[English] 日本語
 Yorodumi
Yorodumi- EMDB-17565: CryoEM structure of a GroEL14-GroES14 cage with two encapsulated ... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | CryoEM structure of a GroEL14-GroES14 cage with two encapsulated disordered MetK substrates in the presence of ADP-BeFx | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Chaperonin / Folding cage / proteostasis / heat shock / ATPase / CHAPERONE | |||||||||
| Function / homology |  Function and homology information Function and homology informationmethionine adenosyltransferase / methionine adenosyltransferase activity / S-adenosylmethionine cycle / S-adenosylmethionine biosynthetic process / GroEL-GroES complex / virion assembly / potassium ion binding / chaperone cofactor-dependent protein refolding / protein folding chaperone / unfolded protein binding ...methionine adenosyltransferase / methionine adenosyltransferase activity / S-adenosylmethionine cycle / S-adenosylmethionine biosynthetic process / GroEL-GroES complex / virion assembly / potassium ion binding / chaperone cofactor-dependent protein refolding / protein folding chaperone / unfolded protein binding / protein folding / one-carbon metabolic process / protein-folding chaperone binding / response to heat / magnesium ion binding / ATP binding / identical protein binding / metal ion binding / cytosol Similarity search - Function | |||||||||
| Biological species |   | |||||||||
| Method | single particle reconstruction / cryo EM / Resolution: 3.22 Å | |||||||||
 Authors Authors | Wagner J / Beck F / Bracher A / Caravajal AI / Wan W / Bohn S / Koerner R / Baumeister W / Fernandez-Busnadiego R / Hartl FU | |||||||||
| Funding support | 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2024 Journal: Nature / Year: 2024Title: Visualizing chaperonin function in situ by cryo-electron tomography Authors: Wagner J / Caravajal AI / Beck F / Bracher A / Wan W / Bohn S / Koerner R / Baumeister W / Fernandez-Busnadiego R / Hartl FU | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17565.map.gz emd_17565.map.gz | 197.8 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17565-v30.xml emd-17565-v30.xml emd-17565.xml emd-17565.xml | 22.9 KB 22.9 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17565_fsc.xml emd_17565_fsc.xml | 12.5 KB | Display |  FSC data file FSC data file |
| Images |  emd_17565.png emd_17565.png | 63.9 KB | ||
| Masks |  emd_17565_msk_1.map emd_17565_msk_1.map | 209.3 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17565.cif.gz emd-17565.cif.gz | 5.6 KB | ||
| Others |  emd_17565_half_map_1.map.gz emd_17565_half_map_1.map.gz emd_17565_half_map_2.map.gz emd_17565_half_map_2.map.gz | 194.5 MB 194.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17565 http://ftp.pdbj.org/pub/emdb/structures/EMD-17565 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17565 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17565 | HTTPS FTP |
-Validation report
| Summary document |  emd_17565_validation.pdf.gz emd_17565_validation.pdf.gz | 1.1 MB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_17565_full_validation.pdf.gz emd_17565_full_validation.pdf.gz | 1.1 MB | Display | |
| Data in XML |  emd_17565_validation.xml.gz emd_17565_validation.xml.gz | 21.5 KB | Display | |
| Data in CIF |  emd_17565_validation.cif.gz emd_17565_validation.cif.gz | 27.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17565 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17565 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17565 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-17565 | HTTPS FTP |
-Related structure data
| Related structure data |  8p4mC  8p4nC  8p4oC  8p4pC  8p4rC  8qxsC  8qxtC  8qxuC  8qxvC C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17565.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17565.map.gz / Format: CCP4 / Size: 209.3 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.07 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17565_msk_1.map emd_17565_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_17565_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_17565_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : GroEL14-GroES14-MetK
| Entire | Name: GroEL14-GroES14-MetK |
|---|---|
| Components |
|
-Supramolecule #1: GroEL14-GroES14-MetK
| Supramolecule | Name: GroEL14-GroES14-MetK / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all / Details: with bound ADP-BeF3 Mg2+ K+ |
|---|---|
| Source (natural) | Organism:  |
| Molecular weight | Theoretical: 1.03 MDa |
-Macromolecule #1: GroEL
| Macromolecule | Name: GroEL / type: protein_or_peptide / ID: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEIT RNTVREIGYV HSDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD Q GLMFGYAT NETDVLMPAP ITYAHRLVQR QAEVRKNGTL PWLRPDAKSQ ...String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEIT RNTVREIGYV HSDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD Q GLMFGYAT NETDVLMPAP ITYAHRLVQR QAEVRKNGTL PWLRPDAKSQ VTFQYDDGKI VG IDAVVLS TQHSEEIDQK SLQEAVMEEI IKPILPAEWL TSATKFFINP TGRFVIGGPM GDC GLTGRK IIVDTYGGMA RHGGGAFSGK DPSKVDRSAA YAARYVAKNI VAAGLADRCE IQVS YAIGV AEPTSIMVET FGTEKVPSEQ LTLLVREFFD LRPYGLIQML DLLHPIYKET AAYGH FGRE HFPWEKTDKA QLLRDAAGLK UniProtKB: S-adenosylmethionine synthase |
-Macromolecule #2: GroES
| Macromolecule | Name: GroES / type: protein_or_peptide / ID: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MNIRPLHDRV IVKRKEVETK SAGGIVLTGS AAAKSTRGEV LAVGNGRILE NGEVKPLDVK VGDIVIFND GYGVKSEKID NEEVLIMSES DILAIVEA UniProtKB: Co-chaperonin GroES |
-Macromolecule #3: MetK
| Macromolecule | Name: MetK / type: protein_or_peptide / ID: 3 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  |
| Recombinant expression | Organism:  |
| Sequence | String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEIT RNTVREIGYV HSDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD Q GLMFGYAT NETDVLMPAP ITYAHRLVQR QAEVRKNGTL PWLRPDAKSQ ...String: MAKHLFTSES VSEGHPDKIA DQISDAVLDA ILEQDPKARV ACETYVKTGM VLVGGEITTS AWVDIEEIT RNTVREIGYV HSDMGFDANS CAVLSAIGKQ SPDINQGVDR ADPLEQGAGD Q GLMFGYAT NETDVLMPAP ITYAHRLVQR QAEVRKNGTL PWLRPDAKSQ VTFQYDDGKI VG IDAVVLS TQHSEEIDQK SLQEAVMEEI IKPILPAEWL TSATKFFINP TGRFVIGGPM GDC GLTGRK IIVDTYGGMA RHGGGAFSGK DPSKVDRSAA YAARYVAKNI VAAGLADRCE IQVS YAIGV AEPTSIMVET FGTEKVPSEQ LTLLVREFFD LRPYGLIQML DLLHPIYKET AAYGH FGRE HFPWEKTDKA QLLRDAAGLK UniProtKB: S-adenosylmethionine synthase |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 10.3 mg/mL | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| ||||||||||||||||||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV Details: 5.9 mM n-octyl-beta-D-glucopyranoside were added before vitrification. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 55.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: OTHER / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 22500 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller








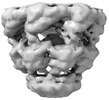






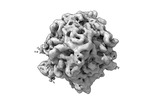




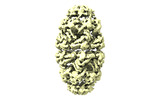

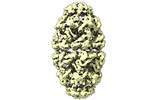










 Z
Z Y
Y X
X




























