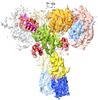
登録情報 データベース : EMDB / ID : EMD-10273タイトル human insulin receptor ectodomain bound by 4 insulin "manually filtered map" in paper, b-factor=-140 複合体 : human insulin receptor ectodomain bound to four insulin複合体 : human insulin receptor ectodomain bound to four insulinタンパク質・ペプチド : Insulin receptorタンパク質・ペプチド : Insulin receptor複合体 : human insulin receptor ectodomain bound to four insulinタンパク質・ペプチド : Insulinタンパク質・ペプチド : Insulin / / / / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
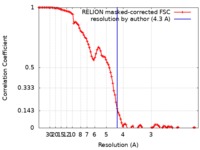
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 4.3 Å Gutmann T / Schaefer IB 資金援助 Organization Grant number 国 German Research Foundation 251981924TRR83 German Research Foundation 347368302 FOR2682 European Research Council CROWDED-PRO-LIPIDS Academy of Finland
ジャーナル : J Cell Biol / 年 : 2020タイトル : Cryo-EM structure of the complete and ligand-saturated insulin receptor ectodomain.
著者 :
Theresia Gutmann / Ingmar B Schäfer / Chetan Poojari / Beate Brankatschk / Ilpo Vattulainen / Mike Strauss / Ünal Coskun / 要旨 :
Glucose homeostasis and growth essentially depend on the hormone insulin engaging its receptor. Despite biochemical and structural advances, a fundamental contradiction has persisted in the current ... Glucose homeostasis and growth essentially depend on the hormone insulin engaging its receptor. Despite biochemical and structural advances, a fundamental contradiction has persisted in the current understanding of insulin ligand-receptor interactions. While biochemistry predicts two distinct insulin binding sites, 1 and 2, recent structural analyses have resolved only site 1. Using a combined approach of cryo-EM and atomistic molecular dynamics simulation, we present the structure of the entire dimeric insulin receptor ectodomain saturated with four insulin molecules. Complementing the previously described insulin-site 1 interaction, we present the first view of insulin bound to the discrete insulin receptor site 2. Insulin binding stabilizes the receptor ectodomain in a T-shaped conformation wherein the membrane-proximal domains converge and contact each other. These findings expand the current models of insulin binding to its receptor and of its regulation. In summary, we provide the structural basis for a comprehensive description of ligand-receptor interactions that ultimately will inform new approaches to structure-based drug design. 履歴 登録 2019年8月29日 - ヘッダ(付随情報) 公開 2019年11月13日 - マップ公開 2019年11月13日 - 更新 2024年10月23日 - 現状 2024年10月23日 処理サイト : PDBe / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報 マップデータ
マップデータ 試料
試料 キーワード
キーワード 機能・相同性情報
機能・相同性情報 Homo sapiens (ヒト)
Homo sapiens (ヒト) データ登録者
データ登録者 ドイツ,
ドイツ,  フィンランド, 4件
フィンランド, 4件  引用
引用 ジャーナル: J Cell Biol / 年: 2020
ジャーナル: J Cell Biol / 年: 2020


 ジャーナル: bioRxiv / 年: 2019
ジャーナル: bioRxiv / 年: 2019 構造の表示
構造の表示 ムービービューア
ムービービューア SurfView
SurfView Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク emd_10273.map.gz
emd_10273.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-10273-v30.xml
emd-10273-v30.xml emd-10273.xml
emd-10273.xml EMDBヘッダ
EMDBヘッダ emd_10273_fsc.xml
emd_10273_fsc.xml FSCデータファイル
FSCデータファイル emd_10273.png
emd_10273.png emd_10273_msk_1.map
emd_10273_msk_1.map マスクマップ
マスクマップ emd-10273.cif.gz
emd-10273.cif.gz emd_10273_additional.map.gz
emd_10273_additional.map.gz emd_10273_half_map_1.map.gz
emd_10273_half_map_1.map.gz emd_10273_half_map_2.map.gz
emd_10273_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-10273
http://ftp.pdbj.org/pub/emdb/structures/EMD-10273 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10273
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-10273 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_10273.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_10273.map.gz / 形式: CCP4 / 大きさ: 91.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) emd_10273_msk_1.map
emd_10273_msk_1.map 試料の構成要素
試料の構成要素 Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト) Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 解析
解析 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN
 ムービー
ムービー コントローラー
コントローラー

































 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)