+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: EMDB / ID: EMD-20590 | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | CryoEM reconstruction of membrane-bound ESCRT-III filament composed of CHMP1B only | ||||||||||||||||||
 マップデータ マップデータ | RELION Refine3D filtered map with helical symmetry imposed on the central 40% | ||||||||||||||||||
 試料 試料 |
| ||||||||||||||||||
 キーワード キーワード | membrane remodeling / membrane-bound protein filament / ESCRT-III / LIPID BINDING PROTEIN | ||||||||||||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報MIT domain binding / amphisome membrane / multivesicular body-lysosome fusion / vesicle fusion with vacuole / ESCRT III complex disassembly / late endosome to lysosome transport / ESCRT III complex / kinetochore microtubule / endosome transport via multivesicular body sorting pathway / regulation of centrosome duplication ...MIT domain binding / amphisome membrane / multivesicular body-lysosome fusion / vesicle fusion with vacuole / ESCRT III complex disassembly / late endosome to lysosome transport / ESCRT III complex / kinetochore microtubule / endosome transport via multivesicular body sorting pathway / regulation of centrosome duplication / nuclear membrane reassembly / midbody abscission / multivesicular body sorting pathway / membrane coat / membrane fission / plasma membrane repair / late endosome to vacuole transport / multivesicular body membrane / ubiquitin-dependent protein catabolic process via the multivesicular body sorting pathway / multivesicular body assembly / regulation of mitotic spindle assembly / mitotic metaphase chromosome alignment / nucleus organization / viral budding via host ESCRT complex / autophagosome membrane / autophagosome maturation / nuclear pore / multivesicular body / viral budding from plasma membrane / establishment of protein localization / kinetochore / autophagy / protein transport / midbody / endosome membrane / protein domain specific binding / cell division / lysosomal membrane / extracellular exosome / nucleoplasm / identical protein binding / plasma membrane / cytosol 類似検索 - 分子機能 | ||||||||||||||||||
| 生物種 |  Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||||||||||||||
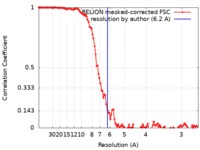
| 手法 | らせん対称体再構成法 / クライオ電子顕微鏡法 / 解像度: 6.2 Å | ||||||||||||||||||
 データ登録者 データ登録者 | Nguyen HC / Frost A | ||||||||||||||||||
| 資金援助 |  米国, 5件 米国, 5件
| ||||||||||||||||||
 引用 引用 |  ジャーナル: Nat Struct Mol Biol / 年: 2020 ジャーナル: Nat Struct Mol Biol / 年: 2020タイトル: Membrane constriction and thinning by sequential ESCRT-III polymerization. 著者: Henry C Nguyen / Nathaniel Talledge / John McCullough / Abhimanyu Sharma / Frank R Moss / Janet H Iwasa / Michael D Vershinin / Wesley I Sundquist / Adam Frost /  要旨: The endosomal sorting complexes required for transport (ESCRTs) mediate diverse membrane remodeling events. These typically require ESCRT-III proteins to stabilize negatively curved membranes; ...The endosomal sorting complexes required for transport (ESCRTs) mediate diverse membrane remodeling events. These typically require ESCRT-III proteins to stabilize negatively curved membranes; however, recent work has indicated that certain ESCRT-IIIs also participate in positive-curvature membrane-shaping reactions. ESCRT-IIIs polymerize into membrane-binding filaments, but the structural basis for negative versus positive membrane remodeling by these proteins remains poorly understood. To learn how certain ESCRT-IIIs shape positively curved membranes, we determined structures of human membrane-bound CHMP1B-only, membrane-bound CHMP1B + IST1, and IST1-only filaments by cryo-EM. Our structures show how CHMP1B first polymerizes into a single-stranded helical filament, shaping membranes into moderate-curvature tubules. Subsequently, IST1 assembles a second strand on CHMP1B, further constricting the membrane tube and reducing its diameter nearly to the fission point. Each step of constriction thins the underlying bilayer, lowering the barrier to membrane fission. Our structures reveal how a two-component, sequential polymerization mechanism drives membrane tubulation, constriction and bilayer thinning. | ||||||||||||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| ムービー |
 ムービービューア ムービービューア |
|---|---|
| 構造ビューア | EMマップ:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| 添付画像 |
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_20590.map.gz emd_20590.map.gz | 100.5 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-20590-v30.xml emd-20590-v30.xml emd-20590.xml emd-20590.xml | 25.5 KB 25.5 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| FSC (解像度算出) |  emd_20590_fsc.xml emd_20590_fsc.xml | 11.6 KB | 表示 |  FSCデータファイル FSCデータファイル |
| 画像 |  emd_20590.png emd_20590.png | 198.5 KB | ||
| マスクデータ |  emd_20590_msk_1.map emd_20590_msk_1.map | 129.7 MB |  マスクマップ マスクマップ | |
| Filedesc metadata |  emd-20590.cif.gz emd-20590.cif.gz | 6 KB | ||
| その他 |  emd_20590_additional_1.map.gz emd_20590_additional_1.map.gz emd_20590_additional_2.map.gz emd_20590_additional_2.map.gz emd_20590_additional_3.map.gz emd_20590_additional_3.map.gz emd_20590_additional_4.map.gz emd_20590_additional_4.map.gz emd_20590_half_map_1.map.gz emd_20590_half_map_1.map.gz emd_20590_half_map_2.map.gz emd_20590_half_map_2.map.gz | 35.2 MB 101.9 MB 102 MB 102 MB 101.4 MB 101.4 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-20590 http://ftp.pdbj.org/pub/emdb/structures/EMD-20590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20590 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-20590 | HTTPS FTP |
-検証レポート
| 文書・要旨 |  emd_20590_validation.pdf.gz emd_20590_validation.pdf.gz | 1 MB | 表示 |  EMDB検証レポート EMDB検証レポート |
|---|---|---|---|---|
| 文書・詳細版 |  emd_20590_full_validation.pdf.gz emd_20590_full_validation.pdf.gz | 1 MB | 表示 | |
| XML形式データ |  emd_20590_validation.xml.gz emd_20590_validation.xml.gz | 18.9 KB | 表示 | |
| CIF形式データ |  emd_20590_validation.cif.gz emd_20590_validation.cif.gz | 24.9 KB | 表示 | |
| アーカイブディレクトリ |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20590 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20590 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20590 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-20590 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  6tz9MC  6tz4C  6tz5C  6tzaC C: 同じ文献を引用 ( M: このマップから作成された原子モデル |
|---|---|
| 類似構造データ | |
| 電子顕微鏡画像生データ |  EMPIAR-10397 (タイトル: CryoEM reconstruction of membrane-bound ESCRT-III filament composed of CHMP1B only EMPIAR-10397 (タイトル: CryoEM reconstruction of membrane-bound ESCRT-III filament composed of CHMP1B onlyData size: 3.8 Data #1: Processed particle stack and metadata for a cryoEM helical reconstruction of membrane-bound ESCRT-III filament composed of CHMP1B only [picked particles - multiframe - processed]) |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_20590.map.gz / 形式: CCP4 / 大きさ: 129.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_20590.map.gz / 形式: CCP4 / 大きさ: 129.7 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D filtered map with helical symmetry imposed on the central 40% | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.345 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
CCP4マップ ヘッダ情報:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-添付データ
-マスク #1
| ファイル |  emd_20590_msk_1.map emd_20590_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
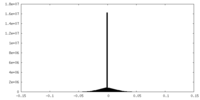
| 投影像・断面図 |
| ||||||||||||
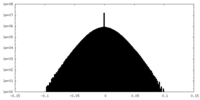
| 密度ヒストグラム |
-追加マップ: RELION postprocessed map with helical symmetry applied to...
| ファイル | emd_20590_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION postprocessed map with helical symmetry applied to the entire map | ||||||||||||
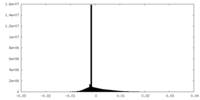
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: RELION Refine3D filtered map with no symmetry applied (C1 map)
| ファイル | emd_20590_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D filtered map with no symmetry applied (C1 map) | ||||||||||||
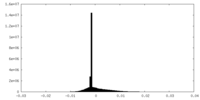
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: RELION Refine3D unfiltered half map1 with no symmetry...
| ファイル | emd_20590_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D unfiltered half map1 with no symmetry applied (C1 half1 map) | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-追加マップ: RELION Refine3D unfiltered half map2 with no symmetry...
| ファイル | emd_20590_additional_4.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D unfiltered half map2 with no symmetry applied (C1 half2 map) | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: RELION Refine3D unfiltered half map1 with helical symmetry...
| ファイル | emd_20590_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D unfiltered half map1 with helical symmetry imposed on the central 40% | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
-ハーフマップ: RELION Refine3D unfiltered half map2 with helical symmetry...
| ファイル | emd_20590_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | RELION Refine3D unfiltered half map2 with helical symmetry imposed on the central 40% | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : membrane-bound ESCRT-III copolymer filament composed of CHMP1B only
| 全体 | 名称: membrane-bound ESCRT-III copolymer filament composed of CHMP1B only |
|---|---|
| 要素 |
|
-超分子 #1: membrane-bound ESCRT-III copolymer filament composed of CHMP1B only
| 超分子 | 名称: membrane-bound ESCRT-III copolymer filament composed of CHMP1B only タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
-分子 #1: Charged multivesicular body protein 1b
| 分子 | 名称: Charged multivesicular body protein 1b / タイプ: protein_or_peptide / ID: 1 / コピー数: 26 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:  Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 分子量 | 理論値: 22.140354 KDa |
| 組換発現 | 生物種:  |
| 配列 | 文字列: MSNMEKHLFN LKFAAKELSR SAKKCDKEEK AEKAKIEKAI QKGNMEVARI HAENAIRQKN QAVNFLRMSA RVDAVAARVQ TAVTMGKVT KSMAGVVKSM DATLKTMNLE KISALMDKFE HQFETLDVQT QQMEDTMSST TTLTTPQNQV DMLLQEMADE A GLDLNMEL ...文字列: MSNMEKHLFN LKFAAKELSR SAKKCDKEEK AEKAKIEKAI QKGNMEVARI HAENAIRQKN QAVNFLRMSA RVDAVAARVQ TAVTMGKVT KSMAGVVKSM DATLKTMNLE KISALMDKFE HQFETLDVQT QQMEDTMSST TTLTTPQNQV DMLLQEMADE A GLDLNMEL PQGQTGSVGT SVASAEQDEL SQRLARLRDQ V UniProtKB: Charged multivesicular body protein 1b |
-実験情報
-構造解析
| 手法 | クライオ電子顕微鏡法 |
|---|---|
 解析 解析 | らせん対称体再構成法 |
| 試料の集合状態 | helical array |
- 試料調製
試料調製
| 緩衝液 | pH: 7.4 |
|---|---|
| グリッド | 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 時間: 30 sec. / 詳細: unspecified |
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 292 K / 装置: FEI VITROBOT MARK III 詳細: Grids were blotted with Whatman No. 1 filter paper for 4-8 seconds with a 0 mm offset at 19C and 100 percent humidity before plunging into liquid ethane. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 特殊光学系 | エネルギーフィルター - 名称: GIF Bioquantum / エネルギーフィルター - スリット幅: 20 eV |
| 撮影 | フィルム・検出器のモデル: GATAN K2 SUMMIT (4k x 4k) 検出モード: SUPER-RESOLUTION / 平均電子線量: 45.0 e/Å2 |
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
 ムービー
ムービー コントローラー
コントローラー













 Z
Z Y
Y X
X