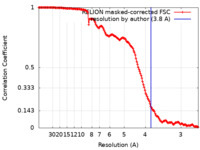
登録情報 データベース : EMDB / ID : EMD-11313タイトル Cryo-EM structure of the dynactin complex at 3.8 Angstrom resolution 複合体 : Dynactin complexリガンド : x 3種 / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Sus scrofa (ブタ)手法 / / 解像度 : 3.8 Å Lau CK / Lacey SE 資金援助 Organization Grant number 国 Medical Research Council (MRC, United Kingdom) MC_UP_A025_1011 Wellcome Trust WT210711
ジャーナル : EMBO J / 年 : 2021タイトル : Cryo-EM reveals the complex architecture of dynactin's shoulder region and pointed end.著者 : Clinton K Lau / Francis J O'Reilly / Balaji Santhanam / Samuel E Lacey / Juri Rappsilber / Andrew P Carter / 要旨 : Dynactin is a 1.1 MDa complex that activates the molecular motor dynein for ultra-processive transport along microtubules. In order to do this, it forms a tripartite complex with dynein and a coiled- ... Dynactin is a 1.1 MDa complex that activates the molecular motor dynein for ultra-processive transport along microtubules. In order to do this, it forms a tripartite complex with dynein and a coiled-coil adaptor. Dynactin consists of an actin-related filament whose length is defined by its flexible shoulder domain. Despite previous cryo-EM structures, the molecular architecture of the shoulder and pointed end of the filament is still poorly understood due to the lack of high-resolution information in these regions. Here we combine multiple cryo-EM datasets and define precise masking strategies for particle signal subtraction and 3D classification. This overcomes domain flexibility and results in high-resolution maps into which we can build the shoulder and pointed end. The unique architecture of the shoulder securely houses the p150 subunit and positions the four identical p50 subunits in different conformations to bind dynactin's filament. The pointed end map allows us to build the first structure of p62 and reveals the molecular basis for cargo adaptor binding to different sites at the pointed end. 履歴 登録 2020年7月6日 - ヘッダ(付随情報) 公開 2020年7月29日 - マップ公開 2020年7月29日 - 更新 2024年5月1日 - 現状 2024年5月1日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報
 データ登録者
データ登録者 英国, 2件
英国, 2件  引用
引用 ジャーナル: EMBO J / 年: 2021
ジャーナル: EMBO J / 年: 2021

 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_11313.map.gz
emd_11313.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-11313-v30.xml
emd-11313-v30.xml emd-11313.xml
emd-11313.xml EMDBヘッダ
EMDBヘッダ emd_11313_fsc.xml
emd_11313_fsc.xml FSCデータファイル
FSCデータファイル emd_11313.png
emd_11313.png emd_11313_msk_1.map
emd_11313_msk_1.map マスクマップ
マスクマップ emd-11313.cif.gz
emd-11313.cif.gz emd_11313_half_map_1.map.gz
emd_11313_half_map_1.map.gz emd_11313_half_map_2.map.gz
emd_11313_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-11313
http://ftp.pdbj.org/pub/emdb/structures/EMD-11313 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11313
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11313 emd_11313_validation.pdf.gz
emd_11313_validation.pdf.gz EMDB検証レポート
EMDB検証レポート emd_11313_full_validation.pdf.gz
emd_11313_full_validation.pdf.gz emd_11313_validation.xml.gz
emd_11313_validation.xml.gz emd_11313_validation.cif.gz
emd_11313_validation.cif.gz https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11313
https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11313 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11313
ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-11313 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_11313.map.gz / 形式: CCP4 / 大きさ: 307.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_11313.map.gz / 形式: CCP4 / 大きさ: 307.5 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_11313_msk_1.map
emd_11313_msk_1.map 試料の構成要素
試料の構成要素 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 画像解析
画像解析
 ムービー
ムービー コントローラー
コントローラー


























 Z
Z Y
Y X
X