[English] 日本語
 Yorodumi
Yorodumi- EMDB-12918: Cryo-EM structure of the Cetacean morbillivirus nucleoprotein-RNA... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-12918 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the Cetacean morbillivirus nucleoprotein-RNA complex | |||||||||
 Map data Map data | Masked postprocessed map | |||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords | Cetacean morbillivirus / nucleocapsid / RNA virus replication / cryo-electron microscopy / helical reconstruction / single particle analysis / VIRAL PROTEIN | |||||||||
| Function / homology | Paramyxovirus nucleocapsid protein / Paramyxovirus nucleocapsid protein / helical viral capsid / viral nucleocapsid / host cell cytoplasm / ribonucleoprotein complex / structural molecule activity / RNA binding / Nucleocapsid Function and homology information Function and homology information | |||||||||
| Biological species |  Morbillivirus sp. Morbillivirus sp. | |||||||||
| Method | helical reconstruction / cryo EM / Resolution: 4.0 Å | |||||||||
 Authors Authors | Zinzula L / Beck F | |||||||||
 Citation Citation |  Journal: J Struct Biol / Year: 2021 Journal: J Struct Biol / Year: 2021Title: Cryo-EM structure of the cetacean morbillivirus nucleoprotein-RNA complex. Authors: Luca Zinzula / Florian Beck / Sven Klumpe / Stefan Bohn / Günter Pfeifer / Daniel Bollschweiler / István Nagy / Jürgen M Plitzko / Wolfgang Baumeister /  Abstract: Cetacean morbillivirus (CeMV) is an emerging and highly infectious paramyxovirus that causes outbreaks in cetaceans and occasionally in pinnipeds, representing a major threat to biodiversity and ...Cetacean morbillivirus (CeMV) is an emerging and highly infectious paramyxovirus that causes outbreaks in cetaceans and occasionally in pinnipeds, representing a major threat to biodiversity and conservation of endangered marine mammal populations in both hemispheres. As for all non-segmented, negative-sense, single-stranded RNA (ssRNA) viruses, the morbilliviral genome is enwrapped by thousands of nucleoprotein (N) protomers. Each bound to six ribonucleotides, N protomers assemble to form a helical ribonucleoprotein (RNP) complex that serves as scaffold for nucleocapsid formation and as template for viral replication and transcription. While the molecular details on RNP complexes elucidated in human measles virus (MeV) served as paradigm model for these processes in all members of the Morbillivirus genus, no structural information has been obtained from other morbilliviruses, nor has any CeMV structure been solved so far. We report the structure of the CeMV RNP complex, reconstituted in vitro upon binding of recombinant CeMV N to poly-adenine ssRNA hexamers and solved to 4.0 Å resolution by cryo-electron microscopy. In spite of the amino acid sequence similarity and consequently similar folding of the N protomer, the CeMV RNP complex exhibits different helical parameters as compared to previously reported MeV orthologs. The CeMV structure reveals exclusive interactions leading to more extensive protomer-RNA and protomer-protomer interfaces. We identified twelve residues, among those varying between CeMV strains, as putatively important for the stabilization of the RNP complex, which highlights the need to study the potential of CeMV N mutations that modulate nucleocapsid assembly to also affect viral phenotype and host adaptation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_12918.map.gz emd_12918.map.gz | 9.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-12918-v30.xml emd-12918-v30.xml emd-12918.xml emd-12918.xml | 21.4 KB 21.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_12918_fsc.xml emd_12918_fsc.xml | 9.2 KB | Display |  FSC data file FSC data file |
| Images |  emd_12918.png emd_12918.png | 162.6 KB | ||
| Masks |  emd_12918_msk_1.map emd_12918_msk_1.map | 64 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-12918.cif.gz emd-12918.cif.gz | 5.8 KB | ||
| Others |  emd_12918_additional_1.map.gz emd_12918_additional_1.map.gz emd_12918_additional_2.map.gz emd_12918_additional_2.map.gz emd_12918_half_map_1.map.gz emd_12918_half_map_1.map.gz emd_12918_half_map_2.map.gz emd_12918_half_map_2.map.gz | 56.3 MB 49.5 MB 49.7 MB 49.8 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-12918 http://ftp.pdbj.org/pub/emdb/structures/EMD-12918 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12918 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-12918 | HTTPS FTP |
-Related structure data
| Related structure data |  7oi3MC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_12918.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_12918.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Masked postprocessed map | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.09 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
-Mask #1
| File |  emd_12918_msk_1.map emd_12918_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
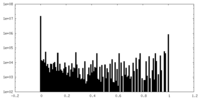
| Density Histograms |
-Additional map: DeepEMhancer map
| File | emd_12918_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | DeepEMhancer map | ||||||||||||
| Projections & Slices |
| ||||||||||||
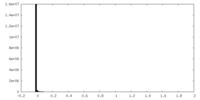
| Density Histograms |
-Additional map: Map before postprocessing
| File | emd_12918_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Map before postprocessing | ||||||||||||
| Projections & Slices |
| ||||||||||||
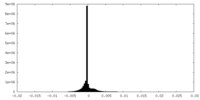
| Density Histograms |
-Half map: Half map 1
| File | emd_12918_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
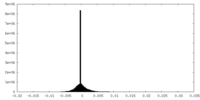
| Density Histograms |
-Half map: Half map 1
| File | emd_12918_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map 1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cetacean morbillivirus nucleoprotein-RNA complex
| Entire | Name: Cetacean morbillivirus nucleoprotein-RNA complex |
|---|---|
| Components |
|
-Supramolecule #1: Cetacean morbillivirus nucleoprotein-RNA complex
| Supramolecule | Name: Cetacean morbillivirus nucleoprotein-RNA complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|
-Supramolecule #2: Cetacean morbillivirus nucleoprotein
| Supramolecule | Name: Cetacean morbillivirus nucleoprotein / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  Morbillivirus sp. Morbillivirus sp. |
-Supramolecule #3: poly-A 6-mer
| Supramolecule | Name: poly-A 6-mer / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:  Morbillivirus sp. Morbillivirus sp. |
-Macromolecule #1: Cetacean morbillivirus nucleoprotein
| Macromolecule | Name: Cetacean morbillivirus nucleoprotein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Morbillivirus sp. Morbillivirus sp. |
| Molecular weight | Theoretical: 46.273625 KDa |
| Recombinant expression | Organism:  |
| Sequence | String: GMATLLRSLA LFKRNKDRTP LTAGSGGAIR GIKHVIVVPV PGDSSIVTRS RLLDRLVRLA GDPYISGPKL TGVMISILSL FVESPSQLI QRITDDPDVS IRLVEVIQSE KSLSGLTFAS RGANMEDEAD DYFSIQAGEE GDTRGTHWFE NKEIVEIEVQ D PEEFNILL ...String: GMATLLRSLA LFKRNKDRTP LTAGSGGAIR GIKHVIVVPV PGDSSIVTRS RLLDRLVRLA GDPYISGPKL TGVMISILSL FVESPSQLI QRITDDPDVS IRLVEVIQSE KSLSGLTFAS RGANMEDEAD DYFSIQAGEE GDTRGTHWFE NKEIVEIEVQ D PEEFNILL ASILAQIWIL LAKAVTAPDT AADSETRRWI KYTQQRRVVG EFRLDKGWLD AVRNRIAEDL SLRRFMVALI LD IKRTPGN KPRIAEMICD IDTYIVEAGL ASFILTIKFG IETMYPALGL HEFSGELTTV ESLMNLYQQM GETAPYMVIL ENS IQNKFS AGSYPLLWSY AMGVGVELEN SMGGLNFGRS YFDPAYFRLG QEMVRRSAGK VSSSLAAELG ITAEDAKLVS EIAA QANDD RVEHHHHHHH H UniProtKB: Nucleocapsid |
-Macromolecule #2: poly-A 6-mer
| Macromolecule | Name: poly-A 6-mer / type: rna / ID: 2 / Number of copies: 1 |
|---|---|
| Source (natural) | Organism:  Morbillivirus sp. Morbillivirus sp. |
| Molecular weight | Theoretical: 1.930277 KDa |
| Sequence | String: AAAAAA |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | helical reconstruction |
| Aggregation state | helical array |
- Sample preparation
Sample preparation
| Concentration | 0.8 mg/mL | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.4 Component:
| |||||||||
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Support film - Material: CARBON / Support film - topology: HOLEY / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 30 sec. | |||||||||
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 95 % / Chamber temperature: 4 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | TFS KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Number real images: 3153 / Average electron dose: 55.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 22500 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller











 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)