[English] 日本語
 Yorodumi
Yorodumi- EMDB-0190: Cryo-EM structure of the ABCG2 E211Q mutant bound to ATP and Magnesium -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-0190 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of the ABCG2 E211Q mutant bound to ATP and Magnesium | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
| Function / homology |  Function and homology information Function and homology informationbiotin transmembrane transporter activity / biotin transport / riboflavin transport / riboflavin transmembrane transporter activity / sphingolipid transporter activity / external side of apical plasma membrane / renal urate salt excretion / urate transmembrane transporter activity / urate metabolic process / Abacavir transmembrane transport ...biotin transmembrane transporter activity / biotin transport / riboflavin transport / riboflavin transmembrane transporter activity / sphingolipid transporter activity / external side of apical plasma membrane / renal urate salt excretion / urate transmembrane transporter activity / urate metabolic process / Abacavir transmembrane transport / organic anion transport / sphingolipid biosynthetic process / Sphingolipid de novo biosynthesis / organic anion transmembrane transporter activity / xenobiotic transport across blood-brain barrier / transepithelial transport / export across plasma membrane / ABC-type xenobiotic transporter / Paracetamol ADME / ABC-type xenobiotic transporter activity / Ciprofloxacin ADME / NFE2L2 regulating MDR associated enzymes / Differentiation of keratinocytes in interfollicular epidermis in mammalian skin / cellular detoxification / Heme biosynthesis / Heme degradation / xenobiotic transmembrane transporter activity / efflux transmembrane transporter activity / transport across blood-brain barrier / ATPase-coupled transmembrane transporter activity / mitochondrial membrane / brush border membrane / Iron uptake and transport / transmembrane transport / apical plasma membrane / membrane raft / protein homodimerization activity / ATP hydrolysis activity / nucleoplasm / ATP binding / identical protein binding / plasma membrane Similarity search - Function | |||||||||
| Biological species |  HOMO SAPIENS (human) / HOMO SAPIENS (human) /  Homo sapiens (human) Homo sapiens (human) | |||||||||
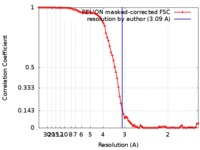
| Method | single particle reconstruction / cryo EM / Resolution: 3.09 Å | |||||||||
 Authors Authors | Manolaridis I / Jackson SM / Taylor NMI / Kowal J / Stahlberg H / Locher KP | |||||||||
| Funding support |  Switzerland, 1 items Switzerland, 1 items
| |||||||||
 Citation Citation |  Journal: Nature / Year: 2018 Journal: Nature / Year: 2018Title: Cryo-EM structures of a human ABCG2 mutant trapped in ATP-bound and substrate-bound states. Authors: Ioannis Manolaridis / Scott M Jackson / Nicholas M I Taylor / Julia Kowal / Henning Stahlberg / Kaspar P Locher /   Abstract: ABCG2 is a transporter protein of the ATP-binding-cassette (ABC) family that is expressed in the plasma membrane in cells of various tissues and tissue barriers, including the blood-brain, blood- ...ABCG2 is a transporter protein of the ATP-binding-cassette (ABC) family that is expressed in the plasma membrane in cells of various tissues and tissue barriers, including the blood-brain, blood-testis and maternal-fetal barriers. Powered by ATP, it translocates endogenous substrates, affects the pharmacokinetics of many drugs and protects against a wide array of xenobiotics, including anti-cancer drugs. Previous studies have revealed the architecture of ABCG2 and the structural basis of its inhibition by small molecules and antibodies. However, the mechanisms of substrate recognition and ATP-driven transport are unknown. Here we present high-resolution cryo-electron microscopy (cryo-EM) structures of human ABCG2 in a substrate-bound pre-translocation state and an ATP-bound post-translocation state. For both structures, we used a mutant containing a glutamine replacing the catalytic glutamate (ABCG2), which resulted in reduced ATPase and transport rates and facilitated conformational trapping for structural studies. In the substrate-bound state, a single molecule of estrone-3-sulfate (ES) is bound in a central, hydrophobic and cytoplasm-facing cavity about halfway across the membrane. Only one molecule of ES can bind in the observed binding mode. In the ATP-bound state, the substrate-binding cavity has collapsed while an external cavity has opened to the extracellular side of the membrane. The ATP-induced conformational changes include rigid-body shifts of the transmembrane domains, pivoting of the nucleotide-binding domains (NBDs), and a change in the relative orientation of the NBD subdomains. Mutagenesis and in vitro characterization of transport and ATPase activities demonstrate the roles of specific residues in substrate recognition, including a leucine residue that forms a 'plug' between the two cavities. Our results show how ABCG2 harnesses the energy of ATP binding to extrude ES and other substrates, and suggest that the size and binding affinity of compounds are important for distinguishing substrates from inhibitors. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_0190.map.gz emd_0190.map.gz | 43.4 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-0190-v30.xml emd-0190-v30.xml emd-0190.xml emd-0190.xml | 12.4 KB 12.4 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_0190_fsc.xml emd_0190_fsc.xml | 8.9 KB | Display |  FSC data file FSC data file |
| Images |  emd_0190.png emd_0190.png | 63.9 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-0190 http://ftp.pdbj.org/pub/emdb/structures/EMD-0190 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0190 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-0190 | HTTPS FTP |
-Validation report
| Summary document |  emd_0190_validation.pdf.gz emd_0190_validation.pdf.gz | 261.7 KB | Display |  EMDB validaton report EMDB validaton report |
|---|---|---|---|---|
| Full document |  emd_0190_full_validation.pdf.gz emd_0190_full_validation.pdf.gz | 260.8 KB | Display | |
| Data in XML |  emd_0190_validation.xml.gz emd_0190_validation.xml.gz | 10.8 KB | Display | |
| Arichive directory |  https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0190 https://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0190 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0190 ftp://ftp.pdbj.org/pub/emdb/validation_reports/EMD-0190 | HTTPS FTP |
-Related structure data
| Related structure data |  6hbuMC  6hzmMC  0196C  6hcoC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_0190.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_0190.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & slices | Image control
Images are generated by Spider. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 0.81 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : inhibitor-bound ABCG2
| Entire | Name: inhibitor-bound ABCG2 |
|---|---|
| Components |
|
-Supramolecule #1: inhibitor-bound ABCG2
| Supramolecule | Name: inhibitor-bound ABCG2 / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1 |
|---|
-Supramolecule #2: A mutant (E211Q) of the ATP-binding cassette sub-family G member 2
| Supramolecule | Name: A mutant (E211Q) of the ATP-binding cassette sub-family G member 2 type: complex / ID: 2 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:  HOMO SAPIENS (human) HOMO SAPIENS (human) |
| Recombinant expression | Organism:  HOMO SAPIENS (human) HOMO SAPIENS (human) |
-Macromolecule #1: ATP-binding cassette sub-family G member 2
| Macromolecule | Name: ATP-binding cassette sub-family G member 2 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 72.384867 KDa |
| Recombinant expression | Organism:  Homo sapiens (human) Homo sapiens (human) |
| Sequence | String: MSSSNVEVFI PVSQGNTNGF PATASNDLKA FTEGAVLSFH NICYRVKLKS GFLPCRKPVE KEILSNINGI MKPGLNAILG PTGGGKSSL LDVLAARKDP SGLSGDVLIN GAPRPANFKC NSGYVVQDDV VMGTLTVREN LQFSAALRLA TTMTNHEKNE R INRVIQEL ...String: MSSSNVEVFI PVSQGNTNGF PATASNDLKA FTEGAVLSFH NICYRVKLKS GFLPCRKPVE KEILSNINGI MKPGLNAILG PTGGGKSSL LDVLAARKDP SGLSGDVLIN GAPRPANFKC NSGYVVQDDV VMGTLTVREN LQFSAALRLA TTMTNHEKNE R INRVIQEL GLDKVADSKV GTQFIRGVSG GERKRTSIGM ELITDPSILF LDQPTTGLDS STANAVLLLL KRMSKQGRTI IF SIHQPRY SIFKLFDSLT LLASGRLMFH GPAQEALGYF ESAGYHCEAY NNPADFFLDI INGDSTAVAL NREEDFKATE IIE PSKQDK PLIEKLAEIY VNSSFYKETK AELHQLSGGE KKKKITVFKE ISYTTSFCHQ LRWVSKRSFK NLLGNPQASI AQII VTVVL GLVIGAIYFG LKNDSTGIQN RAGVLFFLTT NQCFSSVSAV ELFVVEKKLF IHEYISGYYR VSSYFLGKLL SDLLP MRML PSIIFTCIVY FMLGLKPKAD AFFVMMFTLM MVAYSASSMA LAIAAGQSVV SVATLLMTIC FVFMMIFSGL LVNLTT IAS WLSWLQYFSI PRYGFTALQH NEFLGQNFCP GLNATGNNPC NYATCTGEEY LVKQGIDLSP WGLWKNHVAL ACMIVIF LT IAYLKLLFLK KYS |
-Macromolecule #2: ADENOSINE-5'-TRIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-TRIPHOSPHATE / type: ligand / ID: 2 / Number of copies: 2 / Formula: ATP |
|---|---|
| Molecular weight | Theoretical: 507.181 Da |
| Chemical component information |  ChemComp-ATP: |
-Macromolecule #3: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 3 / Number of copies: 2 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method | cryo EM |
|---|---|
 Processing Processing | single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Mesh: 300 / Pretreatment - Type: GLOW DISCHARGE |
| Vitrification | Cryogen name: ETHANE-PROPANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Number real images: 4905 / Average exposure time: 0.2 sec. / Average electron dose: 2.0 e/Å2 |
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD / Nominal magnification: 165000 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller

















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)