+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-23815 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Dimeric (BRAF)2:(14-3-3)2 complex bound to SB590885 Inhibitor | |||||||||
 Map data Map data | Dimeric (B-Raf)2:(14-3-3)2 complex bound to SB590885 Inhibitor | |||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationGolgi reassembly / synaptic target recognition / respiratory system process / NOTCH4 Activation and Transmission of Signal to the Nucleus / regulation of synapse maturation / CD4-positive, alpha-beta T cell differentiation / trehalose metabolism in response to stress / CD4-positive or CD8-positive, alpha-beta T cell lineage commitment / establishment of Golgi localization / negative regulation of synaptic vesicle exocytosis ...Golgi reassembly / synaptic target recognition / respiratory system process / NOTCH4 Activation and Transmission of Signal to the Nucleus / regulation of synapse maturation / CD4-positive, alpha-beta T cell differentiation / trehalose metabolism in response to stress / CD4-positive or CD8-positive, alpha-beta T cell lineage commitment / establishment of Golgi localization / negative regulation of synaptic vesicle exocytosis / Signalling to p38 via RIT and RIN / head morphogenesis / myeloid progenitor cell differentiation / tube formation / ARMS-mediated activation / SHOC2 M1731 mutant abolishes MRAS complex function / Gain-of-function MRAS complexes activate RAF signaling / endothelial cell apoptotic process / Rap1 signalling / negative regulation of fibroblast migration / positive regulation of glucose transmembrane transport / establishment of protein localization to membrane / negative regulation of protein localization to nucleus / mitogen-activated protein kinase kinase binding / regulation of T cell differentiation / Negative feedback regulation of MAPK pathway / KSRP (KHSRP) binds and destabilizes mRNA / positive regulation of axonogenesis / Frs2-mediated activation / GP1b-IX-V activation signalling / stress fiber assembly / positive regulation of axon regeneration / face development / synaptic vesicle exocytosis / somatic stem cell population maintenance / MAP kinase kinase activity / thyroid gland development / Regulation of localization of FOXO transcription factors / Interleukin-3, Interleukin-5 and GM-CSF signaling / phosphoserine residue binding / MAP kinase kinase kinase activity / Activation of BAD and translocation to mitochondria / protein targeting / SARS-CoV-2 targets host intracellular signalling and regulatory pathways / cellular response to glucose starvation / Chk1/Chk2(Cds1) mediated inactivation of Cyclin B:Cdk1 complex / negative regulation of endothelial cell apoptotic process / SARS-CoV-1 targets host intracellular signalling and regulatory pathways / positive regulation of substrate adhesion-dependent cell spreading / RHO GTPases activate PKNs / positive regulation of stress fiber assembly / negative regulation of TORC1 signaling / response to cAMP / protein sequestering activity / cellular response to calcium ion / ERK1 and ERK2 cascade / negative regulation of innate immune response / hippocampal mossy fiber to CA3 synapse / substrate adhesion-dependent cell spreading / regulation of ERK1 and ERK2 cascade / cellular response to nerve growth factor stimulus / thymus development / Translocation of SLC2A4 (GLUT4) to the plasma membrane / Deactivation of the beta-catenin transactivating complex / long-term synaptic potentiation / TP53 Regulates Metabolic Genes / animal organ morphogenesis / Negative regulation of NOTCH4 signaling / RAF activation / Spry regulation of FGF signaling / lung development / Signaling by high-kinase activity BRAF mutants / visual learning / MAP2K and MAPK activation / regulation of protein stability / epidermal growth factor receptor signaling pathway / response to peptide hormone / Negative regulation of MAPK pathway / Signaling by RAF1 mutants / Signaling by moderate kinase activity BRAF mutants / Paradoxical activation of RAF signaling by kinase inactive BRAF / Signaling downstream of RAS mutants / MAPK cascade / Signaling by BRAF and RAF1 fusions / melanosome / cellular response to xenobiotic stimulus / presynapse / positive regulation of peptidyl-serine phosphorylation / T cell receptor signaling pathway / regulation of cell population proliferation / cell body / T cell differentiation in thymus / scaffold protein binding / angiogenesis / DNA-binding transcription factor binding / vesicle / transmembrane transporter binding / negative regulation of neuron apoptotic process / Ras protein signal transduction / positive regulation of ERK1 and ERK2 cascade Similarity search - Function | |||||||||
| Biological species |  Homo sapiens (human) Homo sapiens (human) | |||||||||
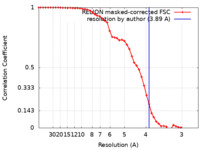
| Method | single particle reconstruction / cryo EM / Resolution: 3.89 Å | |||||||||
 Authors Authors | Martinez Fiesco JA / Ping Z / Durrant DE / Morrison DK | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2022 Journal: Nat Commun / Year: 2022Title: Structural insights into the BRAF monomer-to-dimer transition mediated by RAS binding. Authors: Juliana A Martinez Fiesco / David E Durrant / Deborah K Morrison / Ping Zhang /  Abstract: RAF kinases are essential effectors of RAS, but how RAS binding initiates the conformational changes needed for autoinhibited RAF monomers to form active dimers has remained unclear. Here, we present ...RAF kinases are essential effectors of RAS, but how RAS binding initiates the conformational changes needed for autoinhibited RAF monomers to form active dimers has remained unclear. Here, we present cryo-electron microscopy structures of full-length BRAF complexes derived from mammalian cells: autoinhibited, monomeric BRAF:14-3-3:MEK and BRAF:14-3-3 complexes, and an inhibitor-bound, dimeric BRAF:14-3-3 complex, at 3.7, 4.1, and 3.9 Å resolution, respectively. In both autoinhibited, monomeric structures, the RAS binding domain (RBD) of BRAF is resolved, revealing that the RBD forms an extensive contact interface with the 14-3-3 protomer bound to the BRAF C-terminal site and that key basic residues required for RBD-RAS binding are exposed. Moreover, through structure-guided mutational studies, our findings indicate that RAS-RAF binding is a dynamic process and that RBD residues at the center of the RBD:14-3-3 interface have a dual function, first contributing to RAF autoinhibition and then to the full spectrum of RAS-RBD interactions. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_23815.map.gz emd_23815.map.gz | 1.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-23815-v30.xml emd-23815-v30.xml emd-23815.xml emd-23815.xml | 12.4 KB 12.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_23815_fsc.xml emd_23815_fsc.xml | 4.7 KB | Display |  FSC data file FSC data file |
| Images |  emd_23815.png emd_23815.png | 67.1 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-23815 http://ftp.pdbj.org/pub/emdb/structures/EMD-23815 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23815 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-23815 | HTTPS FTP |
-Validation report
| Summary document |  emd_23815_validation.pdf.gz emd_23815_validation.pdf.gz | 381.1 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_23815_full_validation.pdf.gz emd_23815_full_validation.pdf.gz | 380.7 KB | Display | |
| Data in XML |  emd_23815_validation.xml.gz emd_23815_validation.xml.gz | 7.8 KB | Display | |
| Data in CIF |  emd_23815_validation.cif.gz emd_23815_validation.cif.gz | 9.9 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23815 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23815 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23815 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-23815 | HTTPS FTP |
-Related structure data
| Related structure data |  7mffMC  7mfdC  7mfeC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_23815.map.gz / Format: CCP4 / Size: 8.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_23815.map.gz / Format: CCP4 / Size: 8.4 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Dimeric (B-Raf)2:(14-3-3)2 complex bound to SB590885 Inhibitor | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.348 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Active dimeric (B-Raf)2:(14-3-3)2 complex
| Entire | Name: Active dimeric (B-Raf)2:(14-3-3)2 complex |
|---|---|
| Components |
|
-Supramolecule #1: Active dimeric (B-Raf)2:(14-3-3)2 complex
| Supramolecule | Name: Active dimeric (B-Raf)2:(14-3-3)2 complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: 14-3-3 protein zeta/delta
| Macromolecule | Name: 14-3-3 protein zeta/delta / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 27.777092 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MDKNELVQKA KLAEQAERYD DMAACMKSVT EQGAELSNEE RNLLSVAYKN VVGARRSSWR VVSSIEQKTE GAEKKQQMAR EYREKIETE LRDICNDVLS LLEKFLIPNA SQAESKVFYL KMKGDYYRYL AEVAAGDDKK GIVDQSQQAY QEAFEISKKE M QPTHPIRL ...String: MDKNELVQKA KLAEQAERYD DMAACMKSVT EQGAELSNEE RNLLSVAYKN VVGARRSSWR VVSSIEQKTE GAEKKQQMAR EYREKIETE LRDICNDVLS LLEKFLIPNA SQAESKVFYL KMKGDYYRYL AEVAAGDDKK GIVDQSQQAY QEAFEISKKE M QPTHPIRL GLALNFSVFY YEILNSPEKA CSLAKTAFDE AIAELDTLSE ESYKDSTLIM QLLRDNLTLW TSDTQGDEAE AG EGGEN |
-Macromolecule #2: Serine/threonine-protein kinase B-raf
| Macromolecule | Name: Serine/threonine-protein kinase B-raf / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO / EC number: non-specific serine/threonine protein kinase |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 84.697695 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MAALSGGGGG GAEPGQALFN GDMEPEAGAG AGAAASSAAD PAIPEEVWNI KQMIKLTQEH IEALLDKFGG EHNPPSIYLE AYEEYTSKL DALQQREQQL LESLGNGTDF SVSSSASMDT VTSSSSSSLS VLPSSLSVFQ NPTDVARSNP KSPQKPIVRV F LPNKQRTV ...String: MAALSGGGGG GAEPGQALFN GDMEPEAGAG AGAAASSAAD PAIPEEVWNI KQMIKLTQEH IEALLDKFGG EHNPPSIYLE AYEEYTSKL DALQQREQQL LESLGNGTDF SVSSSASMDT VTSSSSSSLS VLPSSLSVFQ NPTDVARSNP KSPQKPIVRV F LPNKQRTV VPARCGVTVR DSLKKALMMR GLIPECCAVY RIQDGEKKPI GWDTDISWLT GEELHVEVLE NVPLTTHNFV RK TFFTLAF CDFCRKLLFQ GFRCQTCGYK FHQRCSTEVP LMCVNYDQLD LLFVSKFFEH HPIPQEEASL AETALTSGSS PSA PASDSI GPQILTSPSP SKSIPIPQPF RPADEDHRNQ FGQRDRSS(SEP)A PNVHINTIEP VNIDDLIRDQ GFRGDGGSTT GLSATPPAS LPGSLTNVKA LQKSPGPQRE RKSSSSSEDR NRMKTLGRRD SSDDWEIPDG QITVGQRIGS GSFGTVYKGK W HGDVAVKM LNVTAPTPQQ LQAFKNEVGV LRKTRHVNIL LFMGYSTKPQ LAIVTQWCEG SSLYHHLHII ETKFEMIKLI DI ARQTAQG MDYLHAKSII HRDLKSNNIF LHEDLTVKIG DFGLATVKSR WSGSHQFEQL SGSILWMAPE VIRMQDKNPY SFQ SDVYAF GIVLYELMTG QLPYSNINNR DQIIFMVGRG YLSPDLSKVR SNCPKAMKRL MAECLKKKRD ERPLFPQILA SIEL LARSL PKIHRSA(SEP)EP SLNRAGFQTE DFSLYACASP KTPIQAGGYG AFPVH |
-Macromolecule #3: (1Z)-5-(2-{4-[2-(DIMETHYLAMINO)ETHOXY]PHENYL}-5-PYRIDIN-4-YL-1H-I...
| Macromolecule | Name: (1Z)-5-(2-{4-[2-(DIMETHYLAMINO)ETHOXY]PHENYL}-5-PYRIDIN-4-YL-1H-IMIDAZOL-4-YL)INDAN-1-ONE OXIME type: ligand / ID: 3 / Number of copies: 2 / Formula: 215 |
|---|---|
| Molecular weight | Theoretical: 453.536 Da |
| Chemical component information |  ChemComp-215: |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.2 mg/mL | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 8 Component:
| ||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 90 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: SUPER-RESOLUTION / Average electron dose: 57.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller