTINPTASCL VMTTEILRSM LY RGSEVMR EVAWVIFDEI HYMRDSERGV VWEETIILLP DNVHYVFLSA TIPNARQFAE WICHLHKQPC HVIYTDYRPT PLQ HYIFPA GGDGLHLVVD ENGDFREDNF NTAMQVLRDA GDLAKGDQKG RKGGTKGPSN VFKIVKMIME RNFQPVIIFS FSKK DCEAY ALQMTKLDFN TDEEKKMVEE VFSNAIDCLS DEDKKLPQVE HVLPLLKRGI GIHHGGLLPI LKETIEILFS EGLIK ALFA TETFAMGINM PARTVLFTNA RKFDGKDFRW ISSGEYIQMS GRAGRRGMDD RGIVILMVDE KMSPTIGKQL LKGSAD PLN SAFHLTYNMV LNLLRVEEIN PEYMLEKSFY QFQHYRAIPG VVEKVKNSEE QYNKIVIPNE ESVVIYYKIR QQLAKLG KE IEEYIHKPKY CLPFLQPGRL VKVKNEGDDF GWGVVVNFSK KSNVKPNSGE LDPLYVVEVL LRCSKESLKN SATEAAKP A KPDEKGEMQV VPVLVHLLSA ISSVRLYIPK DLRPVDNRQS VLKSIQEVQK RFPDGIPLLD PIDDMGIQDQ GLKKVIQKV EAFEHRMYSH PLHNDPNLET VYTLCEKKAQ IAIDIKSAKR ELKKARTVLQ MDELKCRKRV LRRLGFATSS DVIEMKGRVA CEISSADEL LLTEMMFNGL FNDLSAEQAT ALLSCFVFQE NSSEMPKLTE QLAGPLRQMQ ECAKRIAKVS AEAKLEIDEE T YLSSFKPH LMDVVYTWAT GATFAHICKM TDVFEGSIIR CMRRLEELLR QMCQAAKAIG NTELENKFAE GITKIKRDIV FA ASLYL UniProtKB: Exosome RNA helicase MTR4 |
-Macromolecule #2: Zinc finger CCHC domain-containing protein 8
| Macromolecule | Name: Zinc finger CCHC domain-containing protein 8 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 69.259805 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGDMAAEVYF GDLELFEPFD HPEESIPKPV HTRFKDDDGD EEDENGVGDA ELRERLRQCE ETIEQLRAEN QELKRKLNIL TRPSGILVN DTKLDGPILQ ILFMNNAISK QYHQEIEEFV SNLVKRFEEQ QKNDVEKTSF NLLPQPSSIV LEEDHKVEES C AIKNNKEA ...String: SGDMAAEVYF GDLELFEPFD HPEESIPKPV HTRFKDDDGD EEDENGVGDA ELRERLRQCE ETIEQLRAEN QELKRKLNIL TRPSGILVN DTKLDGPILQ ILFMNNAISK QYHQEIEEFV SNLVKRFEEQ QKNDVEKTSF NLLPQPSSIV LEEDHKVEES C AIKNNKEA FSVVGSVLYF TNFCLDKLGQ PLLNENPQLS EGWEIPKYHQ VFSHIVSLEG QEIQVKAKRP KPHCFNCGSE EH QMKDCPM PRNAARISEK RKEYMDACGE ANNQNFQQRY HAEEVEERFG RFKPGVISEE LQDALGVTDK SLPPFIYRMR QLG YPPGWL KEAELENSGL ALYDGKDGTD GETEVGEIQQ NKSVTYDLSK LVNYPGFNIS TPRGIPDEWR IFGSIPMQAC QQKD VFANY LTSNFQAPGV KSGGAVDEDA LTLEELEEQQ RRIWAALEQA ESVNSDSDVP VDTPLTGNSV ASSPCPNELD LPVPE GKTS EKQTLDEPEV PEIFTKKSEA GHASSPDSEV TSLCQKEKAE LAPVNTEGAL LDNGSVVPNC DISNGGSQKL FPADTS PST ATKIHSPIPD MSKFATGITP FEFENMAEST GMYLRIRSLL KNSPRNQQKN KKASE UniProtKB: Zinc finger CCHC domain-containing protein 8, Zinc finger CCHC domain-containing protein 8 |
-Macromolecule #3: RNA-binding protein 7
| Macromolecule | Name: RNA-binding protein 7 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 9.462986 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: SGDEADRTLF VGNLETKVTE ELLFELFHQA GPVIKVKIPK DKDGKPKQFA FVNFKHEVSV PYAMNLLNGI KLYGRPIKIQ FRS UniProtKB: RNA-binding protein 7 |
-Macromolecule #4: RNA (30-MER)
| Macromolecule | Name: RNA (30-MER) / type: rna / ID: 4 / Number of copies: 2 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 9.017486 KDa |
| Sequence | String: GGCGCGCGCC AAAAAUUUUU AAAAAAAA |
-Macromolecule #5: ZINC ION
| Macromolecule | Name: ZINC ION / type: ligand / ID: 5 / Number of copies: 1 / Formula: ZN |
|---|---|
| Molecular weight | Theoretical: 65.409 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 8 |
|---|---|
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 295 K / Instrument: FEI VITROBOT MARK IV / Details: 30 s wait time, blot for 2.5 s before plunging. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | #0 - Image recording ID: 1 / #0 - Film or detector model: GATAN K2 SUMMIT (4k x 4k) / #0 - Detector mode: SUPER-RESOLUTION / #0 - Average electron dose: 77.5 e/Å2 / #0 - Details: Dataset 1 / #1 - Image recording ID: 2 / #1 - Film or detector model: GATAN K3 (6k x 4k) / #1 - Average electron dose: 66.0 e/Å2 / #1 - Details: Datasets 2 and 3 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 X (Sec.)
X (Sec.) Y (Row.)
Y (Row.) Z (Col.)
Z (Col.)